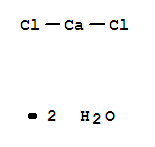
What is the chemical formula for calcium chloride?
Calcium chloride is commonly encountered as a hydrated solid with generic formula CaCl 2 (H 2 O) x, where x = 0, 1, 2, 4, and 6. These compounds are mainly used for de-icing and dust control.
How many atoms are in a mole of calcium chloride?
Expert Answers. One mole of calcium reacts with one mole of chlorine gas to give one mole of calcium chloride. One mole of a substance contains 6.023*10^23 formula units. The molar mass of calcium is 40 g/mole. 5.3 g of calcium contains 0.1325 moles of calcium. This is equivalent to 0.1325*6.023*10^23 ~~ 7.98*10^22 atoms of calcium.
Is calcium chloride ionic or covalent?
It is an ionic compound that comprises two ions i.e calcium and chlorine, (Calcium and Chlorine formula are Ca²⁺ and Cl⁻ respectively). The chemical formula for calcium chloride is CaCl₂. This inorganic compound is a white-colored crystalline solid, odorless, and is highly soluble in water.
What is the molar mass of CaCl2?
Question: Calculate the Molar Mass of CaCl₂ Answer: CaCl₂ chemical name is Calcium Chloride and the molar mass of Calcium is 40.08 grams and molar mass of Chlorine is 35.45 grams. So the molar mass of Calcium Chloride can be calculated as: 40.08 + 2 x 35.45
Question: Write the Chemical Formula of Calcium Chloride and Its Properties.
Answer: The chemical formula for calcium chloride is CaCl₂. Properties: It is water-soluble, hygroscopic, and deliquescent. It absorbs enough water...
Question: Calculate the Molar Mass of CaCl₂
Answer: CaCl₂ chemical name is Calcium Chloride and the molar mass of Calcium is 40.08 grams and molar mass of Chlorine is 35.45 grams. So the mola...
Question: What Happens When Anhydrous CaCl₂ is Exposed to Air for Some Time.
Answer: CaCl₂ compound name is Calcium Chloride, It will absorb a sufficient amount of water from the air to allow it to dissolve. Anhydrous calciu...
What is the formula for calcium chloride?
Answer: The chemical formula for calcium chloride is CaCl₂.
Why is calcium chloride used in food?
Due to its ability to absorb moisture, Calcium chloride is often found as a firming agent in foods.
Is calcium chloride anhydrous or ionic?
In this topic, we will be learning about Calcium chloride, Calcium chloride anhydrous, or Calcium dichloride. It is an ionic compound that comprises two ions i.e calcium and chlorine, (Calcium and Chlorine formula are Ca²⁺ and Cl⁻ respectively). The chemical formula for calcium chloride is CaCl₂. This inorganic compound is a white-colored crystalline solid, odorless, and is highly soluble in water.
What is the formula used to calculate molar mass?
If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight.
How to find molar mass?
Finding molar mass starts with units of grams per mole (g/mol). When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula.
How to find the formula weight?
In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.