What is the concentration of alcohol in a water solution?
In fact, a water/alcohol solution with a alcohol concentration of 95.6% alcohol sees the number of A-A molecules becoming so high that the alcohol becomes as reluctant as water molecules to vaporize of, in fact they will have the same volatility and no more separation or concentration of the alcohol becomes possible by standard distillation.
What is the boiling point of ethanol and water?
Ethanol and water form a constant boiling mixture at 95.6%v/v EtOH with a boiling of 78.2 o C, a temperature near the boiling point of pure ethanol (78.5 o C) and far away from that of water (100 o C) at atmospheric pressure (101.325 kPa).
What is alcohol distillation and how does it work?
Alcoholic distillation is basically the process of separation the more volatile component, alcohol, from the less volatile component, water, from a water/alcohol solution, by heating the solution and condensing and collecting the alcohol rich vapours released, as a high alcohol strength liquid (spirit).
What is the starting material of a distillate?
The starting material for most distillations, such as wine for brandy and beer for whisky, are usually weak alcoholic solutions (5%-7% v/v ethanol) This seems counter intuitive, as it would seem that if the desired product is alcohol, the starting solution should have a high alcohol concentration.

What is the maximum concentration of ethanol in water by simple distillation?
Because the azeotropic mixture boils at a lower temperature, it's impossible to use simple distillation to produce ethanol at concentrations higher than 95.6%.
What is the maximum concentration of ethanol that can be produced by fractional distillation?
The highest concentration of ethanol by volume that can be reached using fractional distillation is 96% which yields 192 proof solution.
Why is fractional distillation better for ethanol and water?
For example, liquid ethanol can be separated from a mixture of ethanol and water by fractional distillation. This method works because the liquids in the mixture have different boiling points.
Is simple or fractional distillation better for ethanol?
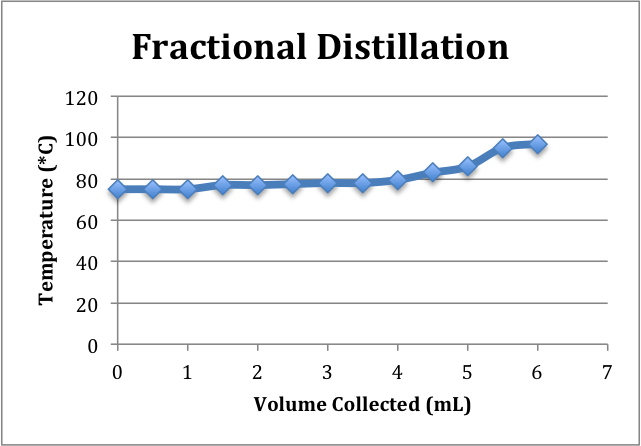
To obtain the highest percent ethanol, fraction distillation is proven to be the most effective. Because the 2 liquids have a boiling point difference of about 25 degrees Celsius, we used both simple and fractional distillation to remove the ethanol from the water.
What is an azeotrope and why does it limit ethanol purity even with fractional distillation?
An azeotrope is a mixture of two liquids with constant composition and boiling point throughout a distillation process. It limits ethanol purity even without fractional distillation as it prevents ethanol from being fully purified.
Is simple distillation effective in separating ethanol from its aqueous mixture?
Azeotropic Mixtures These are called azeotropes. Ethanol is perhaps the most often-cited example; a mixture of 95.6 percent ethanol and 4.4 percent water will actually boil at a lower temperature than either ethanol or water. Consequently, simple distillation cannot change this mixture's composition.
Why is it not possible to separate a mixture of ethanol and water by fractional distillation?
It is impossible to get pure ethanol by distilling any mixture of ethanol and water containing less than 95.6% of ethanol. This particular mixture of ethanol and water boils as if it were a pure liquid. It has a constant boiling point, and the vapor composition is exactly the same as the liquid.
Why is fractional distillation more efficient than simple distillation?
Fractional distillation leads to a better separation than simple distillation because the glass beads in the fractionating column provide "theoretical plates" on which the vapors can condense and then re-evaporate, and re-condense, essentially distilling the compound many times over.
How do you separate ethanol and water using fractional distillation?
When the mixture is heated in a distillation flask, ethanol which has a low boiling point (78oC) than water (100oC) boils faster. Hence ethanol vapor passes into the condenser (the region where vapor is cooled and condensed), collected in a beaker, and hence ethanol gets separated from the mixture.
What method of separation can be used to separate ethanol and water?
Fractional distillationFractional distillation Fractional distillation is used for separating two liquids that are mixed together. Some liquids mix together like ethanol and water and these are called miscible.
Whats the difference between simple distillation and fractional distillation?
Summary: 1. Simple distillation is the method used to separate substances in mixtures with significantly different boiling points, while fractional distillation is used for mixtures containing chemicals with boiling points close to each other.
Is simple distillation and fractional distillation the same?
Simple distillation is used to separate substances in mixtures with widely disparate boiling points, whereas fractional distillation is used for mixtures containing chemicals with similar boiling points.
Why the concentration of ethanol produced by fermentation does not exceed 15?
After sugarcane juice fermentation by yeast, ethanol concentration in the fermented broth is no more than 10% v/v (due to its toxicity). During distillation, the ethanol is recovered and everything left is called vinasse. It is produced in high volumes (12–15 L for each liter of ethanol) and is rich in minerals [8].
What is the concentration of ethanol?
The US and many other countries primarily use E10 (10% ethanol, sometimes known as gasohol) and E85 (85% ethanol) ethanol/gasoline mixtures.
How do you find the concentration of ethanol?
To find out the concentration of ethanol, we need to know the volume of the fermentation medium. If it was 500 ml (0.5 liters), then the concentration is 0.021 moles/0.5 liters = 0.042 moles/liter (or 42 mM) ethanol.
How can the concentration of ethanol be increased?
0:154:33How can we make Ethanol more concentrated? GCSE ... - YouTubeYouTubeStart of suggested clipEnd of suggested clipThe answer is distillation which hopefully you have heard of before. Now there are two types ofMoreThe answer is distillation which hopefully you have heard of before. Now there are two types of distillation. Simple distillation and fractional distillation.
What is the boiling point of ethanol?
Ethanol and water form a constant boiling mixture at 95.6%v/v EtOH with a boiling of 78.2 o C , a temperature near the boiling point of pure ethanol (78.5 o C) and far away from that of water (100 o C) at atmospheric pressure (101.325 kPa).
What is the process of distilling alcohol?
Alcoholic distillation is basically the process of separation the more volatile component, alcohol, from the less volatile component, water, from a water/alcohol solution, by heating the solution and condensing and collecting the alcohol rich vapours released, as a high alcohol strength liquid (spirit).
What is a batch distillation?
Batch distillation and fractional distillation. Pot or batch distillation. A pot still consists of a pot and an outlet to a condenser. The outlet itself also serves as a condenser and to some extent as a fractionating column ( see fractional distillation ). Batch distillation as the name suggest starts with a single batch ...
What can be removed from an alcohol solution by distillation?
Further specialized distillations can remove components with similar volatilities such as methanol from an alcohol solution.
What is the phenomenon of volatilities of substances deviating from that predicted by Raoult's law?
The phenomenon, where volatilities of substances deviate from that predicted by Raoult's law, is refered to as non-ideal behaviour, or deviation from ideality, which happens to be a positive deviation with regards to a water/alcohol solution.
What is the starting material for most distillations?
The starting material for most distillations, such as wine for brandy and beer for whisky, are usually weak alcoholic solutions (5%-7% v/v ethanol)
Is wine ethanol or ethanol?
So far we have assumed that the only alcohol present in wine is ethanol, however there many other important, if minor quantities of other alcohols present. ( see other alcohol) In a batch/pot distillation, the alcoholic vapours coming of the bulk of the liquid are condensed above the liquid on the still outlet, on the way to the condenser.
