Electronegativity is a chemical property that measures the tendency of an atom to attract electrons towards itself. Electronegativity is affected by the atomic number and the distance between the valence electrons and its nucleus.
What is electronegativity and how does it work?
Electronegativity is the property of an atom which increases with its tendency to attract the electrons of a bond. If two bonded atoms have the same electronegativity values as each other, they share electrons equally in a covalent bond. Usually, the electrons in a chemical bond are more attracted to one atom (the more electronegative one) than to the other.
How do you explain electronegativity?
- An atom's electronegativity gets higher as you move to the right in the periodic table.
- An atom's electronegativity gets higher as you move up in the periodic table.
- Thus, the atoms in the top right have the highest electronegativities and the atoms in the bottom left have the lowest ones.
What is the meant by electronegativity?
Electronegativity refers to the ability of an atom to attract shared electrons in a covalent bond. The higher the value of the electronegativity, the more strongly that element attracts the shared electrons.
What is the formula for electronegativity?
When you're using kJ/mol as units for your energies, the equation for Mulliken electronegativity is ENMulliken = (1.97×10−3) (Ei+Eea) + 0.19. Plug your values into the equation and solve for EN Mulliken . EN Mulliken = (1.97×10 −3 ) (E i +E ea) + 0.19

Which is the best definition of electronegativity?
Electronegativity is a function of an atom’s ability to attract an electrons binding pair. The most frequently used is the Pauling scale. Fluorine...
What is high electronegativity?
Electronegativity decrease as it moves from top to bottom and increases over time from left to right. The most electronegative element is, therefor...
What is the electronegativity difference?
The degree to which an atom attracts electrons in a chemical bond is described by electronegativity. If the difference in electronegativity is grea...
What is the difference between electron affinity and electronegativity?
The difference between the two is that electronegativity is a chemical property that shows how well an atom can attract electrons to itself as the...
Is electronegativity a relative quantity?
Electronegativity is an example of an atom’s ability to attract electrons. It is proportional to the difference between the potential for ionizatio...
How does electronegative vary along the period?
Electronegativity increases as we move left to the right in the period because as we move across the period, the effective nuclear charge increases...
How does electronegative vary in a group?
Electronegativity decreases as we move down the group because as we move down the group, the atomic size increases and the effective nuclear charge...
Name the most electronegative element and least electronegative element in the periodic table?
Fluorine is the most electronegative element, and caesium is the least electronegative element in the periodic table.
How does the electronegativity of an element affect its bonding?
The electronegativity of an element affects the bonding of an element. Elements with high electronegativity tend to form ionic bonds with other ele...
Which atom has the highest electronegativity?
The chlorine atom has a higher electronegativity than the hydrogen atom, so the bonding electrons will be closer to the Cl than to the H in the HCl molecule.
What are the factors that affect electronegativity?
Factors that affect electronegativity include the nuclear charge and the number and location of electrons in an atom.
What happens when electrons are attracted to one atom?
Usually, the electrons in a chemical bond are more attracted to one atom (the more electronegative one) than to the other. This results in a polar covalent bond. If the electronegativity values are very different, the electrons aren't shared at all.
What is the property of an atom that increases with its tendency to attract the electrons of a bond?
Electronegativity is the property of an atom which increases with its tendency to attract the electrons of a bond. If two bonded atoms have the same electronegativity values as each other, they share electrons equally in a covalent bond. Usually, the electrons in a chemical bond are more attracted to one atom (the more electronegative one) ...
What is the tendency of an atom to attract electrons to itself in a chemical bond?
Electronegativity is an atom's tendency to attract electrons to itself in a chemical bond.
Which chemists studied the electronegativity of atoms?
The greater the difference between atom electronegativity values, the more polar the chemical bond formed between them. Avogadro and other chemists studied electronegativity before it was formally named by Jöns Jacob Berzelius in 1811. In 1932, Linus Pauling proposed an electronegativity scale based on bond energies.
Which element is the least electronegative?
The most electronegative element on the periodic table is fluorine (3.98). The least electronegative element is cesium (0.79). The opposite of electronegativity is electropositivity, so you could simply say cesium is the most electropositive element.
How does electronegativity decrease down the group?
Electronegativity decreases down the group. Moving down a group, the number of energy shells also increases with the increase of protons and electrons. So because of shielding effects of electrons in the increased inner shells , the attraction between electrons and nucleus (electronegativity) is reduced down the group.
Which element is the least electronegative?
According to the scale Fluorine is the most electronegative element and its value is 4.0. Caesium and Francium are the least electronegative element and its value is 0.7.
What type of bond is formed when two atoms with a greater difference in electronegativity bonded together?
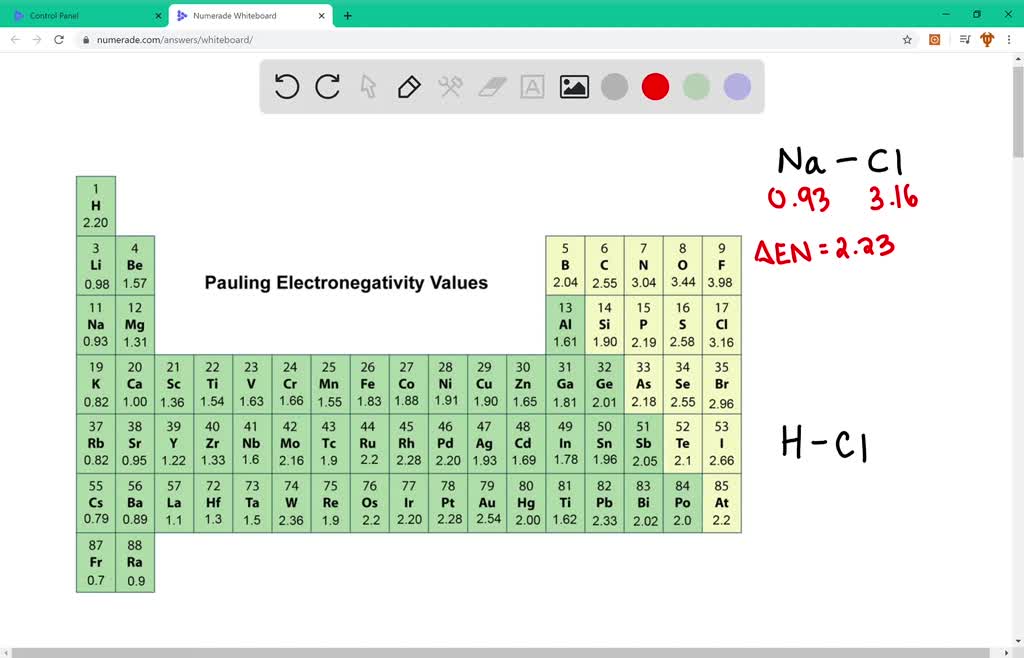
Ionic bond. When two atoms with a greater difference in electronegativity bonded together, then ionic bond is formed. Here one electron is transferred from less electronegative atom to more electronegative atom to form this type of bond. e.g. NaCl.
What is the property of an atom that measures the tendency of an atom to attract electrons towards itself?
Electronegativity is a chemical property that measures the tendency of an atom to attract electrons towards itself. Electronegativity is affected by the atomic number and the distance between the valence electrons and its nucleus. The higher the electronegativity is the more it attracts the electrons towards it.
When two atoms with a slight difference in electronegativities are bonded together, a polar?
When two atoms with a slight difference in electronegativities are bonded together, a polar bond is formed. The atom with more electronegativities attracts electron more than the other atom towards itself to form this type of bond. e.g. HCl, HF etc.
Which element has less electronegativity than boron?
So, for example, the electronegativity of boron is more than beryllium. Again it decreases down the period, so the electronegativity of aluminium is less than boron. Thus berylium and aluminium both have less electronegativity than boron.
When two atoms with equal electronegativity are bonded together in a molecule, a non?
When two atoms with equal electronegativity are bonded together in a molecule, a non polar bond is formed. These two atoms attracts bonding electrons equally towards themselves to form this sort of bond. It is more likely to happen in a molecule composed of same atoms. e.g. H 2, N 2 etc.
