Cholesterol Absorption inhibitors. Ezetimibe, a cholesterol absorption inhibitor, is the first agent of a new class of lipid-lowering compounds that selectively inhibits the intestinal absorption of cholesterol and related phytosterols. Ezetimibe undergoes extensive glucuronidation to an active metabolite in the intestinal mucosa [11].
What are cholesterol absorption inhibitors?
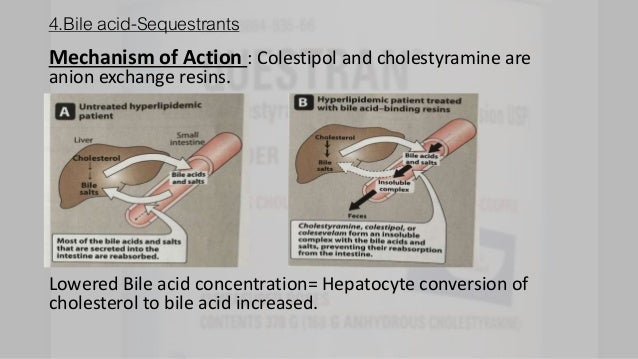
Cholesterol absorption inhibitors. What are Cholesterol absorption inhibitors? Cholesterol absorption inhibitors reduce the absorption of dietary and biliary cholesterol through the intestines. Therefore it deceases the amount of intestinal cholesterol that is delivered to the liver.
What happens to the amount of intestinal cholesterol delivered to the liver?
Therefore it deceases the amount of intestinal cholesterol that is delivered to the liver. Reduced levels of cholesterol delivered to the liver results in increased hepatic LDL (low density lipoprotein) receptor activity, which leads to increased clearance of LDL cholesterol.
What is the mechanism of action of ezetimibe in the treatment of cholesterol?
The cholesterol absorption inhibitor ezetimibe acts by blocking the sterol-induced internalization of NPC1L1. Cell Metab. 2008;7:508–519. [PubMed] [Google Scholar] 42. Santosa S, Varady KA, AbuMweis S, Jones PJH.
What is the role of liver hepatocytes in cholesterol regulation?
1 Sterling Research Group, Cincinnati, OH. 2 Department of Cardiology, University of Chicago Medicine, Chicago, IL. Low-density lipoprotein (LDL) receptors on the surface of liver hepatocytes are the primary way that humans regulate serum LDL cholesterol levels.
What drug inhibits the absorption of cholesterol?
Ezetimibe is the first agent of a novel class of selective cholesterol absorption inhibitors recently approved by the Food and Drug Administration for treatment in the United States.
What is the mechanism of action of statin drugs?
Mechanism of Action Statins work by competitively blocking the active site of the first and key rate-limiting enzyme in the mevalonate pathway, HMG-CoA reductase. Inhibition of this site prevents substrate access, thereby blocking the conversion of HMG-CoA to mevalonic acid.
How do statins inhibit cholesterol synthesis?
In vitro statins act indeed as competitive inhibitors of HMG-CoA reductase (HMGCR), the rate-limiting enzyme of cholesterol synthesis. When statins are bound to HMGCR, they block the access of the natural substrate HMG-CoA to the catalytic site and thereby interfere with cholesterol synthesis (4).
How does cholesterol medication work?
Statin drugs lower LDL cholesterol by slowing down the liver's production of cholesterol. They also increase the liver's ability to remove LDL cholesterol that is already in the blood. The Food and Drug Administration (FDA) offers advice on the risks related to taking statins: Controlling Cholesterol with Statins.
What is the mechanism of action of statins HMG-CoA reductase inhibitors?
Also known as statins, HMG-CoA reductase inhibitors work by inhibiting the synthesis of cholesterol in the liver by the enzyme HMG-CoA reductase.
How does statin work with the liver against cholesterol?
How do statins work? Statins work by slowing down the production of LDL-cholesterol in the liver, where it's made. Because the liver isn't making so much cholesterol, it then takes cholesterol out of your blood to make bile with, so your blood cholesterol levels fall.
What is the effect of a statin?
inflammation of the liver (hepatitis), which can cause flu-like symptoms. inflammation of the pancreas (pancreatitis), which can cause stomach pain. skin problems, such as acne or an itchy red rash. sexual problems, such as loss of libido (reduced sex drive) or erectile dysfunction.
How do statins work HMG-CoA reductase?
Statins act by inhibiting 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase and thereby reducing cholesterol synthesis. In X-ray crystallographic studies, we have determined the structures of the catalytic portions of the enzyme in complex with statin molecules.
What are Cholesterol absorption inhibitors?
Cholesterol absorption inhibitors reduce the absorption of dietary and biliary cholesterol through the intestines. Therefore it deceases the amount of intestinal cholesterol that is delivered to the liver.
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
How does statin therapy affect cholesterol levels?
Serum cholesterol levels are regulated based upon an interactive relationship between hepatic cholesterol production and intestinal cholesterol absorption.42Statin therapy reduces serum LDL-C by inhibiting hepatic cholesterol production through inhibition of the rate-limiting step in cholesterol synthesis catalyzed by HMG-CoA reductase.26In response to the decrease in hepatic cholesterol production, the liver upregulates hepatic LDL receptors, leading to an increase in LDL-C removal from the blood. Additionally, studies have shown that in response to statin treatment, there is a compensatory increase in intestinal cholesterol absorption, possibly through the induction of gene expression of such proteins such as NPC1L1.26,43As a corollary, increases in intestinal absorption can lead to downregulation of intrinsic hepatic cholesterol production.42,44
How do lipids enter the biliary micelles?
Notes:Cholesterol, phytosterols and other lipids enter biliary micelles and are internalized via the NPC1L1 protein and the AP2/clathrin complex. Cholesterol can also reenter hepatocytes from the bile via canalicular NPC1L1. Nearly all phytosterols and some cholesterol are returned to the gut lumen or to the bile from hepatocytes via ABCG5 and ABCG8. Cholesterol can be converted in hepatocytes to primary bile acids, which are effluxed to the bile via ABCB11. ApoA-I is secreted by the liver or enterocyte or enters plasma on a chylomicron. VLDLs and chylomicrons traffic TG to muscle and adipocyte tissues. During lipolysis, surface phospholipids as well as fatty acids from the TG are also released: the resultant chylomicron remnant is cleared by the liver and the VLDL is converted to IDL, most of which are cleared by LDL receptors in the liver but some undergo additional lipolysis and form LDLs. During their plasma residence time the ApoB particles utilizing CETP exchange TG for CE with HDL particles. Smaller HDLs are lipidated at ABCA1, and larger HDLs at ABCG1 or SR B1. Larger HDLs can be delipidated by SR B1 at the liver, on adipocytes, or in steroidogenic tissues.
Where does cholesterol come from?
Circulating plasma levels of cholesterol are derived from two primary sources: cholesterol production from the liver and peripheral tissues , and the absorption of dietary and biliary cholesterol in the gastrointestinal tract (Figure 1).24,25Cholesterol synthesis begins with the conversion of acetyl-CoA to mevalonate, a reaction catalyzed by the enzyme HMG-CoA reductase.26Cholesterol synthesized by hepatocytes undergoes esterification by acyl-CoA acyl transferase (ACAT) and is incorporated into apolipoprotein B (ApoB)-containing lipoproteins such as very-low-density lipoprotein (VLDL) via microsomal transfer protein.27,28Subsequent modification of VLDL with hydrolysis of triglycerides by the enzymes lipoprotein lipase and hepatic lipase produces intermediate-density lipoprotein (IDL) and LDL. The transfer of cholesterol from the peripheral tissues to the liver is mediated by HDL.29Nascent pre-β HDL particles accept free cholesterol from peripheral tissues via ATP-binding cassette transporter A1 (ABCA1). The cholesterol undergoes subsequent esterification by lecithin– cholesterol acetyltransferase. The esterified cholesterol moves into the hydrophobic core of the HDL particle, and as the particles become progressively more lipidated, they mature and become progressively larger and more spherical. The cholesteryl esters in these mature HDL particles can be removed from the circulation by hepatic scavenger receptor BI or undergo transfer of cholesterol to apolipoprotein B-containing lipoproteins such as LDL and IDL via the activity of cholesteryl ester transfer protein.30The liver clears LDL particles from the circulation by the LDL receptor and the LDL receptor-related protein.27,28
Where does cholesterol enter the body?
Intestinal cholesterol absorption, occurring primarily in the duodenum and proximal jejunum , can also contribute to serum cholesterol levels.31,32Dietary intake provides about a quarter of the cholesterol entering the intestinal lumen, while the remaining three-quarters are derived from biliary cholesterol excretion from the liver. A distinction must be drawn between cholesterol entry into enterocytes and systemic cholesterol absorption, which refers to the appearance of cholesterol within lymphatic vessels, as not all of the cholesterol that makes its way into enterocytes will be absorbed into plasma. Intestinal cholesterol absorption is a complex process involving incorporation of free cholesterol, the majority of which is of biliary origin, into mixed biliary micelles, and the subsequent delipidation of micelles via intestinal enterocyte membrane sterol influx transporters.33Once in the enterocyte, free cholesterol can be effluxed to ApoA-1, prebeta HDL, or ApoE, esterified by ACAT into cholesteryl ester for incorporation into ApoB48-containing chylomicrons, or effluxed back to the gut lumen by ABC transporters G5 and G8. Genetic mutations in ABCG5 and ABCG8 proteins result in sitosterolemia, which is associated with an increase in phytosterol accumulation and intestinal cholesterol absorption resulting in significantly elevated plasma cholesterol and plant sterol levels and clinical development of early atherosclerotic heart disease.34–36After secretion into the lymphatic system and drainage via the thoracic duct, chylomicrons and their remnants are cleared from the circulation by the liver. The triglycerides and cholesterol esters derived from chylomicrons can be repackaged into VLDL and secreted.
Does ezetimibe help with atherosclerosis?
A positive impact on carotid atherosclerosis using ezetimibe was observed in the Stop Athero sclerosis in Native Diabetics Study (SANDS).94,100SANDS randomized diabetic subjects to aggressive care with target LDL-C <70 mg/dL and systolic blood pressure <115 mmHg or to standard care with target LDL-C < 100 mg/dL and systolic blood pressure < 130 mmHg. Ezetimibe was added on to statin therapy in subjects not able to meet LDL-C targets. Change in carotid IMT was compared between the aggressive versus standard treatment groups and between subjects receiving statins plus ezetimibe versus statins alone. After 36 months of therapy, LDL-C was reduced similarly in the aggressive treatment group receiving statins plus ezetimibe (−31 mg/dL) or statins alone (−32 mg/dL). Mean baseline CIMT in SANDS was 0.81 mm as compared to 0.69 mm seen in ENHANCE. In the standard therapy group, there was progression of CIMT by +0.039 mm, while the aggressively treated group showed CIMT regression from baseline in both the ezetimibe (−0.025 mm) and nonezetimibe (−0.012 mm) subjects. In multivariate analysis, change in CIMT was related to degree of LDL-C reduction independent of specific choice of lipid-lowering therapy.
Does ezetimibe lower cholesterol?
Ezetimibe inhibits intestinal and biliary cholesterol absorption and can significantly lower LDL-C and nonhigh-density lipoprotein cholesterol (non-HDL-C, defined as total cholesterol minus high-density lipoprotein cholesterol) when used alone or in combination with statin therapy.22Despite the established cholesterol-lowering benefits of ezetimibe, significant controversy exists with respect to ezetimibe’s vascular and clinical benefit, particularly in light of the Ezetimibe and Simvastatin in Hypercholesterolemic Enhances Atherosclerosis Regression (ENHANCE) trial, which showed no difference in carotid atherosclerosis burden as measured by carotid intima-media thickness (CIMT) in patients with heterozygous familial hypercholesterolemia who were treated with simvastatin plus either ezetimibe or placebo.17,23Based upon this controversy, some providers eliminated or reserved the use of ezetimibe as a last-line agent in lipid management. This review aims to detail the biological mechanisms, lipid effects, and safety of ezetimibe treatment and discuss the vascular and clinical outcomes data that may impact the use of ezetimibe in clinical practice.
Can statins lower LDL-C?
Despite growing evidence supporting a lower-is-better approach for LDL-C, treatment with statin therapy alone may not be sufficient to achieve optimal LDL-C targets, with some patients requiring greater than a 50% reduction.13Institutional surveys have shown that only two-thirds of vascular disease patients are at an LDL-C goal of <100 mg/dL and less than a third of very high-risk patients are able to reach an LDL-C goal of <70 mg/dL.14,15Based upon these treatment failures, combination therapies using multiple cholesterol-lowering agents including ezetimibe in addition to statin therapy have been investigated.16,17While ATP III recommends statin therapy as the first-line agent for the treatment of elevated LDL-C, alternative therapies such as ezetimibe, niacin, bile-acid sequestrants, and ileal bypass surgery can also effectively lower LDL-C.17–20A recent meta-analysis has shown that these nonstatin-based treatments can lower cardiac events similar to statin therapies, with an equivalent observed relationship between degree of LDL-C lowering and reduction in coronary heart disease (CHD) risk.21These data suggest that the addition of these therapies to a background of statin treatment may produce an incremental lowering of LDL-C, and possibly result in a further reduction in cardiovascular events.
What is the primary way that humans regulate LDL cholesterol levels?
Low-density lipoprotein (LDL) receptors on the surface of liver hepatocytes are the primary way that humans regulate serum LDL cholesterol levels. Proprotein convertase subtilisin/kexin type 9 (PCSK9) is a proteolytic enzyme that indirectly regulates serum LDL cholesterol (LDL-C) by causing the destruction of LDL receptors. Less LDL receptors result in increased LDL-C in the bloodstream but inhibiting or binding the circulating PCSK9 results in increased LDL receptors with the resultant decrease in serum LDL-C. Two PCSK9 inhibitors are currently approved for use: alirocumab and evolocumab. Both are fully human monoclonal antibodies that bind free PCSK9. Herein we discuss the mechanism of action, efficacy, and safety of PCSK9 inhibitors. clinical problem.
What is the mechanism of action of PCSK9?
PCSK9 Inhibitors: Mechanism of Action, Efficacy, and Safety. Low-density lipoprotein (LDL) receptors on the surface of liver hepatocytes are the primary way that humans regulate serum LDL cholesterol levels.
Does PCSK9 increase LDL?
Less LDL receptors result in increased LDL-C in the bloodstream but inhibiting or binding the circulating PCSK9 results in increased LDL receptors with the resultant decrease in serum LDL-C. Two PCSK9 inhibitors are currently approved for use: alirocumab and evolocumab. Both are fully human monoclonal antibodies that bind free PCSK9.