What are the four properties of nonmetals?
Physical Properties of Non-metals
- Malleability and Ductility. Non-metals are generally non-malleable and non-ductile. ...
- Thermal and Electrical Conductivity. Non-metals are generally poor conductors of heat and electricity. ...
- Lustre. ...
- Tensile Strength. ...
- Density. ...
- Melting and Boiling Points. ...
- Hardness. ...
- Sonority. ...
- Electronic Configuration. ...
- Electronegative Character. ...
Which elements are non metals?
Elements that come under non-metals are sulphur, carbon, all halogens, phosphorus, hydrogen, oxygen, selenium, nitrogen and noble gases. In the periodic table, non-metals are located left of the halogens and to the right of the metalloids.
Which groups represent nonmetals?
Group VIIAGroup VIIA is the only group in periodic table in which all the elements are nonmetals. This group contains F, Cl, Br, I and At. What are the 22 nonmetals in periodic table? Chemically, hydrogen, carbon, nitrogen, oxygen, phosphorus, arsenic and selenium are the non-metallic elements in the periodic table. Where are nonmetals found on Earth?
Which element is a nonmetal?
These nonmetals include hydrogen, carbon, nitrogen, oxygen, phosphorous, sulfur, and selenium. Hydrogen, nitrogen, and oxygen are colorless gases; carbon, phosphorous, and selenium are solids that sometimes have a metallic appearance; and sulfur is a brittle, yellow solid.

What are the top 3 most reactive metals?
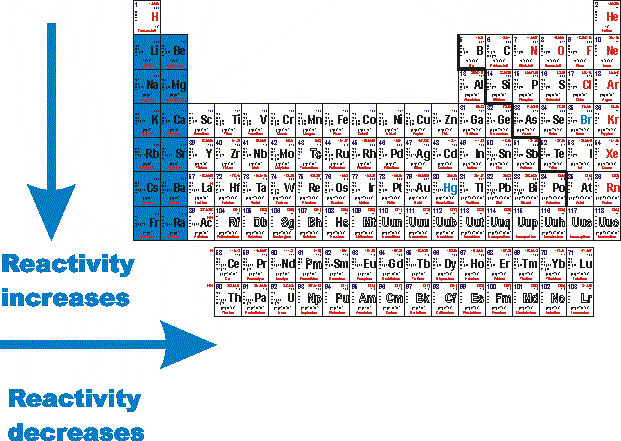
The elements toward the bottom left corner of the periodic table are the metals that are the most active in the sense of being the most reactive. Lithium, sodium, and potassium all react with water, for example.
What is the most non reactive nonmetal?
Helium is the least reactive of the non-metallic elements. Helium is a chemical element with the atomic number and the symbol .
What is a reactive nonmetal?
Some nonmetals are very reactive, whereas others are not reactive at all. It depends on the number of electrons in their outer energy level. Reactive nonmetals tend to gain electrons. This explains why they cannot conduct electricity, which is a flow of electrons.
What are the 4 most reactive metals?
Explanation: Alkali metals are lithium (Li), sodium (Na), potassium (K), rubidiumrubidiumRubidium oxide is the chemical compound with the formula Rb2O. Rubidium oxide is highly reactive towards water, and therefore it would not be expected to occur naturally. The rubidium content in minerals is often calculated and quoted in terms of Rb2O.https://en.wikipedia.org › wiki › Rubidium_oxideRubidium oxide - Wikipedia (Rb), cesium (Cs), and francium (Fr). As you move down the column, the metals become more reactive because the nucleus gains more electrons and protons (more electron levels), weakening their electrostatic force.
Which element is most reactive?
Fluorine: The Most Reactive And Indispensable Chemical Element In Our Daily Lives. Fluorine is a remarkable chemical element - not only because of its unique reactivity.
Which metal is highly reactive?
alkali metal are most reactive metals.
Where are the most reactive non metals on the periodic table?
Answer and Explanation: The most reactive nonmetals are the halogens (of group 17), which are located on the far right-hand side of the periodic table, just before the noble... See full answer below.
Which group contains the most reactive non metals?
1 Answer. The halogens are the most reactive elements; especially fluorine.
Why are halogens the most reactive of the nonmetals?
Because the halogen elements have seven valence electrons, they only require one additional electron to form a full octet. This characteristic makes them more reactive than other non-metal groups.
What is the 2nd most reactive metal?
The alkaline earth metals are the second most reactive family of elements. Beryllium, magnesium, calcium, strontium, barium and radium are all shiny, and silvery-white.
What are the 2 most reactive groups on the periodic table?
The two most reactive groups of elements are the alkali metals and the halogens, because of their valence electrons.
What is the order of reactivity?
The reactivity series follows the order, from most reactive to least reactive:Cesium.Francium.Rubidium.Potassium.Sodium.Lithium.Barium.Radium.More items...•
What group is the most unreactive?
Group 8A — The Noble or Inert Gases. Group 8A (or VIIIA) of the periodic table are the noble gases or inert gases: helium (He), neon (Ne), argon (Ar), krypton (Krkrypton (KrKrypton (from Ancient Greek: κρυπτός, romanized: kryptos 'the hidden one') is a chemical element with the symbol Kr and atomic number 36. It is a colorless, odorless, tasteless noble gas that occurs in trace amounts in the atmosphere and is often used with other rare gases in fluorescent lamps.https://en.wikipedia.org › wiki › KryptonKrypton - Wikipedia), xenon (Xe), and radon (Rn). The name comes from the fact that these elements are virtually unreactive towards other elements or compounds.
What are the least reactive metals?
The least reactive metals would be Platinum, Gold, Palladium, Osmium and Silver and in the decreasing order.
Why is fluorine the most reactive non-metal?
Fluorine is the most reactive non-metal because it is the most electronegative of all of the non-metal elements of the periodic table. Due to its strong electro negativity & small size, Fluorine has a strong tendency to accept electrons from other atoms or ions. As a result it oxidises all other substances.
Which group is least reactive?
Noble gasesNoble gases are nonreactive, nonmetallic elements in group 18 of the periodic table. Noble gases are the least reactive of all elements. That's because they have eight valence electrons, which fill their outer energy level.
Which element is the most reactive in group 7?
The most reactive element from group seven is fluorine which is at the top of that section of the periodic table. The further down the group you go, the less reactive the element is. Halogens are poisonous to humans on the whole, though each one is poisonous to a different degree. The halogens also tend to be poor conductors of electricity and heat.
Why are halogens so reactive?
Halogens are highly reactive because they are all electronegative. They can gain electrons very quickly which is what makes them react so strongly to other elements. Halogens can form many bonds very fast. Fluorine, for example, only has the choice of losing seven electrons or gaining one, so it almost always gains the one electron since this is much easier. As a result, it often rips electrons away from other elements in order to gain this electron and become stable.
Which elemental group is the only one that has this characteristic?
The halogens also tend to be poor conductors of electricity and heat. These chemicals exist at all phases of matter including solid, liquid and gas; they are the only elemental group that has this characteristic. ADVERTISEMENT.
Which nonmetals are the most reactive?
Actually the halogens are the most reactive nonmetals, but we know that as we move down the group, the electronegativity decreases. In other words, Fluorine is at the top of the halogen group and it has less atomic size plus it needs only one electron to complete the octet.
How many nonmetals are there in the periodic table?
There are 18 nonmetals on the Periodic table. All these nonmetals are located on the upper right corner of the Periodic table (Hydrogen is located on the left top corner) In the above image, the nonmetals are represented in yellow color. [ Note: Astatine (atomic number 85) shows characteristics of nonmetals (halogens) as well as metalloids.
What are nonmetals?
Or in other words nonmetals are those elements which do not possess the properties of metals.
How many electrons are in the outermost orbit of a nonmetal?
Nonmetals have 4 to 8 electrons in their outermost orbit. (Halogens have 7 electrons and noble gases have 8 electrons in outermost orbit)
Why are nonmetals oxidizing agents?
Nonmetals are oxidizing agents because they gain electron/s during a chemical reaction and get reduced.
What does it mean when a solid nonmetal breaks?
Solid nonmetals are brittle in nature. That means they break easily when force is applied on them.
Which element has the highest electronegativity?
Also the atomic size of fluorine is very small. (Check this Periodic table guide, for atomic size trend) And because of its smaller size and need of only one electron, it has the highest electronegativity. Electronegativity is nothing but a tendency to attract the electron pair towards it.