The octet rule states that atoms lose, gain, or share electrons in order to acquire a full set of 8 valence electrons. In covalent an bonding the atom share electrons to achieve octet. How does the octet rule relate to bonding? A stable arrangement is attended when the atom is surrounded by eight electrons.
What is octet rule Short answer?
What is the Octet Rule? The octet rule dictates that atoms are most stable when their valence shells are filled with eight electrons.
What is the octet rule and how does it apply to both ionic and covalent bonding?
The Octet Rule: The atoms that participate in covalent bonding share electrons in a way that enables them to acquire a stable electron configuration, or full valence shell. This means that they want to acquire the electronic configuration of the noble gas of their row.
What is octet rule quizlet?
Octet Rule. States that atoms must attain 8 electrons in outermost energy level to become stable. Subatomic Particles. Particles SMALLER than an atom.
How do covalent bonds adhere to the octet rule?
If atoms have similar electronegativities (the same affinity for electrons), covalent bonds are most likely to occur. Because both atoms have the same affinity for electrons and neither has a tendency to donate them, they share electrons in order to achieve octet configuration and become more stable.
How does the octet rule apply to ionic and covalent bonds quizlet?
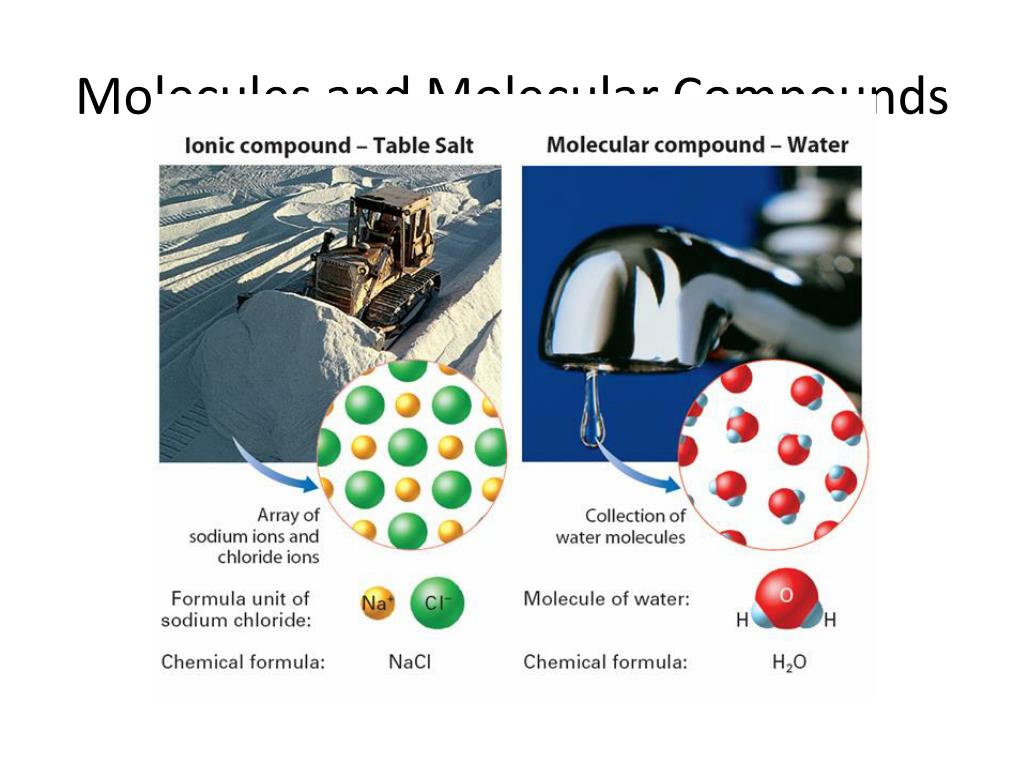
Atoms with covalent bonds satisfy the octet rule by sharing electrons. Nonmetals with ionic bonds satisfy the octet rule by gaining electrons.
What is the octet rule and why is it important?
The octet rule refers to an important principle that the atoms which have bonded share eight outer electrons. This certainly means that the atom's valence shell has a resemblance with a noble gas. The octet rule states that the atoms like to have eight electrons only in their full outer shells.
What is octet rule explain with example?
The tendency of atoms to have eight electrons in the valence shell is known as the 'Octet rule' or the 'Rule of eight'. Example: Sodium with atomic number 11 will readily loose one electron to attain Neon's stable electronic configuration. Concept: Chemical Bond.
What is octet rule in Brainly?
Answer: The octet rule is a chemical rule of thumb that reflects the theory that main-group elements tend to bond in such a way that each atom has eight electrons in its valence shell, giving it the same electronic configuration as a noble gas. Explanation: heart outlined.
Does covalent follows the octet rule?
Since only the valence electrons are shown in these diagrams, the attainment of a noble-gas structure is easily recognized as the attainment of a full complement of eight electron dots (an octet) around each symbol. In other words covalent as well as ionic compounds obey the octet rule.
How are the octet rule and ionic bonding connected?
Ionic bonds require an electron donor, often a metal, and an electron acceptor, a nonmetal. Ionic bonding is observed because metals have few electrons in their outer-most orbitals. By losing those electrons, these metals can achieve noble gas configuration and satisfy the octet rule.
What is the octet rule for ionic compounds?
In the formation of a chemical bond, atoms lose, gain or share valence electrons to complete their outer shell and attain a noble gas configuration. This tendency of atoms to have eight electrons in their outer shell is known as the octet rule.
Does the octet rule apply to ionic as well as to covalent compounds explain using examples as appropriate?
Since only the valence electrons are shown in these diagrams, the attainment of a noble-gas structure is easily recognized as the attainment of a full complement of eight electron dots (an octet) around each symbol. In other words covalent as well as ionic compounds obey the octet rule.
Which is used for both ionic and covalent bonding?
Calcium carbonate is another example of a compound with both ionic and covalent bonds. Here calcium acts as the cation, with the carbonate species as the anion. These species share an ionic bond, while the carbon and oxygen atoms in carbonate are covalently bonded.
How many valence electrons does an atom need to gain to gain an octet?
The octet rule states that atoms lose, gain, or share electrons in order to acquire a full set of 8 valence electrons. In covalent an bonding the atom share electrons to achieve octet.
How many sigma bonds are there in a triple bond?
In a single bond, it has a sigma bond. In a double bond, it has a sigma and pi bond. In a triple bond, it has one sigma bond and two pi bonds.
What is the VSEPR model?
The VSEPR model is based on an arrangement that minimizes the repulsion of shared and unshared pairs of electrons around the central atom.
What chapter is the periodic table?
The periodic table and periodic law chapter 6
Do electron clouds repel each other?
Electron clouds repel each other and they will get as far from each other in 3-D space as they can.
