
Just as wavelength and frequency are related to light, they are also related to energy. The shorter the wavelengths and higher the frequency corresponds with greater energy. So the longer the wavelengths and lower the frequency results in lower energy.
How to calculate energy with wavelength?
Part 3 Part 3 of 3: Catching Mistakes
- Check your answer by multiplying the wavelength by the frequency. ...
- Use scientific notation to avoid calculator rounding errors. Wavelength calculations often involve very large numbers, especially if you're working with light speed.
- Do not change frequency when a wave enters a different medium. ...
- Check your units. ...
How can I calculate the wavelength from energy?
User Guide
- Formula
- Photon Energy. Enter the energy per photon for the electromagnetic wave. ...
- Wavelength. This is the distance of one complete cycle of oscillation, for an electromagnetic wave, produced by a photon propagating through a vacuum.
How to find the wavelength when frequency is given?
The Formula for Wavelength to Frequency
- λ is the wavelength of the wave under consideration expressed in the units of a metre,
- C is the speed of the wave in the given medium, expressed in terms of m/s
- f is the frequency of the wave expressed in terms of Hertz.
What is the equation for wavelength and frequency?
Wavelength and frequency are therefore inversely related. As the wavelength of a wave increases, its frequency decreases. The equation that relates the two is: (5.2.1) c = λ ν The variable c is the speed of light.
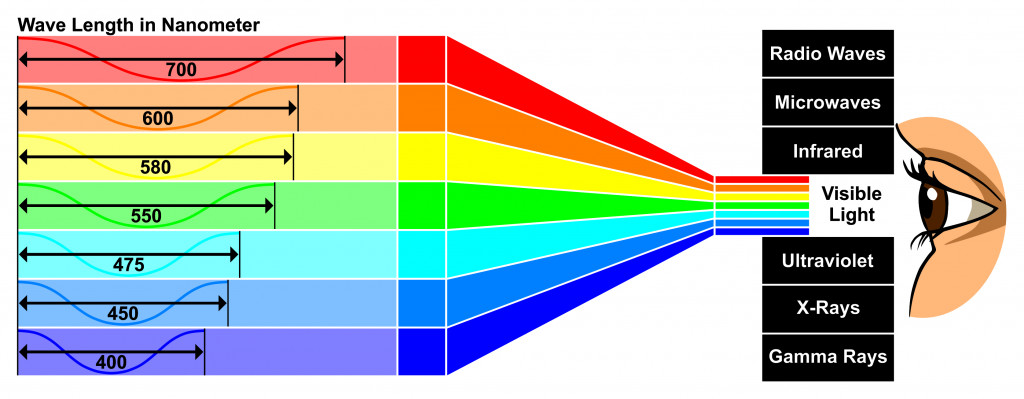
What is the wavelength of a wave?
The wavelength is the distance between two crests or two troughs in a wave. The peak point of the wave is crest whereas the lowest point of the waveform is a trough. The units of wavelength are meters, cms, mms, nms, etc. The wavelength (λ) equation is = λ = v/f. Where. V = Phase speed or velocity.
What is guided wavelength?
The guided wavelength can be defined as the space between two equivalent phase planes with the waveguide. This wavelength is a function used to operate frequency as well as the low-cutoff wavelength. The guide wavelength equation is shown below.
What is the distance between two wave crests called?
The distance among two wave crests is called wavelength and for troughs also it will be the same. The number of ripples that flow through a specified point within 1 sec is known as the frequency, and it is calculated in cycles/second known as HZ (Hertz). This article discusses the relationship between wavelength and frequency.
How does each position on a signal reaches the same rate after a single period?
Each position on the signal reaches to the same rate after a single period, as a signal goes through one oscillation throughout a single stage. This happens when every session result of oscillation travels through a wavelength distance within the single phase to close.
How fast can electromagnetic waves travel?
The traveling of electromagnetic or EM waves can be done with a speed of 299,792 km/sec. This is one of the important characteristics. There are numerous types of waves are available which varies with frequency as well as wavelength.
Can the frequency change when the sound waves travel from one medium to another?
Once the sound wave starts traveling from one medium to another medium then the sound speed can be changed . But, usually, the frequency remains very similar as it is similar to a driven oscillation. If ‘Vw’ alters & the frequency remains the same, after that the wavelength must be changed.
Relationship between Frequency and Energy of Wave
The energy of a wave is characterized by the frequency of the occurrence of particles in a wave. The energy of any body is related to its frequency by the equation
Graph of Energy and Frequency of Wave
The particle will travel at high speed if the amount of kinetic energy acquired by the particle is more. The speed of the particle is directly proportional to its wavelength. If the length of the wave of the particle is more, then the frequency of its occurrence will be lessened. The wavelength is inversely related to the energy of the particle.
Relationship between Frequency and Energy of Radiation
The frequency of the wave radiated from the object is more, when the radiations received by the surface of the object are high. The frequency of the wave is reduced at cooler temperatures as the wavelength of the radiated wave increases.
Relationship between Frequency and Energy of the Photon
The photon is a quantum particle of light that has zero rest mass. The energy of the photon in the electromagnetic radiation is directly proportional to the frequency of the photon and is given by the relation,
Relation between the Kinetic Energy and the Frequency
The kinetic energy of the particle having a mass ‘m’ and travelling with velocity ‘v’ is given by the formula
How does the frequency affect the power of the radiations?
The power of the radiations depends upon the emissivity of the objects and the temperature at which they are exposed.
Answer
Just as wavelength and frequency are related to light, they are also related to energy. The shorter the wavelengths and higher the frequency corresponds with greater energy. So the longer the wavelengths and lower the frequency results in lower energy. The energy equation is E = hν.
Answer
Just as wavelength and frequency are related to light, they are also related to energy. The shorter the wavelengths and higher the frequency corresponds with greater energy. So the longer the wavelengths and lower the frequency results in lower energy

Energy and Wavelength Relationship Formula
- The energy of the particle can be related to its speed during propagation. The speed of the particle gives the idea of the frequency and the length of the wave. If the wavelength is minute then the frequency and hence the energy of the particle will increase. If the oscillations of the pa…
Energy and Wavelength Relationship Graph
- As the wavelength increases, the frequency of the wave falls off thus declining the energy possessed by the wave. If we plot a graph of Energy v/s Wavelength of the emerging particle, then the graph will look like as shown below The above graph clearly indicated that, as the wavelength increases, the energy associated with the particle decreases exponentially.
Kinetic Energy and Wavelength Relationship
- If the speed of the particle is greater, it is evident that the kinetic energy of the particle is high.The kinetic energy is given by the equation Where m is a mass of the object or particle V is a velocity of the mass We can write the above equation as Multiplying ‘m’ on both sides of the equation The momentum of the object is given as the product of object mass and the velocity at which it is m…
Electron Energy and Wavelength Relationship
- The energy of the electron is given by the simple equation as Where ‘h’ is a Planck’s constant and is a frequency of occurrence of the electron The frequency of the electron is given as Where v is a velocity of the electron and is a wavelength of the electron wave Hence, the energy is related to the wavelength of the electron as This is a relation to find the energy associated with the single …
Radiant Energy and Wavelength Relationship
- Every object absorbs light rays during daylight depending upon its shape, size, and its composition. If the temperature of the surface of the object reaches above the absolute zero temperature, the object will emit the radiations in the form of waves. This emitted radiation is proportional to the fourth power of the absolute temperature of the object and is given by the eq…
Energy of Photon and Wavelength Relationship
- The energy possessed by a photon is termed as photon energy, and is inversely proportional to the electromagnetic wave of the photon, by the relation Where ‘h’ is a Planck’s constant C is a speed of light is a wavelength of the photon The frequency of the photon is given by the equation Where f is a frequency Hence, the photon with a large wavelength possesses a small unit of ene…
What Is The Absolute Temperature of The System?
- It is a non-variable and a perfect value of the temperature of a system. The absolute temperature of the system is measured on the scale of degree Celsius, Fahrenheit, or Kelvin which measure zero as absolute zero degrees.
How Does The Wavelength of The Photon Be Dependent on The temperature?
- The temperature of the system specifies the agility of the particles of the system. The more radiations received by the system at higher temperatures, the more emission will be given out from the system. At higher temperatures, shorter wavelength radiations are omitted and at lower temperatures, longer wavelengths are radiated.