Conditions for writing the Lewis dot structures
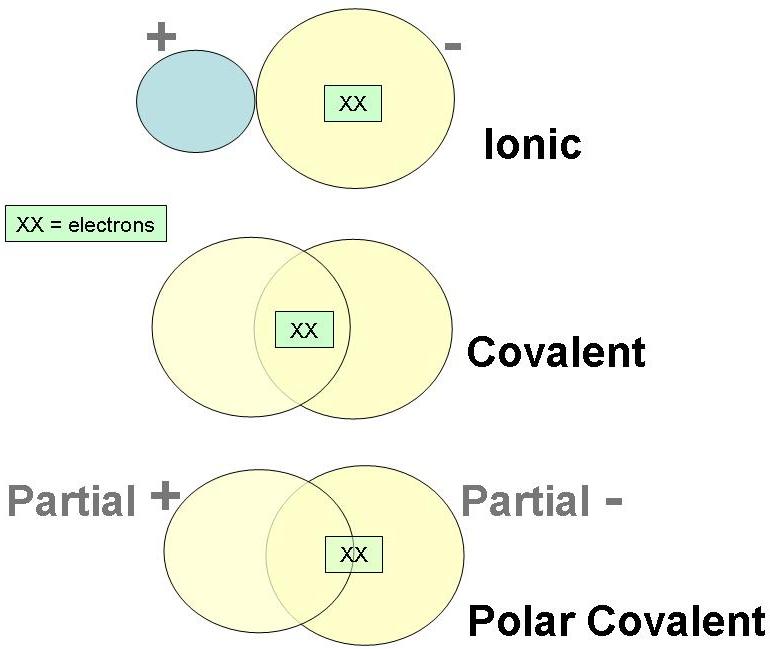
- Sharing of an electron pair between the atoms results in the formation of covalent bonds.
- During bond formation, each bond consists of two electrons which are contributed by each one of the combining atoms.
- By the mutual sharing of electrons, each atom attains octet configuration in its valence shell.
What result results in the formation of covalent bonds?
Sharing of an electron pair between the atoms results in the formation of covalent bonds. During bond formation, each bond consists of two electrons which are contributed by each one of the combining atoms.
What type of bond is formed by sharing of electrons?
Covalent Bond A covalent bond is formed by equal sharing of electrons from both the participating atoms. The pair of electrons participating in this type of bonding is called shared pair or bonding pair. The covalent bonds are also termed as molecular bonds.
What is a shared pair in a covalent bond?
A covalent bond is formed by equal sharing of electrons from both the participating atoms. The pair of electrons participating in this type of bonding is called shared pair or bonding pair.
How many electrons does each atom share during bond formation?
During bond formation, each bond consists of two electrons which are contributed by each one of the combining atoms. By the mutual sharing of electrons, each atom attains octet configuration in its valence shell.

What is the result of electron sharing in covalent bonds quizlet?
What is the result of electron sharing in covalent bonds? In covalent bonds, electron sharing usually occurs so that atoms attain the electron configuration of noble gases.
How are electrons shared in covalent bonds?
A covalent bond consists of the mutual sharing of one or more pairs of electrons between two atoms. These electrons are simultaneously attracted by the two atomic nuclei. A covalent bond forms when the difference between the electronegativities of two atoms is too small for an electron transfer to occur to form ions.
Do covalent bonds involve electron sharing?
Covalent bonds involve the sharing of electron pairs between atoms. Electron pairs shared between atoms of equal or very similar electronegativity constitute a nonpolar covalent bond (e.g., H–H or C–H), while electrons shared between atoms of unequal electronegativity constitute a polar covalent bond (e.g., H–O).
What is formed when sharing electrons?
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.
How many electrons are shared in a covalent bond?
two electronsA single covalent bond is formed when two electrons are shared between the same two atoms, one electron from each atom.
How covalent bonds are formed?
A covalent bond is formed by equal sharing of electrons from both the participating atoms. The pair of electrons participating in this type of bonding is called shared pair or bonding pair. The covalent bonds are also termed as molecular bonds.
What are the characteristics of covalent bond?
Properties of Covalent Compounds:The covalent compounds exist as gases or liquids or soft solids.The melting and boiling points of covalent compounds are generally low.Covalent compound are insoluble in water but dissolve in organic solvents.They are non-conductors of electricity in solid, molten or aqueous state.
What particle is produced after covalent bonding?
Particles formed from the covalent bonding of atoms are called ions.
What is covalent bond explain with example?
These electron pairs are known as shared pairs orbonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding. example could be "Water, H2O" as it is formed by the share of electrons of hydrogen and oxygen (which are both non-metals).
What is formed when two or more atoms bond covalently?
A covalent bond is the force of attraction that holds together two atoms that share a pair of valence electrons. The shared electrons are attracted to the nuclei of both atoms. This forms a molecule consisting of two or more atoms.
How are covalent bonds formed quizlet?
A covalent bond forms when two or more valence electrons are attracted by the positively charged nuclei of two atoms and thus are shared between both atoms.
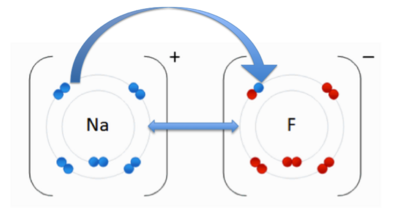
Are electrons shared in ionic bonds?
In ionic bonding, atoms transfer electrons to each other. Ionic bonds require at least one electron donor and one electron acceptor. In contrast, atoms with the same electronegativity share electrons in covalent bonds, because neither atom preferentially attracts or repels the shared electrons.
Are valence electrons shared in covalent bonds?
A covalent bond is the force of attraction that holds together two atoms that share a pair of valence electrons. The shared electrons are attracted to the nuclei of both atoms. This forms a molecule consisting of two or more atoms. Covalent bonds form only between atoms of nonmetals.
How are electrons transferred between atoms?
The atom losing one or more electrons becomes a cation—a positively charged ion. The atom gaining one or more electron becomes an anion—a negatively charged ion. When the transfer of electrons occurs, an electrostatic attraction between the two ions of opposite charge takes place and an ionic bond is formed.
What type of bond is formed by equal sharing of electrons from both the participating atoms?
Covalent Bond. A covalent bond is formed by equal sharing of electrons from both the participating atoms. The pair of electrons participating in this type of bonding is called shared pair or bonding pair. The covalent bonds are also termed as molecular bonds. Sharing of bonding pairs will ensure that the atoms achieve stability in their outer shell ...
How is covalent bonding achieved?
Covalent Bonding can be Achieved in two Ways: Sharing of electrons between atoms of the same kind E .g. Formation of H 2 , Cl 2, O 2, etc. Sharing of electrons between atoms of different kind E.g. Formation of CH 4, H 2 O, NH 3, etc.
What is the Octet Rule?
All atoms except noble gases have less than eight electrons in their val ence shell. In other words, the valence shells of these atoms do not have stable configurations. Therefore, they combine with each other or with other atoms to attain stable electronic configurations.
What are the properties of covalent bonds?
Some of the properties of covalent bonds are: Covalent bonding does not result in the formation of new electrons. The bond only pairs them. They are very powerful chemical bonds that exist between atoms. A covalent bond normally contains the energy of about ~80 kilocalories per mole (kcal/mol).
What type of bond is formed between two metals?
A covalent bond is formed between two similar electronegative non-metals. This type of bond is formed between a metal and non-metal. Bonds formed from covalent bonding have a Definite shape. Ionic Bonds have No definite shape. Low Melting Point and Boiling Point.
What type of bond is between oxygen and carbon?
Since two electron pairs are shared there is a double bond between the two oxygen atoms. Ethylene Molecule: In ethylene, each carbon atom shares two of its valence electron with two hydrogen atoms and remaining two electrons with the other carbon atom. So there is a double bond between the carbon atoms.
Why do covalent bonds have unequal electrons?
This type of covalent bond exists where the unequal sharing of electrons occurs due to the difference in the electronegativity of combining atoms. More electronegative atom will have a stronger pull for electrons. The electronegative difference between the atoms is greater than zero and less than 2.0. As a result, the shared pair of electrons will be closer to that atom.