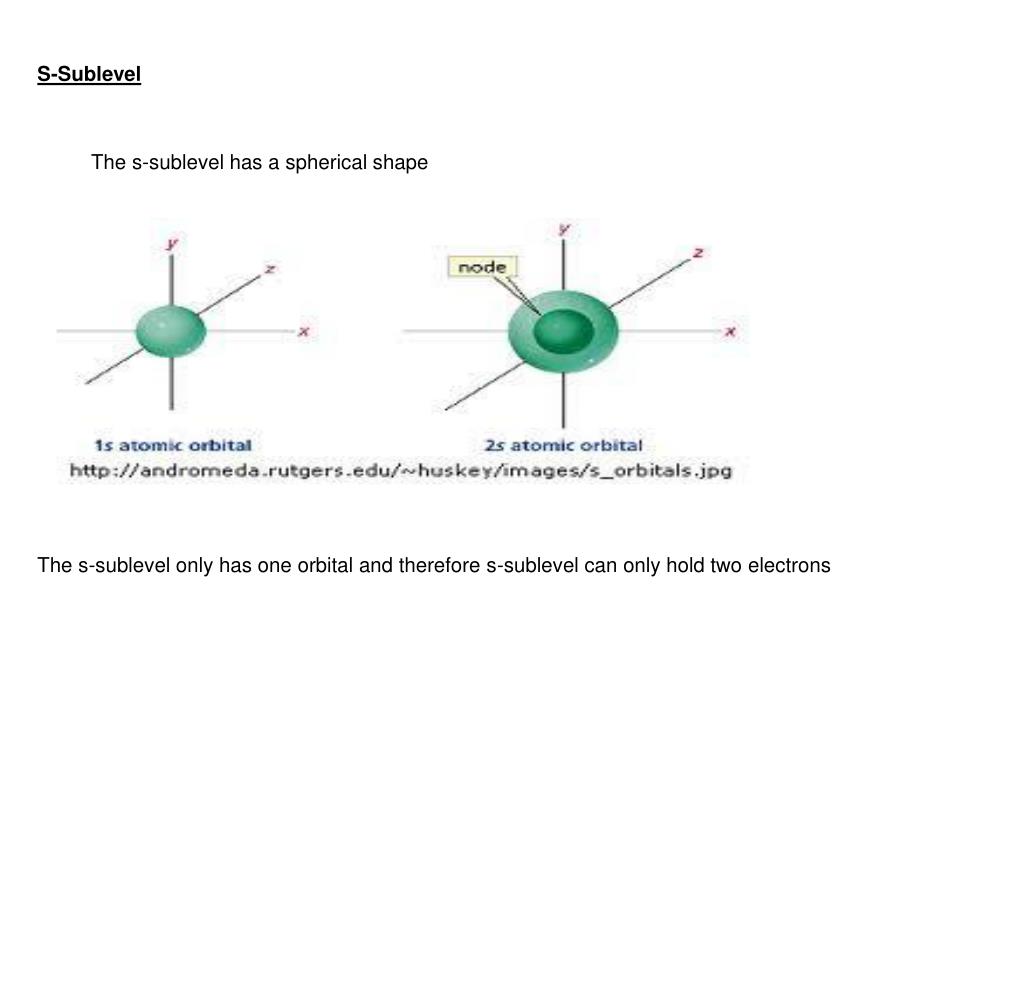
How many orbitals are in each sublevel?
Each sublevel has differing numbers of orbitals. Sublevels are designated with lower-case letters. Sublevel s contains one orbital, p contains three, d has five, f has seven, g has nine, h has 11 and i has 13. The periodic table of elements contains seven rows; therefore, elements can have up to seven energy levels, depending on how many ...
What is the difference between an orbital and a sublevel?
What is the difference between orbitals and sublevels? The sublevels contain orbitals. Orbitals are spaces that have a high probability of containing an electron. In other words, an orbital is an area where the electrons live. There can be two electronsin one orbital maximum. The s sublevel has just one orbital, so can contain 2 electrons max.
How many orbitals in F?
There are seven f orbitals in an f subshell. How many orbitals does 4f have? For any atom, there are seven 4f orbitals. The f-orbitals are unusual in that there are two sets of orbitals in common use. What do f orbitals look like? The f orbital has 15 protons to complete a fifth level of a tetrahedral structure.
How many orbitals are in 5p?
There are 3 orbitals in an atom that has a designation of 5p. How many orbitals are in 5p? For any atom, there are three 5p orbitals. These orbitals have the same shape but are aligned differently in space. How many orbitals does the 5 sublevel have? The d sublevel has 5 orbitals, so can contain 10 electrons max. Does the 5p orbital exist?
What shape is an f orbital?
There is no discrete shape in the f-subshell. This is because of the complexity of its nature. Therefore the answer would be option D, No definite shape.
What is the shape of each sublevel?
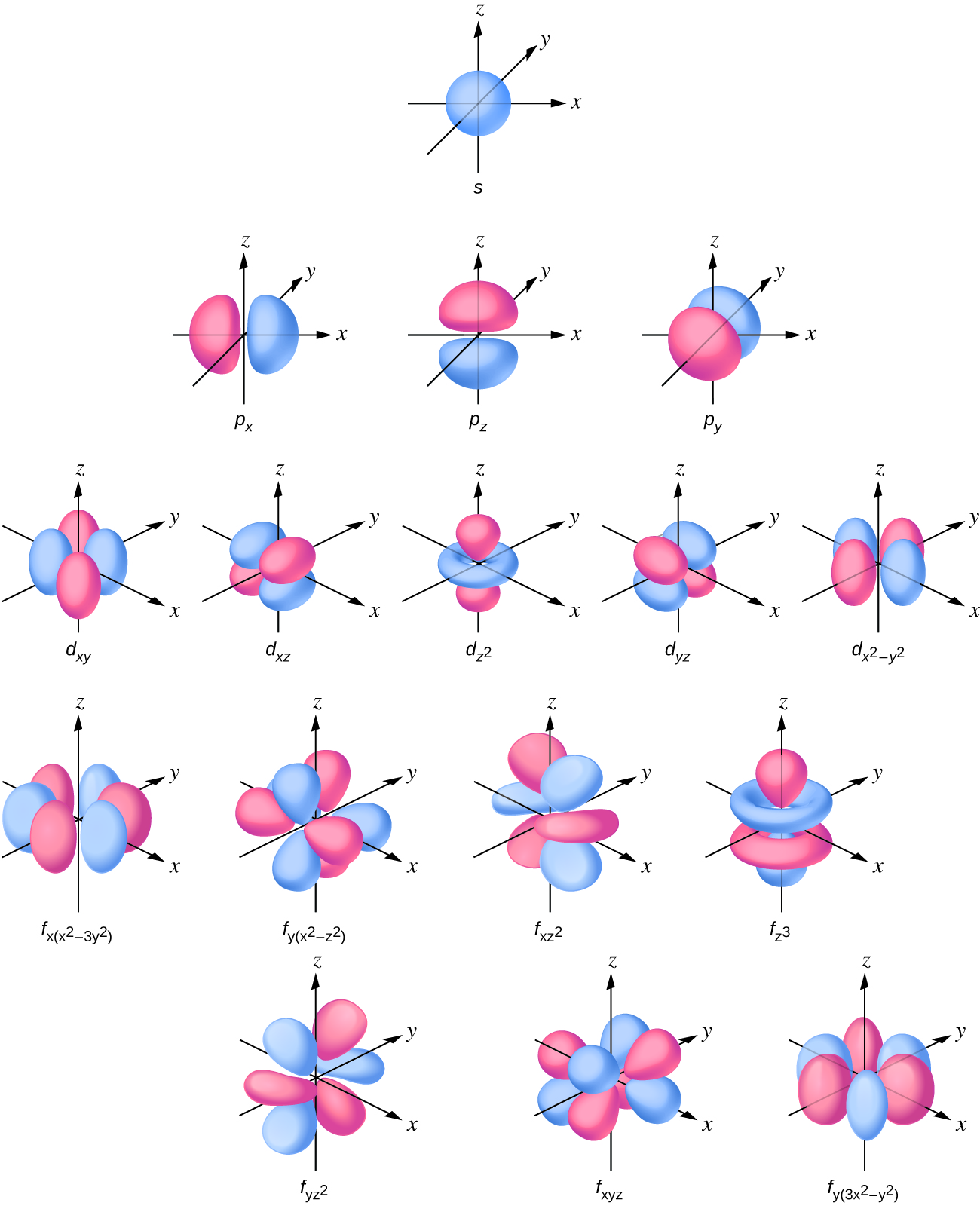
Each of the four sublevels have a specific shape or area where electrons can be found. As shown in Figure 3.6, the s sublevel has a spherical shape, the p sublevel has a dumbbell shape, and the d sublevel has a four leaf clover shape.
Can we draw the shape of f orbitals?
If you absolutely have to draw them, you can draw the shapes as below. One orbital looks like a p orbital with two doughnuts around its middle. Two orbitals have eight lobes pointing towards the corners of a cube. Four orbitals have six lobes oriented in various planes (easiest to draw).
What is the shape of d orbital and f orbital?
An s-orbital is spherical with the nucleus at its centre, a p-orbitals is dumbbell-shaped and four of the five d orbitals are cloverleaf shaped. The fifth d orbital is shaped like an elongated dumbbell with a doughnut around its middle. The orbitals in an atom are organized into different layers or electron shells.
How many f orbitals are there?
1 Answer. Ernest Z. There are seven f orbitals in an f subshell.
How many orbitals are in the f sublevel?
OrbitalsSublevelOrbitalss1p3d5f7
What are the f orbitals?
An f orbital is an orbital for which the secondary quantum number l = 3. There are seven f orbitals, with ml = -3, -2, -1, 0, 1, 2, and 3. The f orbitals aren't occupied in the ground state until element 58 (cerium). The electron configuration of cerium is [Xe] 6s24f5d .
What is the name of f orbitals?
Three of the orbitals are common to both sets. These are are the 7fxyz, 7fz 3, and 7fz(x 2-y 2) orbitals. The higher f-orbitals (8f, 9f, ...) are more complex since they have more spherical nodes while the lower orbitals (4f, 5f, and 6f) have fewer.
How many lobes are in the f orbital?
seven lobesVisualizing Electron Orbitals As shown in Table 1, the s subshell has one lobe, the p subshell has three lobes, the d subshell has five lobes, and the f subshell has seven lobes.
What is the d orbital shaped like?
The d orbital is a clover shape because the electron is pushed out four times during the rotation when an opposite spin proton aligns gluons with three spin-aligned protons.
What is the shape of G orbital?
The higher g-orbitals (6g and 7g) are more complex since they have spherical nodes. The shapes of the nine 5g orbitals.
What is the shape of s orbit?
The shape of the s orbital is a sphere; s orbitals are spherically symmetric.
What is the shape of an s sublevel orbital?
spherical shapeThe s orbital is a spherical shape. The p orbital is a dumbbell shape. There are three p orbitals that differ in orientation along a three-dimensional axis. There are five d orbitals, four of which have a clover shape with different orientations, and one that is unique.
What are the sublevels on the periodic table?
As illustrated, the Periodic Table is divided into the s sublevel block, the p sublevel block, the d sublevel block, and the f sublevel block.
What is the meaning of s sublevel?
The sublevels contain orbitals. Orbitals are spaces that have a high probability of containing an electron. In other words, an orbital is an area where the electrons live. There can be two electrons in one orbital maximum. The s sublevel has just one orbital, so can contain 2 electrons max.
What element is 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p5?
Electron Configuration Match 1--Complete AddressABcalcium1s2 2s2 2p6 3s2 3p6 4s2chromium1s2 2s2 2p6 3s2 3p6 4s1 3d5 !copper1s2 2s2 2p6 3s2 3p6 4s1 3d 10 !bromine1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p516 more rows