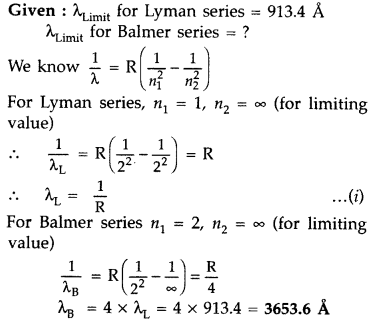
What is the shortest wavelength in the Paschen series of hydrogen?
The shortest wavelength transition in the paschen series in hydrogen occurs at 821 nm. What is the shortest wavelength in the Paschen series of li2+ ion? The shortest wavelength of the light emitted is when the electron jumps from Infinity to 3. For Li²⁺, λ = 9/R × 3² m. = 81/R m. This is the Required Answer.
What is the Paschen series in spectroscopy?
HINT: Paschen series corresponds to the transitions between the higher and lower energy states with principal quantum number 3 or higher. The Paschen series does not lie in the visible spectra. To calculate the shortest wavelength in this series, we can use Rydberg's formula.
What is the value of Rydberg's constant for Paschen series?
The spectral lines obtained due to the emissions are grouped into a series according to their lower energy state (n’). The Paschen series lies in the infrared region. The value of Rydberg’s constant is 1.09677 × 10 7 m − 1.
What is the wavelength of the shortest wave emitted from an electron?
Its wavelength is 1875 nm. Similarly, the shortest wave has been emitted when the electron makes the transition from infinity energy level to level 3 and its wavelength will be 820nm.
What are the spectral lines of hydrogen atoms?
What is the spectral emission?
Where is the Paschen series?
Is the Rydberg constant fixed for every series?
See 1 more
About this website
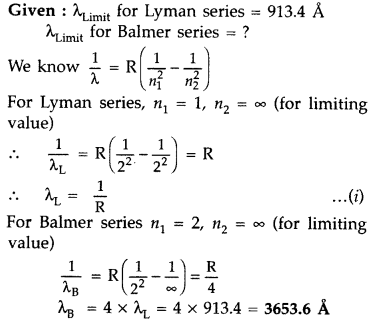
What is the shortest and longest wavelength of Paschen series?
SolutionCalculation:∴ λL = 18750 ÅThe longest wavelength in the Paschen series is 18750 Å.
How the shortest wavelength in the Paschen series is produced?
Solution : The wavelength for Paschen series,
`(1)/(lambda)=R[(1)/(3^2)-(1)/(n^2)]`
For shortest wavelength `n=prop`
`therefore (1)/(lambda)=R[(1)/(9)-(1)/(prop^2)]=(R)/(9)rArrlambda=(9)/(R)=(9)/(1.097xx10^7)`
`=8.20xx10^-7m=820nm`.
What is the wavelength of the line in the Paschen series?
The wavelength of a certain line in the Paschen series of H-spectrum is 1093.6 nm.
What is the shortest and longest wavelength of Lyman series?
The shortest and the longest wavelength in Lyman series of hydrogen spectrum are: Rydberg constant ,RH=109678 cm−1.
What is the shortest wavelength?
Gamma rays have the shortest wavelength and the highest energy among the other waves in the electromagnetic spectrum. The wavelength of the gamma rays is less than 0.01 nanometers.
How is Paschen series calculated?
The wavelengths of the Paschen series for hydrogen are given by 1/λ=RH(1/32−1/n2) 1 / λ = R H ( 1 / 3 2 − 1 / n 2 ) , n = 4, 5, 6, . . . (a) Calculate the wavelengths of the first three lines in this series.
What is Alpha line of Paschen series?
The transitions are named sequentially by Greek letter: n = 4 to n = 3 is called Paschen-alpha, 5 to 3 is Paschen-beta, 6 to 3 is Paschen-gamma, etc.
What is the last line of Paschen series?
This is Expert Verified Answer We have to Find: What is the wavelength of the last line of paschen series? Angstrom. Angstrom.
What is the region of Paschen series?
All the wavelength of the Paschen series falls in the Infrared region of the electromagnetic spectrum.
How do you find the shortest wavelength?
For Lyman series, n1=1.For shortest wavelength in Lyman series (i.e., series limit), the energy difference in two states showing transition should be maximum, i.e., n2=∞.For longest wavelength in Lyman series (i.e., first line), the energy difference in two states showing transition should be minimum, i.e., n2=2.
What is the shortest wavelength present in the Lyman series?
912 ÅThe shortest wavelength of spectral line in Lyman series is 912 Å.
Which line of Lyman series has shortest wavelength?
The shortest wavelength for Lyman series is 912 oA .
What is the shortest and longest wavelength present in the Paschen series II Balmer series of spectral lines?
λ=819nm.
How do you find the shortest wavelength in Balmer series?
Since, we know that for Balmer series,→v = 1λ=R(122−1n2i) 1 λ = R ( 1 2 2 − 1 n i 2 ) , n2 = 3, 4, 5, ......For shortest wavelength of Balmer series ni → ∞1λmin=R(122−1∞) 1 λ m i n = R ( 1 2 2 − 1 ∞ ) = R4 ⇒ λmin=4R=41.097×107m.
What is the shortest wavelength color?
violet lightBlue or violet light has the shortest wavelength. White light is a combination of all colors in the color spectrum. It has all the colors of the rainbow. Combining primary colors of light like red, blue, and green creates secondary colors: yellow, cyan, and magenta.
What do you mean by Paschen series ?`?
Paschen series are the series of lines in the spectrum of the hydrogen atom which corresponds to transitions between the state with principal quantum number n = 3 and successive higher states.
What is the shortest wavelength present in the Paschen series of ...
Rydberg’s formula is given as: ` "hc"/lambda = 21.76 xx 10^(-19) [1/"n"_1^2 - 1/"n"_2^2]` Where, h = Planck’s constant = 6.6 × 10 −34 Js. c = Speed of light = 3 × 10 8 m/s (n 1 and n 2 are integers). The shortest wavelength present in the Paschen series of the spectral lines is given for values n 1 = 3 and n 2 = ∞. `"hc"/lambda = 21.76 xx 10^(-19) [1/(3)^2 - 1/(∞)^2]`
What is the shortest wavelength present in the Paschen series of ...
The Rydberg's formula for the hydrogen atom is. Where R is Rydberg constant for the Hydrogen atom and equals to 1.1 10 7 m-1. For shortest wavelength in Paschen Series n 1 =2 and n 2 =. The shortest wavelength in Paschen Series is therefore 818 nm.
What is the shortest wavelength line in the Paschen series of Li^2 + ion
Click here👆to get an answer to your question ️ What is the shortest wavelength line in the Paschen series of Li^2 + ion ?
CBSE Free NCERT Solution of 12th physics Atoms what is the ... - SaralStudy
Popular Questions of Class 12 Physics. Q:-What is the force between two small charged spheres having charges of 2 x 10-7 C and 3 x 10-7 C placed 30 cm apart in air?. Q:-An infinite line charge produces a field of 9 × 10 4 N/C at a distance of 2 cm. Calculate the linear charge density.. Q:-
What is the capacitance of a parallel plate capacitor?from saralstudy.com
A parallel plate capacitor with air between the plates has a capacitance of 8 pF (1pF = 10 -12 F). What will be the capacitance if the distance between the plates is reduced by half, and the space between them is filled with a substance of dielectric constant 6?
How is a 600 pf capacitor charged?from saralstudy.com
A 600 pF capacitor is charged by a 200 V supply. It is then disconnected from the supply and is connected to another uncharged 600 pF capacitor. How much electrostatic energy is lost in the process?
How much charge does a regular hexagon have?from saralstudy.com
A regular hexagon of side 10 cm has a charge 5 µC at each of its vertices. Calculate the potential at the centre of the hexagon.
What is the charge of polythene?from saralstudy.com
A polythene piece rubbed with wool is found to have a negative charge of 3 × 10 −7 C.
What is the resistance of a galvanometer coil?from saralstudy.com
A galvanometer coil has a resistance of 15 Ω and the metre shows full scale deflection for a current of 4 mA. How will you convert the metre into an ammeter of range 0 to 6 A?
What is the speed of an electron shot into the magnetic field?from saralstudy.com
In a chamber, a uniform magnetic field of 6.5 G (1 G = 10 -4 T) is maintained. An electron is shot into the field with a speed of 4.8 x 10 6 m s -1 normal to the field. Explain why the path of the electron is a circle. Determine the radius of the circular orbit. ( e = 1.6 x 10 -19 C, me = 9.1 x 10 -31 kg)
What is the field of an infinite line charge?from saralstudy.com
An infinite line charge produces a field of 9 × 10 4 N/C at a distance of 2 cm. Calculate the linear charge density.
What are the spectral lines of hydrogen atoms?
ADDITIONAL INFORMATION:For a hydrogen atom, the spectral lines are given special names. For n’=1 the series is called the Lyman series, which lies in the ultraviolet region. n’=2 is given the name Balmer series which has a few lines in the visible region, n’=3 is the Paschen series lying in the infrared region and there are others for n’=4, 5 and 6 named as Brackett series, Pfund series and Humphrey series respectively.
What is the spectral emission?
We know that spectral emission occurs when an electron transitions from a higher energy state to a lower energy state. The lower energy state is designated as n’ and the higher energy state is designated as n. The spectral lines obtained due to the emissions are grouped into a series according to their lower energy state (n’).
Where is the Paschen series?
The Paschen series lies in the infrared region.
Is the Rydberg constant fixed for every series?
NOTE:It is important to remember the n’ value for the series as it is fixed for every series, for hydrogen atoms. We should not forget to mention the units I.e. metres. The value of Rydberg constant is different for heavier metals so it is important to consider it.
What is the spectrum of electrons in an atom?
When electron in the atom or a molecule is making a transition from higher to lower energy level, it emits the spectrum of frequencies of electromagnetic radiation. This Spectrum is called emission spectrum. Such emission spectrum of hydrogen atom has been divided into different number of spectral series depends on wavelength of emitted electromagnetic waves. These wavelengths can be determined by Rydberg Formula.
What is the Paschen series?
Paschen series is a sequence of absorption or emission lines along the near infrared part of the hydrogen atom. This is caused by the electron jump between the 3rd energy level and the higher levels.
How is the spectrum of hydrogen classified?
The spectrum of Hydrogen atom can be classified as different series depends on the transition of electrons in the atom from higher to lower energy levels.
Which atom emits the longest wave?
So, the electromagnetic wave with longest wavelength in Paschen series is emitted by the Hydrogen atom when electron jumps from level 4 to level 3. Its wavelength is 1875 nm. Similarly, the shortest wave has been emitted when the electron makes the transition from infinity energy level to level 3 and its wavelength will be 820nm.
Which series has the shortest wavelength?
These wavelengths fall within the infrared region of the electromagnetic spectrum. Brackett series has the shortest wavelength and it overlaps with the Paschen series. Similarly, the other sequences also overlap.
What are the spectral lines of hydrogen atoms?
ADDITIONAL INFORMATION:For a hydrogen atom, the spectral lines are given special names. For n’=1 the series is called the Lyman series, which lies in the ultraviolet region. n’=2 is given the name Balmer series which has a few lines in the visible region, n’=3 is the Paschen series lying in the infrared region and there are others for n’=4, 5 and 6 named as Brackett series, Pfund series and Humphrey series respectively.
What is the spectral emission?
We know that spectral emission occurs when an electron transitions from a higher energy state to a lower energy state. The lower energy state is designated as n’ and the higher energy state is designated as n. The spectral lines obtained due to the emissions are grouped into a series according to their lower energy state (n’).
Where is the Paschen series?
The Paschen series lies in the infrared region.
Is the Rydberg constant fixed for every series?
NOTE:It is important to remember the n’ value for the series as it is fixed for every series, for hydrogen atoms. We should not forget to mention the units I.e. metres. The value of Rydberg constant is different for heavier metals so it is important to consider it.
