What is uuf and CRRT?
UF is used to remove fluid and UF rate can be controlled UF removes fluid with composition close to plasma water Solutes removed to varying degrees depending upon membrane characteristic Replacement Replacement fluid may be used to replace varying amounts of the fluid removed Composition of the replacement fluid can be varied CRRT Fluid Balance
What is the role of CRRT in fluid management?
Adjust patient fluid balance using CRRT as a tool for fluid management Goal is to adjust fluid removal/administration to achieve patient needs Substitution Fluid Ultrafiltrate Common Strategy Approaches to Fluid Balance with CRRT Fluid Regulation Substitution Fluid Ultrafiltrate Common Strategy Alternate Strategy UF is Varied UF is fixed
What is the role of SCUF in CRRT?
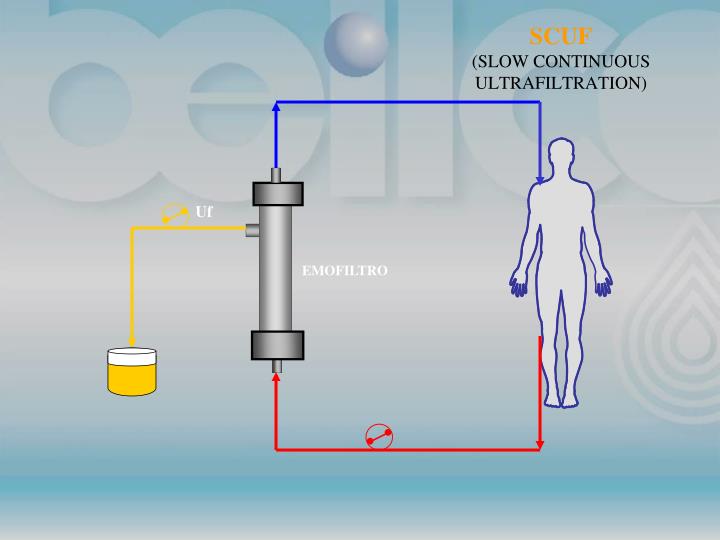
Gabriella Salvatori, in Critical Care Nephrology (Second Edition), 2009 SCUF is the least complex treatment from among all CRRTs. It is employed usually for 24 hours per day in an intensive care setting, but it also may be applied for some hours per day with an arteriovenous or a venovenous access circuit ( Fig. 247-1 ).
How many hours a day is CRRT?
•Renal replacement therapy for critically ill patients •24 hrs/day, 7 days/wk •Available in most US & Canadian ICUs •Common in Europe, Australia, New Zealand 5 Who gets CRRT?
What is CRRT in ICU?
What happens if a CRRT circuit stops cycling?
When in doubt of the circuit clotting, what to do?
Can a fistula be used with a CRRT?
How is UF calculated in CRRT?
UF NET intensity ml / kg / day = Total UF NET volume ml Hospital admission weight kg X RRT duration days . For instance, if an 80-kg patient is on CVVH with an UF rate of 2000 ml/h and substitution fluid of 1500 ml/h, the total UFNET produced is 500 ml/h (2000 – 1500 = 500 ml) or 500 × 24 = 12,000 ml/day.
What is ultrafiltration rate in CRRT?
CRRT is usually initiated with a blood flow rate of 100mls/mt and gradually increased up to 200mls/mt. In CVVH, the ultrafiltrate volume is usually set around 1 to 3 litres/hr. Ronco et al showed in a randomised controlled trial that ultrafiltrate volumes of 35mls/kg/hr are superior to 20 or 45mls/kg/hr.
What is UF on dialysis?
The UF volume is the amount of water that must be removed in a single treatment to return a patient to his or her target (or base) weight. The ultrafiltration volume is commonly expressed in terms of weight (where 1 litre = 1 kilogram). The UF rate is the speed at which that volume is removed.
What is safe ultrafiltration rate in dialysis?
For each treatment, we computed the maximum UF volume (L) allowable under the constraint of a maximum prescribed UF rate of 13 mL/h/kg using the patient's prescribed TT and post-dialysis weight from the previous treatment.
How do you calculate ultrafiltration rate?
For both measures, the UF rate is calculated as UF rate (milliliters per hour per kilogram) = (predialysis weight − postdialysis weight [milliliters])/delivered TT (hours)/postdialysis weight (kilograms).
What is high TMP?
What is a high temperature? Normal body temperature is different for everyone and changes during the day. A high temperature is usually considered to be 38C or above. This is sometimes called a fever.
What is a good ultrafiltration rate?
Currently, the Centers for Medicare & Medicaid Services (CMS) is considering an UF rate threshold of 13 mL/h/kg as a quality measure to assess dialysis facility fluid management, and such a threshold has been incorporated into the CMS 2016 End Stage Renal Disease Core Survey.
What is low UF in dialysis?
UFF, which can be defined as ultrafiltration (UF) of less than 400 mL after a 4-hour dwell duration with a 4.25% dextrose-based peritoneal dialysis fluid (PDF), is a clinical condition that has an increasing incidence with chronic PD duration.
Why is ultrafiltration important?
Ultrafiltration is an effective means of reducing the silt density index of water and removing particulates that can foul reverse osmosis membranes. Ultrafiltration is frequently used to pretreat surface water, seawater and biologically treated municipal water upstream of the reverse osmosis unit.
What is the meaning of ultrafiltration?
Definition of ultrafiltration : filtration through a medium (such as a semipermeable capillary wall) which allows small molecules (as of water) to pass but holds back larger ones (as of protein)
What is a good dialysis clearance?
If a patient's blood flow rate is good, further improvements in clearance can be obtained by using a big dialyzer or, in some cases, by increas ing the flow rate for dialysis solution from the usual 500 mL/min to 600 or 800 mL/min. A good flow rate for adult patients is 350 mL/min and higher.
What happens if too much fluid is removed during dialysis?
If too much fluid is removed and a person goes below their dry weight, a patient may experience dehydration causing: Thirst. Dry mouth. Lightheadedness that goes away when laying down.
What is the meaning of ultrafiltration?
Ultrafiltration is the removal of fluid from a patient and is one of the functions of the kidneys that dialysis treatment replaces. Ultrafiltration occurs when fluid passes across a semipermeable membrane (a membrane that allows some substances to pass through but not others) due to a driving pressure.
What is the difference between convection and ultrafiltration?
Different membranes are utilized for different techniques. Convection is the process whereby solutes pass through membrane pores, dragged by fluid movement (ultrafiltration) caused by a hydrostatic and/or osmotic transmembrane pressure gradient.
How do you calculate filtration fraction on CRRT?
FormulasFiltration Fraction (FF) % = GFR / RPF * 100.Filtration Fraction = Ultrafiltrate flow rate / [Blood flow rate x (1 – Hct) + Pre-dilution replacement flow rate]
Is ultrafiltration the same as dialysis?
The key difference between dialysis and ultrafiltration is that the dialysis is an artificial process of blood filtration that aids patients suffering from kidney failures while the ultrafiltration is one of the three steps of natural blood filtration that occurs in our kidneys.
CRRT Procedures and Guidelines - ResearchGate
CRRT Procedures and Guidelines 2013 Guidelines for the utilization of CRRT (Continuous Renal Replacement Therapies) at Monroe Carell Jr. Children’s Hospital at
Nurses' role in continuous renal replacement therapy
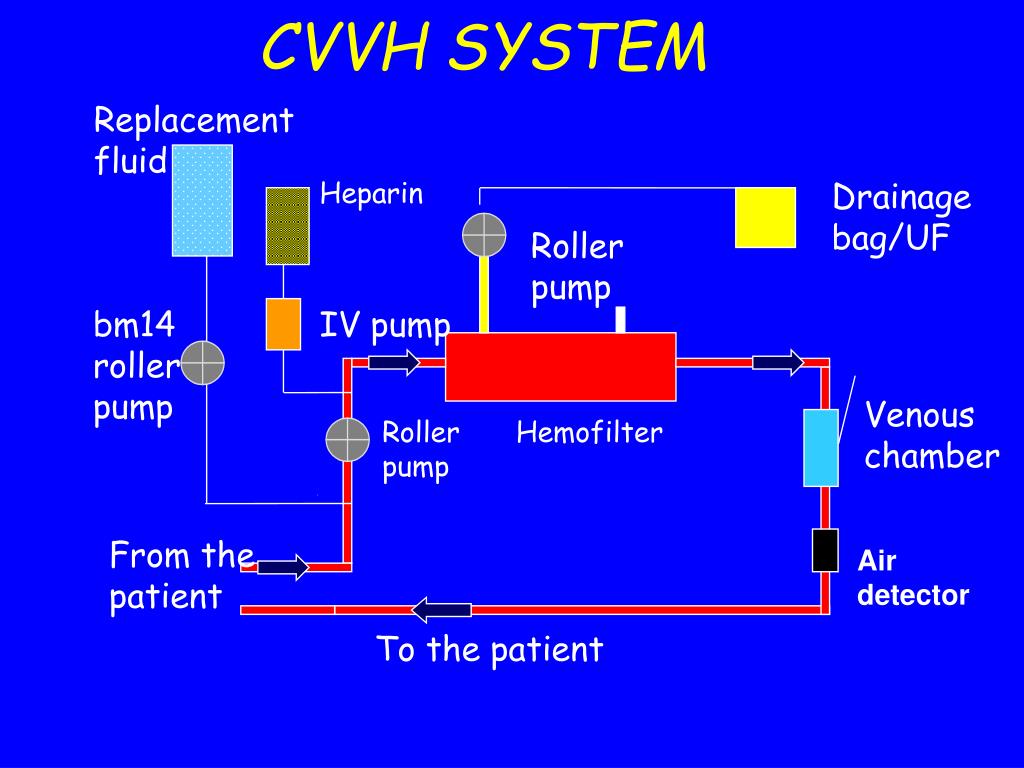
How CRRT works . Continuous renal replacement therapy (CRRT) is a 24-hour blood filtration process. Via hemodialysis access, it slowly purifies the patient’s blood using a cartridge and special filter that removes fluids, toxins, and other substances.
Fluid Management with CRRT
Fluid needs 500 ml/hr. Dilution fluids 700 ml/hr (500 pre-filter and 200 post filter)
Continuous Renal Replacement Therapy: Who, When, Why, and How
Continuous renal replacement therapy (CRRT) is commonly used to provide renal support for critically ill patients with acute kidney injury, particularly patients who are hemodynamically unstable. A variety of techniques that differ in their mode of solute clearance may be used, including continuous …
CRRT Review and Refresh - UCLA Health
CRRT Review and Refresh Pam Waters, RN Acute Field Mentor-West US Region Baxter-Gambro Renal Los Angeles, CA June 11, 2014 Prismaflex 5.1 SW only
What is CRRT in ICU?from criticalcarenow.com
Continuous Renal Replacement Therapy (CRRT) is a treatment option for patients in need of dialysis or fluid removal. It is typically only utilized in the ICU setting and patients require this particular therapy because of their hemodynamic instability. CRRT is a much slower type of dialysis than regular HD, as it pulls fluid or cleans the blood continuously, 24 hours a day, rather than over a 2-4 hr treatment. Some facilities only use this treatment option in ICU patients with renal failure, even if they are hemodynamically stable. This type of therapy relies on the bedside nurse, who has special training in this technology and the equipment. It requires you to be aware of how the patient responds to the treatment both metabolically and hemodynamically at all times.
What is NUF rate in CRRT?from ccforum.biomedcentral.com
A high NUF rate in CRRT may be harmful [ 12 ], although optimal values are not yet established. Because fluid overload is common and undesirable, we regularly reassess fluid status and adjust NUF rate accordingly. We avoid very high NUF rates (> 2 mL/kg/h), unless aggressive fluid removal is indicated by life-threatening fluid overload.
Why is maintaining a CRRT circuit important?from crrtonline.com
Maintaining the CRRT circuit is crucial for delivering CRRT effectively
What happens if a CRRT circuit stops cycling?from criticalcarenow.com
The bedside nurse should always have saline flushes and heparin (or the anticoagulant ordered) within reach if the machine decides to clot off and stop cycling the blood. If the machine stops cycling and appears to be trying to clot, the patient’s blood should be returned and therapy discontinued with the current circuit. Each circuit can hold approximately 150-250 mL blood, which can add up, especially if circuits are replaced 2-3 times a shift.
Can CRRT be discontinued?from ccforum.biomedcentral.com
The decision to discontinue CRRT is based on clinical judgement. However, higher urine output, higher creatinine clearance, and lower serum creatinine can predict successful CRRT cessation [ 5 ]. A trial of CRRT cessation is appropriate when spontaneous urine output is > 500 mL/day and endogenous creatinine clearance is > 15 mL/min. We delay exposure to IRRT until at least 24 h after cessation of vasopressor drugs.
When in doubt of the circuit clotting, what to do?from criticalcarenow.com
When in doubt of the circuit clotting, rinse back. If the patient is getting more hypotensive or unstable, stop treatment and rinse back.
Does citrate affect hemodynamics?from ccforum.biomedcentral.com
Once established, typical blood flow rates (150–250 mL/min) do not affect haemodynamics. For citrate CVVHDF, we prescribe a lower blood flow rate of 120 mL/min because higher rates necessitate a higher dose of citrate, which increases the risk of citrate toxicity.
What is the blood flow rate of an ultrafiltration system?
Although the preceding analysis suggests that a blood flow rate as low as 50 mL/min may be used in isolated ultrafiltration, two caveats must be discussed. First, the filtration fraction calculation mentioned is based on a hematocrit of 35% at the start of therapy. However, as ultrafiltration proceeds and net volume removal occurs, hematocrit increases. Therefore, for a given volume of blood flowing through the filter, an increasing percentage of that volume is composed of red blood cells and a decreasing percentage is composed of plasma water during ongoing ultrafiltration therapy. At a fixed blood flow rate and ultrafiltration rate, this implies an increasing filtration fraction, since plasma water flow rate is the denominator in the filtration fraction equation. Thus, from the relatively narrow perspective of filtration fraction, a seemingly adequate blood flow rate at the onset of ultrafiltration may be inadequate after several hours of therapy. A second important consideration related to blood flow rate involves its effect on blood rheology at the membrane surface. The velocity that blood achieves while passing through an individual hollow fiber membrane is directly proportional to its blood flow rate. 51 In turn, the velocity (or more rigorously, the velocity gradient) of blood at the membrane surface is directly proportional to its shear rate at that membrane-blood interface. When continuous arteriovenous therapies were popular in the early years of CRRT, filters were designed specifically to overcome some of the drawbacks of the characteristically low blood flow rates achieved with these therapies. However, the design characteristics of contemporary filters are based on the higher blood flow rates achieved with continuous venovenous therapies. Thus, whether or not contemporary filters can achieve adequate ultrafiltration rates at blood flow rates delivered from a peripheral venous access (i.e., approximately 50 mL/min) will need to be assessed carefully.
What is extracorporeal ultrafiltration?
Extracorporeal ultrafiltration is being used increasingly as an adjunctive therapy for patients with refractory heart failure. 5,6 In this context, an important consideration is the type of vascular access required. Because slow continuous ultrafiltration is not a blood-cleansing modality such that solute clearance is not a relevant consideration, it is possible to use a smaller-bore catheter in a peripheral vein as vascular access. From one perspective, the minimum blood flow rate is that required to avoid excessive hemoconcentration. To quantify this phenomenon, the filtration fraction (ratio of the ultrafiltration rate to the plasma flow rate delivered to the filter) has been employed traditionally. In general, a maximal filtration fraction of 25% usually guides prescription in acute postdilution hemofiltration, which is the relevant comparison in this instance. At filtration fractions beyond this value, hemoconcentration is associated with an environment that promotes interactions between formed elements and proteins in the blood and the filter membrane, leading to a high risk of filter resulting from widespread pore occlusion (“clogging”). However, the ultrafiltration rates typically employed in SCUF (<10 mL/min) are significantly less than those in hemofiltration, which may be 40 mL/min or higher. Therefore, although the minimum blood flow rate may be 200 mL/min or higher in the setting of postdilution hemofiltration, a blood flow rate of 50 mL/min in SCUF may be adequate to maintain the filtration fraction less than 25%.
What is SCUF used for?
Given its minimal solute clearance (equal to the UFR at generally 4 to 5 ml/min), SCUF has primarily proved useful for treatment of the cardiorenal syndrome (see Chapter 71 ).
What is continuous renal replacement therapy?
Continuous renal replacement therapy is actually an umbrella term for the four different continuous modalities: slow continuous ultrafiltration (SCUF), continuous venovenous hemofiltration (CVVH), continuous venovenous hemodialysis (CVVHD), and continuous venovenous hemodiafiltration (CVVHDF). The type of modality chosen depends on the goal of therapy; treatment may be used for solute removal, fluid removal, or both. Solute clearance through a dialysis filter occurs by diffusion, convection, or combination, called hemodiafiltration (Tables 28.1 to 28.3 and Fig. 28.1 ).
What is CIU in a patient?
CIU is also known as slow continuous ultrafiltration (SCUF). UF is carried out continuously, but slowly over a longer period of time (e.g., up to a matter of days in a single sitting) to remove accumulated fluid. The volume of ultrafiltrate obtained can reach several liters per day. Arteriovenous and venovenous variants of continuous isolated UF (AV-SCUF and VV-SCUF) have been described. Because of the slow rate of excess fluid removal, even patients with relatively unstable cardiovascular systems can often respond favorably to continuous isolated UF. Because SCUF also fits into the previous definition of IU, it is included here as being synonymous with CIU.
What is the blood flow rate for SCUF?
Generally SCUF can be performed with low blood flow rates, between 50 and 200 mL/min in venovenous modality, and ultrafiltration rate is usually between 100 and 300 mL/hr according to fluid balance needs. Because of the low ultrafiltration and blood flow rates required, relatively small surface area filters can be employed with reduced heparin doses to maintain circuit patency. 4 Convective clearance of solutes is not significant and blood purification is ineffective. The main purpose of treatment is to achieve volume control; therefore, no fluids are administered either as dialysate or replacement fluids. 5,6 Synthetic high-flux membranes of polysulfone, polyamide, polymethylmethacrylate, or polyacrylonitrile are generally employed because of their excellent permeability and biocompatibility characteristics. 7,8
What is SCUF in medical terms?
Slow continuous ultrafiltration (SCUF), based only on slow removal of plasma water, is used for patients with refractory fluid overload, with or without renal dysfunction. Its primary aim is to achieve safe and effective correction of fluid overload.
What is CRRT in nursing?
Nursing is vital during CRRT as they have the most exposure to the vascular access site, the CRRT circuit itself, and the patient. Nursing staff and patient care technicians should be aware of complications so that intervention can be initiated early, and the physician team alerted promptly. In addition, pharmacists and nutritionists are vital to ensure proper medication doses and nutrition while on CRRT.
What is CRRT therapy?
Continuous renal replacement therapy (CRRT) is one of the renal replacement methods that include intermittent hemodialysis and peritoneal dialysis. The role of the interprofessional team is reviewed. This activity details the principals, indications, and complications of continuous renal replacement therapy. Additionally, it discusses when to consider initiating CRRT and when to discontinue.
How long does CRRT last?
It is intended to be applied for 24 hours or longer through continuous, slower dialysis. CRRT acts as renal support through blood purification to allow solute and fluid homeostasis. It requires appropriate vascular access, pumps to allow blood circulation, a permeable membrane, and varying solutions to allow fluid balance. There are different techniques of CRRT that are distinguished by their method of solute removal.
How to monitor renal function after CRRT?
There are no standards for dialysis discontinuation. One way of monitoring renal recovery is through measuring urine output; increased urine output is an indicator of improving renal function. In addition to renal function, otherfactors such as fluid overload, ongoing hemodynamic instability, or continued need for nephrotoxic medication or large volumes may need to be considered before stopping CRRT. [6]
What are the risks of CRRT?
The risks of the therapy itself include electrolyte disturbances, clearance of trace elements or antibiotics, hypothermia, and hypotension. Although hypotension should occur less commonly than in IHD, if the net ultrafiltration rate exceeds the rate of intravascular filling, then hypotension may occur. Monitoring of electrolytes and acid-base status should be done every 6-12 hours when starting CRRT. If remaining stable after the first 24 to 48 hours, the interval may be increased to 12-24 hours. The exception, as discussed above, is when using citrate as regional anticoagulation because this requires frequent monitoring of ionized calcium levels. The removal of medications during CRRT is variable, and so it is advised to check the dose of required medications when on CRRT. This practice is especially important when it comes to the administration of antibiotics, as the trough concentrations of these medications will determine their bacteriocidal or bacteriostatic effectiveness. Many patients who meet indications for CRRT will do so as a result of sepsis, meaning that appropriate antibiotic dosage is vital. [4][5]Finally, the risks associated with the extracorporeal circuit include hypersensitivity to the circuit, air embolization, as well as blood loss with filter or circuit changes. [1]
What are the factors that determine if a patient should undergo CRRT?
There are many factors to consider when deciding whether to initiate CRRT. Two main factors are the severity of illness and the necessity of the procedure. The severity of the illness may be judged through the severity of the AKI and the observed trend. Further supporting factors would include the presence ofelectrolyte and acid-base disorders, evidence of fluid overload, and other significant organ dysfunction that requires renal support for promoting recovery. The necessity of the procedure may be judged through the assessment of the likelihood of the reversibility of the acute kidney injury (AKI), the presence of oliguria, and the nature and timing of the renal insult.
Is CRRT delivered safely?
With this team approach and good education for each member, CRRT may be delivered as safely and effectively as possible. [7]
What is CRRT in nursing?from ncbi.nlm.nih.gov
Nursing is vital during CRRT as they have the most exposure to the vascular access site, the CRRT circuit itself, and the patient. Nursing staff and patient care technicians should be aware of complications so that intervention can be initiated early, and the physician team alerted promptly. In addition, pharmacists and nutritionists are vital to ensure proper medication doses and nutrition while on CRRT.
What is NUF rate in CRRT?from ccforum.biomedcentral.com
A high NUF rate in CRRT may be harmful [ 12 ], although optimal values are not yet established. Because fluid overload is common and undesirable, we regularly reassess fluid status and adjust NUF rate accordingly. We avoid very high NUF rates (> 2 mL/kg/h), unless aggressive fluid removal is indicated by life-threatening fluid overload.
What is CRRT therapy?from ncbi.nlm.nih.gov
Continuous renal replacement therapy (CRRT) is one of the renal replacement methods that include intermittent hemodialysis and peritoneal dialysis. The role of the interprofessional team is reviewed. This activity details the principals, indications, and complications of continuous renal replacement therapy. Additionally, it discusses when to consider initiating CRRT and when to discontinue.
How does citrate impede coagulation?from freseniusmedicalcare.ae
The citrate solution impedes coagulation by forming chelate complexes with the ionized calcium of the extracorporeal blood. The citrate solution impedes coagulation by forming chelate complexes with the ionized calcium of the extracorporeal blood. When time is of the essence, every convenience helps.
How long does CRRT last?from ncbi.nlm.nih.gov
It is intended to be applied for 24 hours or longer through continuous, slower dialysis. CRRT acts as renal support through blood purification to allow solute and fluid homeostasis. It requires appropriate vascular access, pumps to allow blood circulation, a permeable membrane, and varying solutions to allow fluid balance. There are different techniques of CRRT that are distinguished by their method of solute removal.
How to monitor renal function after CRRT?from ncbi.nlm.nih.gov
There are no standards for dialysis discontinuation. One way of monitoring renal recovery is through measuring urine output; increased urine output is an indicator of improving renal function. In addition to renal function, otherfactors such as fluid overload, ongoing hemodynamic instability, or continued need for nephrotoxic medication or large volumes may need to be considered before stopping CRRT. [6]
What is the contraindication for CRRT?from ncbi.nlm.nih.gov
The main contraindication for CRRT is the need to have treatment outcomes reached more rapidly than the CRRT treatment can accomplish.
Why do people use CRRT?
Maybe that’s because they have heard of dialysis and can relate to it better than the other therapies patients receive in the ICU setting. CRRT is used until patients start showing signs of their own kidneys recovering — or until they have more stable blood pressure and can tolerate regular dialysis.
How do you explain continuous renal replacement therapy (CRRT) to your patients?
Continuous renal replacement therapy is a special type of dialysis that we do for unstable patients in the ICU whose bodies cannot tolerate regular dialysis. It is a very different type of dialysis from the routine type that patients may be familiar with, and it requires special skills and expertise. Regular hemodialysis is meant to be mostly an outpatient procedure. It is done usually three times a week for three to four hours at a time. The flow rates used to clear waste products and remove fluid from the patient are very fast, potentially putting stress on a patient’s heart and blood pressure. If a patient already has a low or unstable blood pressure or has heart issues, he or she will not tolerate regular dialysis. CRRT is a slower type of dialysis that puts less stress on the heart. Instead of doing it over four hours, CRRT is done 24 hours a day to slowly and continuously clean out waste products and fluid from the patient. It requires special anticoagulation to keep the dialysis circuit from clotting.
What is citrate used for?
Citrate is the yellowish substance you’ve seen at the bottom of the tubes your doctor’s office uses to collect blood, in order to keep that blood from coagulating, or clotting. In 2004, Tolwani developed a new kind of anticoagulant solution based on citrate that has helped make CRRT safer and simpler, allowing it to spread worldwide. That anticoagulant, Prismocitrate 18, is now used in Europe, Canada, India, Australia, New Zealand and most of the rest of the planet. Approval from the U.S. FDA is still pending, although UAB and other U.S. hospitals formulate the compound in their pharmacies. (Meanwhile, Tolwani used royalties from sales of the anticoagulant to start an innovation fund that backs new kidney-related therapies.)
Is CRRT more familiar to doctors?
CRRT is becoming more familiar to doctors and patients alike. “I’m constantly getting email from both critical care and kidney doctors around the world,” Tolwani said. So we asked her to explain the therapy, when it is used and the most common questions she hears from patients and families.
What is CRRT in nursing?from ncbi.nlm.nih.gov
Nursing is vital during CRRT as they have the most exposure to the vascular access site, the CRRT circuit itself, and the patient. Nursing staff and patient care technicians should be aware of complications so that intervention can be initiated early, and the physician team alerted promptly. In addition, pharmacists and nutritionists are vital to ensure proper medication doses and nutrition while on CRRT.
What is CRRT in AKI?from ncbi.nlm.nih.gov
CRRT has become a mainstay in the management of AKI in critically ill patients. In patients who do not have objective indications for the emergent initiation of renal support, the optimal timing of RRT remains controversial. Although the use of continuous therapies may facilitate management in hemodynamically unstable patients, existing data do not show that use of CRRT results in improved survival or recovery of kidney function compared with alternatives such as conventional IHD and PIRRT. Large, well-designed clinical trials have established that for the majority of patients, augmenting solute clearance using effluent flow rates > 20 to 25 mL/kg per hour is not associated with improved outcomes; however, optimal strategies for volume management still must be defined. Similarly, other aspects of the management of CRRT are subject to substantial variations in practice, including strategies for anticoagulation. Finally, the role of CRRT needs to be considered in the setting of overall goals of care and the use of other life-sustaining treatments.
What is the criterion for discontinuing CRRT?from ncbi.nlm.nih.gov
There are no specific criteria for discontinuation of CRRT because of recovery of kidney function or transition to other modalities of RRT. 59, 94, 95 An initial manifestation of recovery of kidney function is increased urine output, although specific criteria are sparse. In the observational Beginning and Ending Supportive Therapy for the Kidney (BEST Kidney) study, a urine output > 400 mL/d without concomitant diuretic therapy was a predictor of successful CRRT discontinuation. 96 In this observational cohort, patients who were successfully discontinued from CRRT without requiring re-initiation were more likely to survive to hospital discharge compared with those requiring re-initiation of CRRT. In another study, urine output > 500 mL/d was proposed as a criterion for discontinuation of RRT in a study of initiation and discontinuation of therapy in patients with AKI. 97 The usefulness of this criterion is uncertain, however, as the treating clinicians continued RRT despite this recommendation approximately two thirds of the time, citing continued volume overload as the most common reason for continuation of RRT. In the ATN study, a 6-h timed urine collection was obtained when the urine output was > 750 mL/d. 62 RRT was continued if the measured creatinine clearance was < 12 mL/min, was discontinued if > 20 mL/min, and was left to clinician judgment if the measured creatinine clearance was between 12 and 20 mL/min. Although these strategies can inform clinical decision-making, precise criteria for discontinuation of RRT are lacking.
What is the most common complication during CRRT?from ncbi.nlm.nih.gov
Clotting of the extracorporeal circuit is the most common complication during CRRT. Practice patterns regarding the use of anticoagulation vary widely, with estimates of 30% to 60% of patients undergoing CRRT without anticoagulation. 62, 70 Although the use of anticoagulation is often avoided in patients who are coagulopathic, thrombocytopenic, or are having active hemorrhage, anticoagulation-free treatment may also be successful in the absence of coagulopathy and thrombocytopenia. Strategies to minimize the risk of clotting of the extracorporeal circuit include the following: use of higher blood flow rates; minimization of filtration fraction (the ratio of ultrafiltration to plasma flow) by using CVVHD rather than CVVH, or by infusing replacement fluids prefilter during CVVH and CVVHDF; ensuring optimal catheter function and responding promptly to machine alarms to minimize interruptions in blood flow; and increasing the frequency of scheduled replacement of the extracorporeal circuit. 62, 64 In the absence of anticoagulation, increased vigilance is required to ensure there is no compromise of delivered dose. 71
What is CRRT therapy?from ncbi.nlm.nih.gov
Continuous renal replacement therapy (CRRT) is commonly used to provide renal support for critically ill patients with acute kidney injury, particularly patients who are hemodynamically unstable. A variety of techniques that differ in their mode of solute clearance may be used, including continuous venovenous hemofiltration with predominantly convective solute clearance, continuous venovenous hemodialysis with predominantly diffusive solute clearance, and continuous venovenous hemodiafiltration, which combines both dialysis and hemofiltration. The present article compares CRRT with other modalities of renal support and reviews indications for initiation of renal replacement therapy, as well as dosing and technical aspects in the management of CRRT.
How does ultrafiltration work in CVVH?from ncbi.nlm.nih.gov
In CVVH, a high rate of ultrafiltration across the semi-permeable hemofilter membrane is created by a hydrostatic gradient, and solute transport occurs by convection ( Fig 2 A). Solutes are entrained in the bulk flow of water across the membrane, a process often referred to as “solvent drag.” 1, 27 High ultrafiltration rates are needed to achieve sufficient solute clearance, and the ultrafiltrate volume beyond what is required to achieve desired net fluid removal is replaced with balanced IV crystalloid solutions. These replacement solutions may be infused into the extracorporeal circuit either prior to or following the hemofilter. Because the high ultrafiltration rate hemoconcentrates the blood as it passes through the hemofilter fibers, the risk of sludging and fiber occlusion is increased. Prefilter infusion of replacement fluid dilutes the blood entering the hemofilter, mitigating this hemoconcentration. However, prefilter administration of replacement fluid dilutes the solute content of the blood, reducing effective solute clearance at a fixed ultrafiltration rate. Postfilter infusion has no such effects.
How long does CRRT last?from ncbi.nlm.nih.gov
It is intended to be applied for 24 hours or longer through continuous, slower dialysis. CRRT acts as renal support through blood purification to allow solute and fluid homeostasis. It requires appropriate vascular access, pumps to allow blood circulation, a permeable membrane, and varying solutions to allow fluid balance. There are different techniques of CRRT that are distinguished by their method of solute removal.
Is real time GFR being evaluated in clinical trials?
Real time GFR measurements are being evaluated in clinical trials
Did 40% of the sulfate concentrations achieve the higher target concentrations?
40% did not achieve the higher target concentrations
What is ultrafiltration in dialysis?
What is Ultrafiltration? Ultrafiltration is the removal of fluid from a patient and is one of the functions of the kidneys that dialysis treatment replaces.
What can you do to help with ultrafiltration?
Do not skip or shorten hemodialysis treatments – adequate dialysis is important for overall health
How does ultrafiltration work in hemodialysis?
Ultrafiltration in Hemodialysis. In hemodialysis, fluid is removed by ultrafiltration using the dialysis membran e. The pressure on the dialysate side is lower so water moves from the blood (place of higher pressure) to the dialysate (place of lower pressure). This is how the hemodialysis treatment removes fluid.
What can cause ultrafiltration to fail?
Some things that can cause ultrafiltration to fail include uremia (high blood urea nitrogen), peritonitis (infection of the peritoneal membrane), and high dextrose PD solution (especially 4.25%). These factors cause inflammation of the peritoneal membrane.
What is CRRT in ICU?
Continuous Renal Replacement Therapy (CRRT) is a treatment option for patients in need of dialysis or fluid removal. It is typically only utilized in the ICU setting and patients require this particular therapy because of their hemodynamic instability. CRRT is a much slower type of dialysis than regular HD, as it pulls fluid or cleans the blood continuously, 24 hours a day, rather than over a 2-4 hr treatment. Some facilities only use this treatment option in ICU patients with renal failure, even if they are hemodynamically stable. This type of therapy relies on the bedside nurse, who has special training in this technology and the equipment. It requires you to be aware of how the patient responds to the treatment both metabolically and hemodynamically at all times.
What happens if a CRRT circuit stops cycling?
The bedside nurse should always have saline flushes and heparin (or the anticoagulant ordered) within reach if the machine decides to clot off and stop cycling the blood. If the machine stops cycling and appears to be trying to clot, the patient’s blood should be returned and therapy discontinued with the current circuit. Each circuit can hold approximately 150-250 mL blood, which can add up, especially if circuits are replaced 2-3 times a shift.
When in doubt of the circuit clotting, what to do?
When in doubt of the circuit clotting, rinse back. If the patient is getting more hypotensive or unstable, stop treatment and rinse back.
Can a fistula be used with a CRRT?
Fistulas are common for established dialysis patients but cannot be used with CRRT. This is because it is pump-driven and incorporates venovenous extracorporeal circuits. There is a red and a blue line, but it will both be venous and should never be arterial. Once the line is established, it needs to be anticoagulated as soon as it is inserted. The amount of required heparin will be labeled on each port hub. CRRT machine orders will be given by the nephrologist and can be adjusted during the treatment. Patients will either run with a positive, negative, even, or zero fluid balance. Blood flow rate can be adjusted to help prevent clotting, and dialysate rate is usually calculated depending on patient weight and electrolytes. Lab orders and ABGs are followed based on physician preference, patient condition, and/or for any acute change.
