Three of the four major classes of biological macromolecules (complex carbohydrates, nucleic acids, and proteins), are composed of monomers that join together via dehydration synthesis reactions. Complex carbohydrates are formed from monosaccharides, nucleic acids are formed from mononucleotides, and proteins are formed from amino acids.
Do all macromolecules use dehydration synthesis?
Dehydration and hydrolysis reactions are similar for all macromolecules, but each monomer and polymer reaction is specific to its class. Dehydration reactions typically require an investment of energy for new bond formation, while hydrolysis reactions typically release energy by breaking bonds.
What molecules undergo dehydration synthesis?
Complex carbohydrates, nucleic acids, and proteins are all examples of polymers that are formed by dehydration synthesis. Monomers like glucose can join together in different ways and produce a variety of polymers.
Do lipids use dehydration synthesis?
And, dehydration synthesis reactions are also involved in the assembly of certain types of lipids, even though the lipids are not polymers 3).
How do proteins use dehydration synthesis?
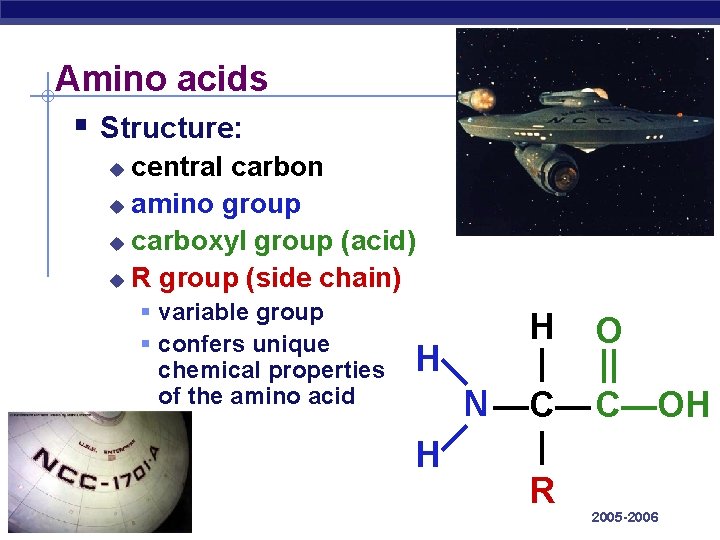
Amino acids are attached to other amino acids by covalent bonds, known as peptide bonds, which are formed by dehydration synthesis reactions. The carboxyl group of one amino acid and the amino group of the incoming amino acid combine, releasing a molecule of water and forming a peptide bond (Figure 3.7).
How does dehydration synthesis work in carbohydrates?
Dehydration synthesis involves the formation of new chemical bonds between two molecules which leads to the formation of new compounds. A reaction occurs with the loss of water molecules at each step. The loss of water molecules can occur due to reactions between two functional groups like –OH, -NH2 or –COOH.
Do ribosomes use dehydration synthesis?
Answer: The cell organelle that performs condensation and dehydration synthesis of the polypeptide is Ribosomes.
What types of molecules are used for the synthesis of a lipid?
Lipids are important energy storage molecules and the major constituent of cell membranes. They are synthesized from acetyl CoA, which is formed from the breakdown of carbohydrates, in a series of reactions that resemble the reverse of fatty acid oxidation.
What is the synthesis of lipids?
Structural lipids are mostly synthesized in the endoplasmic reticulum (ER), from which they are actively transported to the membranes of other organelles.
What are the macromolecules?
Macromolecules are basically polymers, long chains of molecular sub-units called monomers. Carbohydrates, proteins and nucleic acids are found as long polymers. Due to their polymeric nature and large size, they are known as macromolecules.
What functional group is key to dehydration synthesis of both fats and proteins?
Hydroxyl groups are polar. They are components of all four types of organic compounds discussed in this chapter. They are involved in dehydration synthesis and hydrolysis reactions. Carboxyl groups are found within fatty acids, amino acids, and many other acids.
Why is building a protein molecule called dehydration synthesis?
Dehydration means "loss of water". Synthesis means "to put together". Explain why the chemical process responsible for building a protein molecule is called dehydration synthesis. When the chemicals of water are put together they break down and are taken away from the orginal structure.
How does dehydration synthesis help organisms store and use glucose?
Explanation: When your body is queued by insulin, it removes glucose from the bloodstream and stores it as the molecule glycogen (animal starch) - many glucose are linked together into a chain through dehydration synthesis.
What functional groups can undergo hydrolysis?
Hydrolysis has been observed for many drugs. This can occur for compounds that contain the functional groups: ester, amide, thioester, imide, imine, carbamate, acetal, alkyl chloride, nitrate, lactam, lactone, and sulfonamide.
Which of these molecules is not formed by dehydration reactions?
Hence, the correct answer is 'Collagen'
Which bond is not formed by dehydration?
The H (hydrogen) molecule comes from one unit and OH (hydroxyl) comes from the other unit. Complete answer: A dehydration reaction is observed in the formation of all except collagen.
What two types of molecules are needed to form a fat molecule?
A fat molecule consists of two kinds of parts: a glycerol backbone and three fatty acid tails. Glycerol is a small organic molecule with three hydroxyl (OH) groups, while a fatty acid consists of a long hydrocarbon chain attached to a carboxyl group.
How are dehydration and hydrolysis similar?
Dehydration and hydrolysis reactions are similar for all macromolecules, but each monomer and polymer reaction is specific to its class. Dehydration reactions typically require an investment of energy for new bond formation, while hydrolysis reactions typically release energy by breaking bonds.
What are macromolecules in biology?
As you’ve learned, biological macromolecules are large molecules, necessary for life, that are built from smaller organic molecules. There are four major classes of biological macromolecules (carbohydrates, lipids, proteins, and nucleic acids); each is an important cell component and performs a wide array of functions. Combined, these molecules make up the majority of a cell’s dry mass (recall that water makes up the majority of its complete mass). Biological macromolecules are organic, meaning they contain carbon. In addition, they may contain hydrogen, oxygen, nitrogen, and additional minor elements.
How is water formed in dehydration?
In a dehydration synthesis reaction (Figure 3.1. 1 ), the hydrogen of one monomer combines with the hydroxyl group of another monomer, releasing a molecule of water. At the same time, the monomers share electrons and form covalent bonds.
What is the difference between dehydration and hydrolysis?
Dehydration and hydrolysis reactions are catalyzed, or “sped up,” by specific enzymes; dehydration reactions involve the formation of new bonds, requiring energy, while hydrolysis reactions break bonds and release energy.
What type of reaction is when polymers are broken down into smaller units?
When polymers are broken down into smaller units (monomers), a molecule of water is used for each bond broken by these reactions; such reactions are known as hydrolysis reactions.
What happens to the hydrogen in a dehydration reaction?
This creates an opening in the outer shells of atoms in the monomers, which can share electrons and form covalent bonds.
What are the four major classes of macromolecules?
Proteins, carbohydrates, nucleic acids, and lipids are the four major classes of biological macromolecules—large molecules necessary for life that are built from smaller organic molecules. Macromolecules are made up of single units known as monomers that are joined by covalent bonds to form larger polymers. The polymer is more than the sum of its ...
What is Dehydration Synthesis?
The hydrogen ion and hydroxide then combine to form water, hence, the components appear to “ lose water ” or “ dehydrate “.
Why is dehydration synthesis called condensation?
Dehydration synthesis is also called as “ condensation reaction ” because both are characterized by the condensation and formation of water from the large molecule.
How is hydrolysis done?
Relative to the process of dehydration synthesis, hydrolysis is merely the reverse. Using water molecules complex molecules are broken down into smaller units. Large molecules are broken down by breaking the bond between water molecules. In this process, a hydrogen ion (H+) is added to one component and a hydroxide ion (OH-) is added to another one.
Why is water important in cellular processes?
In fact, in the presence of water, dehydration synthesis and hydrolysis are the biochemical processes that are used to either build or break down the said biological molecules.
What is hydrolysis in biology?
Since water is split, it is termed as “ hydrolysis ” which literally means “ water separation “. Examples of hydrolytic reactions are the breaking down of complex sugars, proteins, complex fats, and nucleic acids into monosaccharides, amino acids, fatty acids, and nucleotides.
What are the building blocks of dehydration?
Dehydration Synthesis: Despite being very diverse, life can still be broken down into its 4 major building blocks: carbohydrates, proteins, lipids, and nucleic acids. Being a constituent of living organisms, a more general name for this group of organic compounds is biomolecules. These biomolecules are needed for survival: carbohydrates ...
Why do autotrophs use dehydration?
In most autotrophic organisms utilize dehydration synthesis in order to form long chains of small molecules. For instance, this process used for storing excess glucose by converting it to glycogen (storage form of carbohydrates in animals) or starch (storage form of carbohydrates) in plants.
How are dehydration and hydrolysis reactions similar?
These reactions are similar for most macromolecules, but each monomer and polymer reaction is specific for its class. For example, in our bodies, food is hydrolyzed, or broken down, into smaller molecules by catalytic enzymes in the digestive system. This allows for easy absorption of nutrients by cells in the intestine. Each macromolecule is broken down by a specific enzyme. For instance, carbohydrates are broken down by amylase, sucrase, lactase, or maltase. Proteins are broken down by the enzymes pepsin and peptidase, and by hydrochloric acid. Lipids are broken down by lipases. Breakdown of these macromolecules provides energy for cellular activities.
What are macromolecules in biology?
As you’ve learned, biological macromolecules are large molecules, necessary for life, that are built from smaller organic molecules. There are four major classes of biological macromolecules (carbohydrates, lipids, proteins, and nucleic acids); each is an important cell component and performs a wide array of functions. Combined, these molecules make up the majority of a cell’s dry mass (recall that water makes up the majority of its complete mass). Biological macromolecules are organic, meaning they contain carbon. In addition, they may contain hydrogen, oxygen, nitrogen, and additional minor elements.
What is the name of the splitting or lysis of a bond between monomers within a polymer, using?
The remaining oxygen atom is used to link the two monomers together. Hydrolysis is the splitting or lysis of a bond between monomers within a polymer, using water.
How are polymers broken down?
Polymers are broken down into monomers in a process known as hydrolysis, which means “to split with water.” Hydrolysis is a reaction in which a water molecule is used during the breakdown of another compound ( Figure 3.3 ). During these reactions, the polymer is broken into two components: one part gains a hydrogen atom (H+) and the other gains a hydroxyl molecule (OH–) from a split water molecule.
What are the elements that make up the majority of a cell's dry mass?
Combined, these molecules make up the majority of a cell’s dry mass (recall that water makes up the majority of its complete mass). Biological macromolecules are organic, meaning they contain carbon. In addition, they may contain hydrogen, oxygen, nitrogen, and additional minor elements.
What are the four categories of macromolecules?
The four categories of macromolecules are carbohydrates, lipids, proteins, and nucleic acids.
What enzyme breaks down carbohydrates?
For instance, carbohydrates are broken down by amylase, sucrase, lactase, or maltase. Proteins are broken down by the enzymes pepsin and peptidase, and by hydrochloric acid. Lipids are broken down by lipases. Breakdown of these macromolecules provides energy for cellular activities.
How are dehydration and hydrolysis reactions similar?
These reactions are similar for most macromolecules, but each monomer and polymer reaction is specific for its class. For example, catalytic enzymes in the digestive system hydrolyze or break down the food we ingest into smaller molecules. This allows cells in our body to easily absorb nutrients in the intestine. A specific enzyme breaks down each macromolecule. For instance, amylase, sucrase, lactase, or maltase break down carbohydrates. Enzymes called proteases, such as pepsin and peptidase, and hydrochloric acid break down proteins. Lipases break down lipids. These broken down macromolecules provide energy for cellular activities.
What happens to the hydrogen in a dehydration reaction?
This creates an opening in the outer shells of atoms in the monomers, which can share electrons and form covalent bonds.
What happens when polymers break down into smaller units?
When polymers break down into smaller units (monomers), they use a water molecule for each bond broken by these reactions. Such reactions are hydrolysis reactions. Dehydration and hydrolysis reactions are similar for all macromolecules, but each monomer and polymer reaction is specific to its class.
What are macromolecules made of?
Macromolecules are comprised of single units scientists call monomers that are joined by covalent bonds to form larger polymers. The polymer is more than the sum of its parts: it acquires new characteristics, and leads to an osmotic pressure that is much lower than that formed by its ingredients.
How do polymers break down?
Breaking a covalent bond with this water molecule in the compound achieves this ( Figure ). During these reactions, the polymer breaks into two components: one part gains a hydrogen atom (H+) and the other gains a hydroxyl molecule (OH–) from a split water molecule.
What are monomer and polymer reactions?
These reactions are similar for most macromolecules, but each monomer and polymer reaction is specific for its class. For example, catalytic enzymes in the digestive system hydrolyze or break down the food we ingest into smaller molecules. This allows cells in our body to easily absorb nutrients in the intestine.
What are the four macromolecules?
There are four major biological macromolecule classes (carbohydrates, lipids, proteins, and nucleic acids ). Each is an important cell component and performs a wide array of functions. Combined, these molecules make up the majority of a cell’s dry mass (recall that water makes up the majority of its complete mass).
How are dehydration and hydrolysis reactions similar?
These reactions are similar for most macromolecules, but each monomer and polymer reaction is specific for its class, for example, catalytic enzymes in the digestive system hydrolyse or break down the food we ingest into smaller molecules. This allows cells in our body to easily absorb nutrients in the intestine. A specific enzyme breaks down each macromolecule. For instance, amylase, sucrase, lactase, or maltase break down carbohydrates. Enzymes called proteases, such as pepsin and peptidase, and hydrochloric acid break down proteins. Lipases break down lipids. These broken-down macromolecules provide energy for cellular activities.
What are the elements that make up the cell's dry mass?
Biological macromolecules are organic, meaning they contain carbon. In addition, they may contain hydrogen, oxygen, nitrogen, and additional minor elements.
How do polymers break down?
Polymers break down into monomers during hydrolysis. A chemical reaction occurs when inserting a water molecule across the bond. Breaking a covalent bond with this water molecule in the compound achieves this (Figure 2.1.2). During these reactions, the polymer breaks into two components: one part gains a hydrogen atom (H+) and the other gains a hydroxyl molecule (OH–) from a split water molecule.
What is the name of the reaction where two glucose molecules link to form a disaccharide?
Figure 2.1.1. Dehydration synthesis. In the dehydration synthesis reaction above, two glucose molecules link to form the disaccharide maltose. In the process, it forms a water molecule.
What are macromolecules?
As you have learned, biological macromolecules are large molecules, necessary for life, that are built from smaller organic molecules. There are four major biological macromolecule classes (carbohydrates, lipids, proteins, and nucleic acids). Each is an important cell component and performs a wide array of functions.
What are the four major classes of macromolecules?
There are four major biological macromolecule classes (carbohydrates, lipids, proteins, and nucleic acids ).
What enzyme breaks down carbohydrates?
A specific enzyme breaks down each macromolecule. For instance, amylase, sucrase, lactase, or maltase break down carbohydrates. Enzymes called proteases, such as pepsin and peptidase, and hydrochloric acid break down proteins.