- Soil organic matter, for example, contains phosphorus. ...
- Soil pH is another factor that can affect phosphorus uptake Acidic soil contains high levels of iron and aluminum which can tie up phosphorus. ...
- Soils with a higher cation exchange capacity (CEC) and more clay content can hold more nutrients such as iron, aluminum, calcium and magnesium. ...
Why does phosphorus have a low melting point?
Why phosphorus has low melting point? The four molecular elements Their melting or boiling points will be lower than those of the first four members of the period which have giant structures. Phosphorus contains P4 molecules. To melt phosphorus you don’t have to break any covalent bonds – just the much weaker van der Waals forces between the molecules. Why does sulfur have a higher melting point than sodium thiosulfate?
What does high phosphorus indicate?
Other possible causes of hyperphosphatemia include:
- low parathyroid hormone levels (hypoparathyroidism)
- damage to cells
- high vitamin D levels
- diabetic ketoacidosis — high levels of acids called ketones in the blood of people with diabetes
- injuries — including those that cause muscle damage
- serious body-wide infections
What is the best source of phosphorus?
- Dairy: milk, yogurt, cheese
- Salmon
- Beef
- Poultry
- Pork
- Legumes
- Nuts, seeds
- Whole wheat breads and cereals
- Some vegetables: asparagus, tomatoes, cauliflower
- Processed foods (as inorganic phosphorus), especially deli meats, bacon, sausage, sodas, sports drinks, and other bottled beverages
Why does phosphorus have a higher IE than sulfur?
Why does phosphorus have a high ionization energy? Either all unpaired or all paired makes it stable, so if phosphorus is all unpaired it take more energy to remove an electron than it would from sulfur in which one electron can be removed making it more stable. Because it takes more energy for phosphorus, it has a higher ionization energy.

How do I know if my soil has too much phosphorus?
The main symptom of excessive phosphorus in soil is stunted plant growth. High P interferes with N absorption. Also there may be symptoms of deficiencies of zinc, iron, cobalt or calcium, because the P has locked up these nutrients.
How do you get rid of high phosphorus in soil?
0:101:46How to Lower Phosphorus in Soil : Green Savvy - YouTubeYouTubeStart of suggested clipEnd of suggested clipDesign. Today I'm going to talk about how to lower the phosphorus in your soil. Now often times youMoreDesign. Today I'm going to talk about how to lower the phosphorus in your soil. Now often times you get high phosphorus by over fertilizing or by adding certain types of manure. So what you need to do
How do you fix high phosphorus levels?
Here are seven methods to help control high levels of phosphorus:Reduce the amount of phosphorus you eat. ... Take phosphorus binders. ... Take vitamin D. ... Take a calcimimetic medicine. ... Stay on dialysis the entire time. ... Start an exercise program approved by a doctor. ... Get an operation to remove some of the parathyroid glands.
What will neutralize phosphorus?
The most common metal salts used are in the form of alum (aluminum sulfate), sodium aluminate, ferric chloride, ferric sulfate, ferrous sulfate, and ferrous chloride. Chemical treatment is the most common method used for phosphorus removal to meet effluent concentrations below 1.0 mg/L.
How can I lower my phosphorus naturally?
Lower-phosphorus alternatives to choose:Fresh fruits and vegetables.Rice milk, unenriched.Breads.Pasta.Rice.Fish.Corn and rice cereals.Soda without phosphate additives.More items...•
How long does phosphorus stay in the soil?
Phosphorus will be most available to the plant within a few day s to two weeks after fertilizer addition, slowly dropping as time goes on. When applied in the fall, P will stay in the soil for as long as four to six months before plant uptake.
What do high phosphorus levels mean?
High phosphorus, also called hyperphosphatemia, means you have extra phosphorus in your blood. High phosphorus is often a sign of kidney damage. For someone with healthy kidneys, the normal amount of phosphorus (phosphate) in the blood is between 2.5 to 4.5 mg/dL.
What should phosphorus levels be in soil?
25 to 50 ppmHealthy levels of P in soil ranges from 25 to 50 ppm. The University of Nebraska-Lincoln gives the following guidelines to consider when analyzing your P levels in soil: Healthy levels between 25 and 50 ppm should receive light maintenance application of phosphorus annually (1/8-1/4 lb.
What percentage of phosphorus is in soil?
Approximately 30 to 65 percent of total soil phosphorus is in organic forms, which are not plant available, while the remaining 35 to 70 percent is in inorganic forms. Organic forms of phosphorus include dead plant/animal residues and soil micro-organisms.
Which soil property favors phosphorus adsorption?
Another soil property that favors phosphorus adsorption is the clay content. Soils with greater clay content have higher adsorption capacity than coarse textured sandy soils.
How is phosphorus converted into inorganic phosphorus?
Immobilization, on the other hand, is the reverse of mineralization. During immobilization, inorganic phosphorus forms are converted back to organic forms and are absorbed into the living cells of soil microbes. Immobilization typically occurs when crop residues are incorporated in the soil. As crop residues decompose, more phosphorus becomes available in the soil solution through mineralization. Because mineralization and immobilization processes are biological processes, they are highly influenced by soil moisture, temperature, pH, organic carbon to organic phosphorus ratio of crop residues, microbial population, etc.
What is phosphorus pool?
Plant-available (soil solution) phosphorus: This pool is comprised of inorganic phosphorus dissolved in water/soil solution that is readily available for plant uptake.
What is the total amount of phosphorus in soil?
Although total soil phosphorus is generally high, with concentrations ranging from 200 to 6,000 pounds per acre, 80 percent of this phosphorus is immobile and not available for uptake by the plant.
Why is clay good for soil?
Soils with higher clay content have high phosphorus retention capacity because clay particles have very large surface area per unit volume, which can adsorb phosphorus easily.
Why is organic matter important?
Organic matter is an important factor in controlling phosphorus availability. With the addition of organic matter, availability of phosphorus increases. This is due to the following reasons: Mineralization of organic matter releases plant- available forms of phosphorus into soils.
Which soils have the highest phosphorus sorption capacity?
Volcanic soils and highly weathered soils (such as Ultisols and Oxisols) have high phosphorus sorption capacity and thus lower phosphorus availability.
Why do plants take up phosphorus?
Despite the relatively small amount of phosphate in the soil solution at any given time, plants can take up substantial amounts due to replenishment of the solution phosphorus pool through desorption from soil particles and dissolution from soil minerals.
What is the phosphate that is adsorbed to soil particles?
Consists of inorganic phosphate adsorbed to soil particles, secondary phosphate minerals (phosphate bound to cations such as calcium and aluminum), and organic phosphorus that is readily mineralized.
What is the term for the release of phosphorus from soil?
Dissolution – Release of phosphorus that occurs when soil minerals dissolve. Occurs slowly over long periods of time.
How long does phosphorus stay in the pool?
Dissolution of fixed phosphorus into the active pool occurs very slowly over time; phosphorus can remain in the fixed pool for years and have little effect on the fertility of the soil.
What is the simplest form of phosphorus?
The simplest form of phosphate is PO 4-3 (orthophosphate), which is the predominant form of phosphorus taken up by plants.
What is the term for the binding of phosphates to soil particles?
Adsorption – Binding of phosphates to soil particles; also referred to as fixation.
Why is phosphorus important for plants?
Phosphorus is needed for cell division, hence to promote root formation and growth, vigorous seedlings, flowering, crop maturity and seed production, and to improve winter hardiness in fall plantings. Phosphorus is important in fat, carbon, hydrogen and oxygen metabolism, in respiration, and in photosynthesis.
Why is salt bad for soil?
Some of the salt problems are caused by having very high organic matter levels , due to heavy amendment with composts or manures. In high OM soils, when warm or when irrigated after a dry spell, large flushes of nitrate can occur. This makes it difficult to manage nitrogen levels.
What are some organic soil amendments?
Organic soil amendments include soil inoculants, organic mulches, biochar and other soil conditioners, lime and other natural minerals, manure and other organic fertilizers, such as alfalfa meal, soybean meal, fish meal, kelp, composted chicken litter and compost. Screening compost from a huge pile, for spring seedling compost.
How to reduce excess soil P?
The quickest way to reduce excess soil P (which can take years!) is to stop any manure or compost application while continuing to grow crops that can be eaten or sold. One solution for vegetable growers may be to grow cover crops as forage crops, and graze or bale grass crops to sell off the farm as livestock feed.
How does compost help plants?
Compost can add a range of beneficial bacteria and fungi, which can inoculate plants against diseases by inducing systemic acquired resistance. The plants produce antibodies and other protective compounds before any infection can occur. Compost improves the soil structure, organic matter and humus.
What is the P pool in soil?
In the soil water, P is only present in very small amounts, but when removed by plants, supplies are quickly replenished from the “active P pool” (P in solid compounds which is relatively easily released to the soil solution). The “fixed P pool” contains inorganic phosphate compounds that are very insoluble, and organic compounds that are resistant to mineralization by microorganisms in the soil. Although some slow conversion between the fixed P pool and the active P pool does occur in the soil, phosphate may remain for years without being available to plants and may have very little impact on soil fertility.
Why are salts bad for plants?
Some of the salt problems are caused by having very high organic matter levels, due to heavy amendment with composts or manures.
Why is phosphorus in my soil?
High phosphorous levels in your soil are usually the culprit of over-fertilizing or adding too much manure. Not only does excessive phosphorous harm plants, it can also stay in your soil for years.
What is the first number in phosphorous free fertilizer?
Add phosphorous-free fertilizer. All fertilizers contain a string of numbers that follow this format: "X-X-X." The first number is nitrogen, the second phosphorous and the third potassium. Phosphorous-free fertilizers will always have a "0" in the second column.
What is the first number in fertilizer?
All fertilizers contain a string of numbers that follow this format: "X-X-X.". The first number is nitrogen, the second phosphorous and the third potassium. Phosphorous-free fertilizers will always have a "0" in the second column. Spray foliar zinc and iron on plants in high phosphorous soils.
What vegetables can you plant to increase nitrogen?
Plant nitrogen-fixing vegetables to increase nitrogen without increasing phosphorous. Examples of such vegetables include beans and peas. If nitrogen levels are extremely low, planting nitrogen-fixing vegetables alone will not fix your problem.
Can you add manure to fertilizer?
Avoid adding manure as fertilizer. Manure is typically high in phosphorous and can quickly lead to a spike in phosphorous levels. This is especially true if manure is being added to increase nitrogen levels in the soil. Plants can usually remove slightly excessive amounts of phosphorous, but there's a limit to how much phosphorous each plant can ...
Why is phosphorus important in Minnesota?
In Minnesota, most agricultural soils contain between 100 and about 4,000 pounds of total P per acre. Efficiently using this phosphorus is becoming more important due to economic and environmental concerns .
What is the pH of Minnesota soil?
To determine the available phosphorus (P) of Minnesota plants, use the Bray-1 test when soil pH is 7.4 or less and the Olsen test when soil pH exceeds 7.4. Soil has both organic and inorganic P forms, ...
Why is the soil in western Minnesota so low?
Soils throughout most of western Minnesota usually have natively low levels of available P because of the materials of the soils it’s formed in. Appropriately managing phosphate fertilizers is a major concern for these soils.
What is soil test used for in Minnesota?
To determine the need for supplemental P, soil tests are often used to estimate how much phosphate will be available for a crop. The two most common types of soil tests in Minnesota are:
Why is the P cycle similar to other mineral nutrient cycles?
The P cycle is similar to several other mineral nutrient cycles because it exists in soils, minerals, living organisms and water. Phosphorus serves a unique purpose, wherever it is in the cycle. Rain and weathering of rocks cause phosphate ions to release, which are then distributed in soils and water.
How many pools of P are there in soil?
However, in practical terms, think of P in soils as existing in three pools: The solution P, active P and fixed P pools.
Which element reacts with calcium or aluminum to form soluble solids?
Phosphate that reacted with elements such as calcium or aluminum to form somewhat soluble solids.
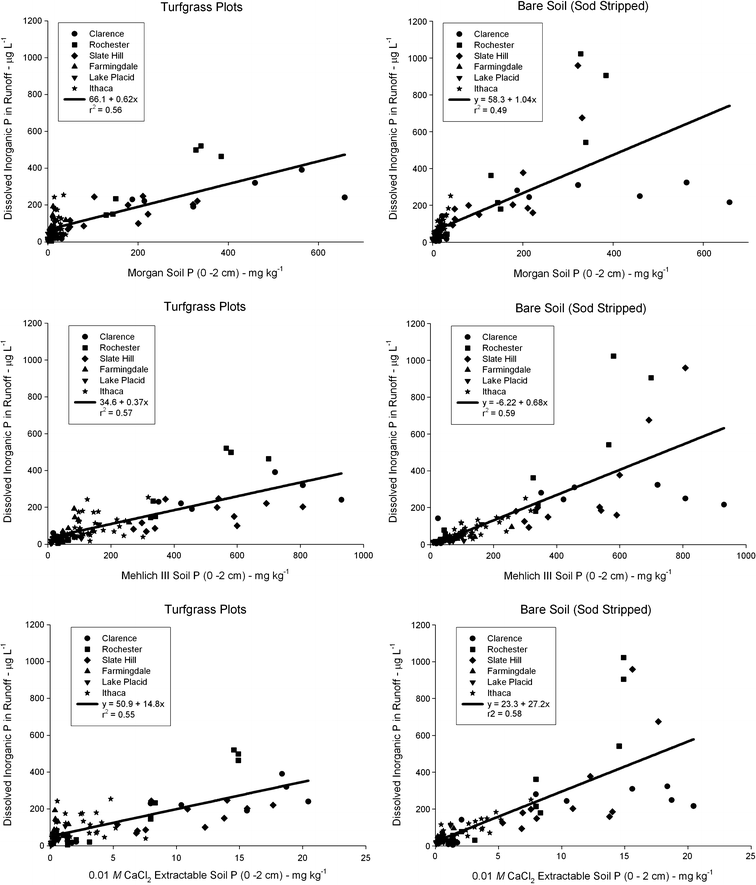
Introduction
Five of the eight eastern panhandle counties in West Virginia’s portion of the Chesapeake Bay watershed include 98% of the state’s poultry industry.
Results
To help farmers in the eastern panhandle better understand the environmental risks of phosphorus saturated soils, a WVU student was hired in the fall of 2019 to collect soil samples from crop, hay and pasture fields. Our survey area included Grant, Hardy, Mineral, Hampshire, Pendleton and Jefferson counties.
Conclusions
Having farmers better understand phosphorus soil chemistry is key to reducing or eliminating applications of poultry litter to phosphorus-saturated fields. The Mehlich 3 soil test enables famers to identify fields that are vulnerable to phosphorus losses and then, take steps to manage the fields to minimizes losses.
What causes phosphorus to accumulate in soil?
The accumulation of phosphorus is conditioned by the excessive use of inorganic fertilizers or the application of phosphorus-rich compounds and manure. High levels of phosphorus in soil may also pose a threat to streams, rivers, lakes, and oceans. Phosphorus can become water-soluble and mobile, affecting surface waters and causing the growth ...
Why is phosphorus in the soil bad for plants?
High levels of phosphorus in the soil are usually the cause of over-fertilization or the addition of too much manure. Excessive phosphorus is not only harmful to plants but can also remain in the soil for many years.
Why do my azaleas have yellow leaves?
Control and correction of high phosphorus content in the soil are essential for optimal plant growth in your garden. Excess soil phosphorus causes the leaves of plants to turn yellow or bleach , especially in species that do not have deep roots. Excess phosphorus sometimes kills even plants that need acidic soil, such as azaleas.
What is soil phosphorus test?
A soil phosphorus test is essentially an indicator to help predict the fertilizer requirements of a crop. Recommendations on the application of fertilizer are based on numerous field tests on many soils and crops.
What are the nutrients that grow in a garden?
Analyzing and maintaining the right amount of nutrients in the soil is an important aspect of growing a beautiful garden. Phosphorus, potassium, and Nitrogen are essential nutrients in plant growth. While nitrogen helps plants produce lush foliage and leaves, phosphorus helps plants flower and produce strong seeds and roots.
How to remove phosphorus from soil?
Watering the soil – by spraying carefully , phosphorus is removed from the soil. Be careful with this method, because it also removes other important nutrients. Then test the soil with a mud test kit to check the effectiveness of this method. I recommend it as the last test in the trench.
What to do if your soil has low phosphorus?
Stop adding organic products that increase the phosphorus content in the soil. The worst culprits are manure and other organic compounds. If you need organic mulch or nitrogen generator, replace pine bark or blood meal. These products have low phosphorus content.