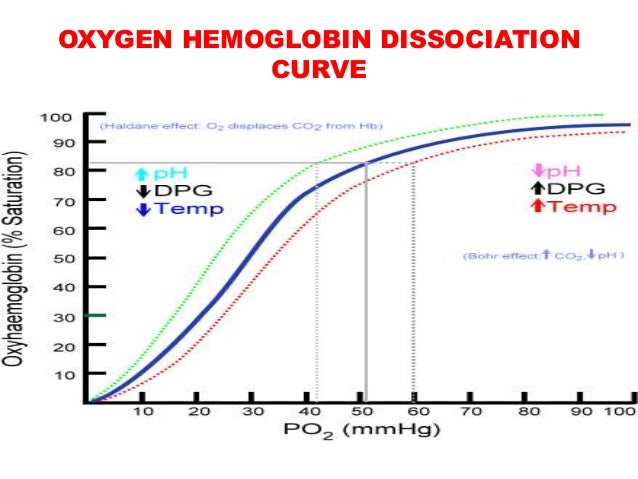
Factors that affect the standard dissociation curve
| Control factors | Change | Shift of curve |
| Temperature | ↑ | → |
| Temperature | ↓ | ← |
| 2,3-BPG | ↑ | → |
| 2,3-BPG | ↓ | ← |
When does the dissociation curve shifts to the left?
When hemoglobin-oxygen affinity increases, the oxyhemoglobin dissociation curve shifts to the left and decreases p50. When hemoglobin-oxygen affinity decreases, the oxyhemoglobin dissociation curve shifts to the right and increases p50 (Figure 1).
What does an oxygen dissociation curve show?
P 50
- Temperature
- Carbon Dioxide
- 2,3-DPG
What is the oxyhemoglobin dissociation curve?
The oxyhemoglobin dissociation curve (ODC) is one of the most recognized teachings of basic physiology. It describes the relationship between the saturation of hemoglobin and the partial pressure of arterial oxygen. Intuitively, it makes sense that the more oxygen that’s available (a higher PO2), the more saturated hemoglobin will be (% saturation).
Why is that oxygen dissociation curve sigmoid?
The oxygen dissociation curve has a sigmoid shape because of the co-operative binding of oxygen to the 4 polypeptide chains. Co-operative binding means that haemoglobin has a greater ability to bind oxygen after a subunit has already bound oxygen. Haemoglobin is, therefore, most attracted to oxygen when 3 of the 4 polypeptide chains are bound ...

What causes a shift in the oxygen dissociation curve?
Factors which result in shifting of the oxygen-dissociation curve to the right include increased concentration of pCO2, acidosis, raised temperature and high concentrations of 2,3 diphosphoglycerate (2,3 DPG). These factors, in effect, cause the Hb to give up oxygen more readily.
What affects dissociation curve shift?
The oxygen–hemoglobin dissociation curve can be displaced such that the affinity for oxygen is altered. Factors that shift the curve include changes in carbon dioxide concentration, blood temperature, blood pH, and the concentration of 2,3-diphosphoglycerate (2,3-DPG).
What does the right and left shift of oxygen dissociation curve indicate?
A rightward shift of the curve indicates that hemoglobin has a decreased affinity for oxygen, thus, oxygen actively unloads. A shift to the left indicates increased hemoglobin affinity for oxygen and an increased reluctance to release oxygen.
Which change would cause a shift to the left in the oxygen hemoglobin binding curve?
Leftward shifts indicate an increased affinity for the binding of hemoglobin, so that more oxygen binds to hemoglobin, but less oxygen is unloaded from it into the tissues. Causes of leftward shifts include increased blood pH, decreased temperature, and carbon monoxide exposure.
Which change would cause a shift to the left in the oxygen hemoglobin binding curve quizlet?
What causes a left shift in the hemoglobin-O2 dissociation curve? Fetal hemoglobin is shifted to the left relative to adult hemoglobin, primarily due to a decrease in the affinity of fetal hemoglobin for 2, 3-BPG.
What does a shift to the left mean dissociation curve?
increased oxygen affinityThe oxygen dissociation curve can be shifted right or left by a variety of factors. A right shift indicates decreased oxygen affinity of haemoglobin allowing more oxygen to be available to the tissues. A left shift indicates increased oxygen affinity of haemoglobin allowing less oxygen to be available to the tissues.
Which of the following will shift the o2 dissociation curve to the left quizlet?
'An increase in H+ concentration and decrease in pH will shift hemoglobin's oxygen-binding curve to the right, while a decrease in H+ concentration will shift the curve to the left'.
What shifts ODC to the right?
Deep hypoxia is known to increase the intraerythrocytic 2,3 diphosphoglycerate (DPG) level and therefore to induce a right shift of the oxyhemoglobin dissociation curve (ODC), which is considered to be a protective mechanism against tissular hypoxia.
Which of the following will not cause the oxygen-hemoglobin dissociation curve to shift to the right?
Correct answer: The oxygen partial pressure will affect the saturation percentage of hemoglobin in the blood, but will not shift the curve itself.
How does altitude affect oxygen dissociation curve?
As altitude increases or exercise intensity increases, partial pressure of oxygen (PaO2) decreases as a result. In response to this lowered oxygen pressure, hemoglobin releases more bound O2. It is apparent that the dissociation curve follows a curvilinear, downward direction (Fig.
What does right shift in dissociation curve mean?
decreased oxygen affinityThe oxygen dissociation curve can be shifted right or left by a variety of factors. A right shift indicates decreased oxygen affinity of haemoglobin allowing more oxygen to be available to the tissues. A left shift indicates increased oxygen affinity of haemoglobin allowing less oxygen to be available to the tissues.
What causes the shift of the oxygen dissociation curve to the right?
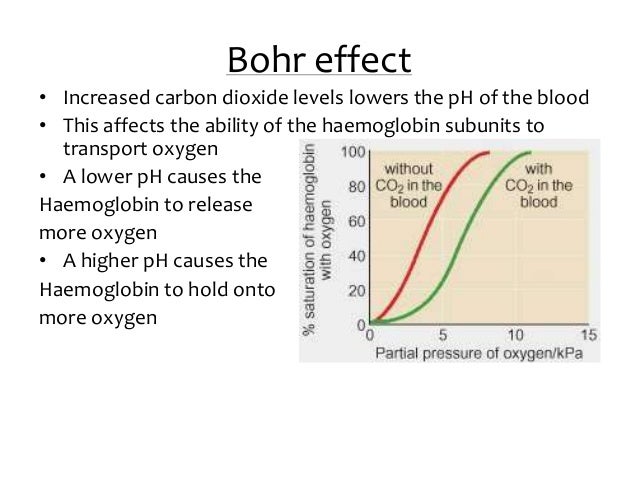
The shift of the oxygen dissociation curve to the right occurs in response to an increase in the partial pressure of carbon dioxide (Pco2 ), a decrease in pH , or both , the last of which is known as the Bohr effect.
What is the oxygen dissociation curve?
The oxygen dissociation curve is the expression of the relationship between the partial pressure of oxygen and oxygen saturation of haemoglobin. Details of this relationship and the physiological importance of changes in this relationship were worked out in detail at the beginning of the last century by the great physiologists Hüfner, Bohr, Barcroft, Henderson and many others. Their work was summarised by Peters and Van Slyke in Quantitative Clinical Chemistry. 58 The relevant chapters of this book have been reprinted and it would be difficult to improve their description of the importance of the oxygen dissociation curve.
What is the S-shaped oxyhaemoglobin dissociation curve?
The Kelman equation, which uses seven coefficients, generates a curve indistinguishable from the true curve above a Po2 of about 1 kPa (7.5 mm Hg) and this has remained the standard. Calculation of Po2 from saturation requires an iterative approach, but saturation may be conveniently determined from Po2 by computer, a calculation that is automatically performed by most blood gas analysers in clinical use. The following simplified version of the Kelman equation is convenient to use and yields similar results at Po2 values greater than 4 kPa (30 mm Hg): 13
How does acidosis affect oxygen delivery?
Acidosis causes a right shift in the oxyhaemoglobin dissociation curve (the Bohr effect, see p. 90) and this facilitates oxygen delivery to tissues. An increase in hydrogen ion concentration decreases erythrocyte 2,3-diphosphoglycerate (2,3-DPG) concentrations through effects on both synthesis and breakdown; this causes a left shift in the curve, but, whereas the Bohr effect is immediate, the fall in 2,3-DPG takes place over a matter of hours. The reverse is also true; so, if hydrogen ion concentration is restored to normal rapidly, oxygen delivery will be compromised until 2,3-DPG concentrations are restored to normal. This is a potential hazard if an attempt is made to correct an acidosis rapidly by the intravenous infusion of bicarbonate.
What does it mean when the hemoglobin curve is left?
A shift to the left indicates increased affinity and so an increased tendency for haemoglobin to take up and retain oxygen.
How much oxygen is in a 100 ml blood sample?
The oxygen content is about 20 ml, when is hemoglobin at 15 g per 100 ml of blood is 100% saturated with oxygen. It has the characteristic advantages of taking in oxygen via the lungs and dissociation of oxygen in organs.
Where is oxygen transported?
Oxygen and carbon dioxide are transported in the blood as a result of changes in blood partial pressures ( Figure 5.1 ). Most oxygen is taken into the hemoglobin in red blood cells, although trace levels of oxygen exist in the dissolved form.
Which direction does oxygen affinity shift?
Oxygen affinity for haemoglobin plays a critical role in the delivery of oxygen to the tissues and is changed by shifting to the left or right. A shift to the left implies an increased oxygen affinity and, hence, tighter binding due to the higher oxygen saturation in relation to the pO2.
What is the ODC curve?
The oxygen dissociation curve: quantifying the shift. An oxyhaemoglobin dissociation curve (ODC) quantifies the most important function of red blood cells and that is the affinity for oxygen and its delivery to the tissues. Oxygen affinity for haemoglobin plays a critical role in the delivery of oxygen to the tissues and is changed by shifting ...
What does it mean when the ODC shifts to the right?
On the other hand, a shift to the right corresponds to a decreased oxygen affinity and easier release of oxygen to the tissues. It is well known that the ODC shifts in response to changes in pH, pCO2 and 2,3 diphosphoglycerate. However, how much the ODC shifts has never been quantified.
Overview
Factors that affect the standard dissociation curve
The strength with which oxygen binds to hemoglobin is affected by several factors. These factors shift or reshape the oxyhemoglobin dissociation curve. A rightward shift indicates that the hemoglobin under study has a decreased affinity for oxygen. This makes it more difficult for hemoglobin to bind to oxygen (requiring a higher partial pressure of oxygen to achieve the same oxygen saturation), but it makes it easier for the hemoglobin to release oxygen bound to it. The …
Background
Hemoglobin (Hb) is the primary vehicle for transporting oxygen in the blood. Each hemoglobin molecule has the capacity to carry four oxygen molecules. These molecules of oxygen bind to the iron of the heme prosthetic group.
When hemoglobin has no bound oxygen, nor bound carbon dioxide, it has the unbound conformation (shape). The binding of the first oxygen molecule induces change in the shape of t…
Sigmoid shape
The curve is usually best described by a sigmoid plot, using a formula of the kind:
A hemoglobin molecule can bind up to four oxygen molecules in a reversible method.
The shape of the curve results from the interaction of bound oxygen molecules with incoming molecules. The binding of the first molecule is difficult. Howeve…
Fetal hemoglobin
Fetal hemoglobin (HbF) is structurally different from normal adult hemoglobin (HbA), giving HbF a higher affinity for oxygen than HbA. HbF is composed of two alpha and two gamma chains whereas HbA is composed of two alpha and two beta chains. The fetal dissociation curve is shifted to the left relative to the curve for the normal adult because of these structural differences.
See also
• Automated analyzer
• Bohr effect
External links
• Nosek, Thomas M. "Section 4/4ch5/s4ch5_18". Essentials of Human Physiology. Archived from the original on 2016-03-24.
• The Interactive Oxyhemoglobin Dissociation Curve
• Simulation of the parameters CO2, pH and temperature on the oxygen–hemoglobin dissociation curve (left or right shift)