
Many factors influence the secretion of vasopressin:
- Ethanol (alcohol) reduces the calcium-dependent secretion of AVP by blocking voltage-gated calcium channels in neurohypophyseal nerve terminals in rats. [23]
- Angiotensin II stimulates AVP secretion, in keeping with its general pressor and pro-volumic effects on the body. [24]
- Atrial natriuretic peptide inhibits AVP secretion, in part by inhibiting Angiotensin II-induced stimulation of AVP secretion. ...
What stimulates the release of ADH and vasopressin?
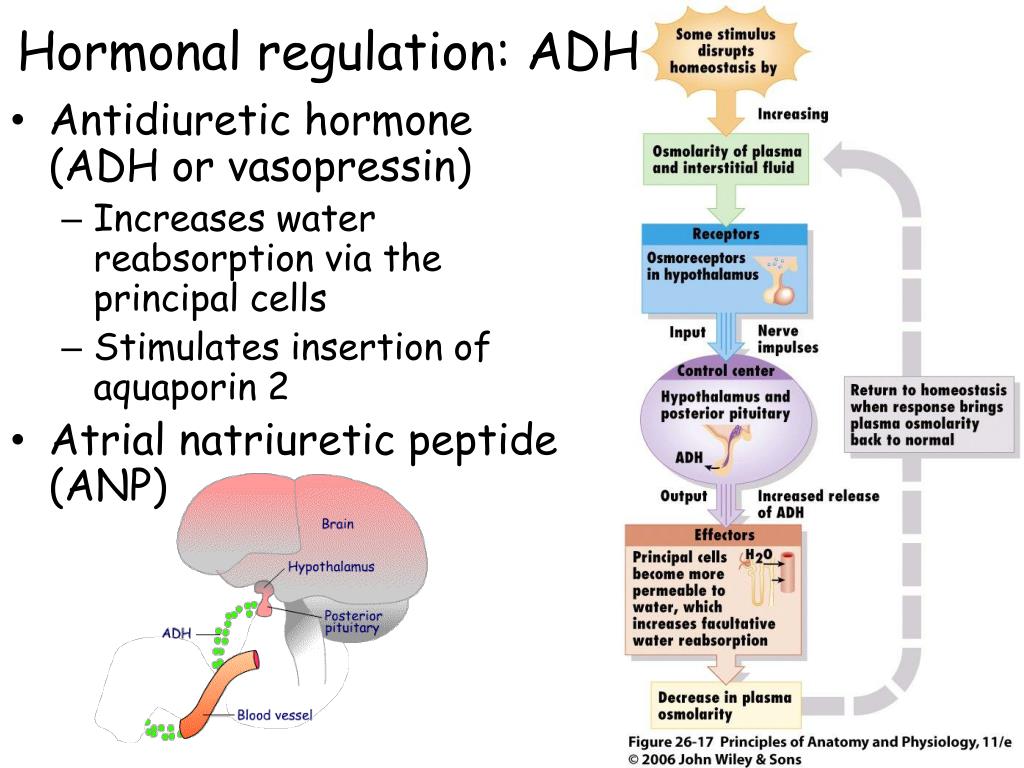
The decrease in the arteriole volume of blood stimulates the secretion of the vasopressin. The ADH that is measured in the peripheral blood is derived from the secretion of vasopressin from the region of the posterior pituitary gland. Cortisol can inhibit the release of ADH.
What is the function of vasopressin hormone?
Vasopressin is a hormone of the posterior pituitary that is secreted in response to high serum osmolarity. Excitation of atrial stretch receptors inhibits vasopressin secretion. Vasopressin is also released in response to stress, inflammatory signals, and some medications.
What are the physiologic effects of vasopressin?
Physiologic Effects of Vasopressin. Vasopressin causes direct systemic vasoconstriction via the V1Rs. In vitro, AVP is a more potent vasoconstrictor than angiotensin II, norepinephrine, or phenylephrine on a molar basis. It is vital for osmoregulation and maintenance of normovolemia, mediated by the V2Rs.
How is vasopressin released from the hypothalamus?
Vasopressin release is regulated by osmoreceptors in the hypothalamus, which are exquisitely sensitive to changes in plasma osmolality. Under hyperosmolar conditions, osmoreceptor stimulation leads to vasopressin release and stimulation of thirst.
See more
How does vasopressin affect the kidneys?
Vasopressin has its major effect in maintaining the osmolality of the extracellular compartment. Vasopressin selectively raise s free water reabsorption in the kidneys and results in blood pressure elevation (Elliot et al, 1996). Plasma levels of vasopressin are markedly elevated in the neonate, especially after vaginal delivery, and its cardiovascular actions facilitate neonatal adaptation (Pohjavuori and Raivio, 1985). The high vasopressin levels are in part also responsible for the diminished urine output of the healthy term neonate during the first day of life. Under certain pathologic conditions, the dysregulated release of, or the end-organ unresponsiveness to, vasopressin significantly affects renal and cardiovascular functions and electrolyte and fluid status in the sick preterm and term infant. In the syndrome of inappropriate secretion of antidiuretic hormone (SIADH), an uncontrolled release of vasopressin occurs in sick preterm and term infants, with resulting water retention, hyponatremia, and oliguria. In the syndrome of diabetes insipidus (DI), the lack of pituitary production of vasopressin or renal unresponsiveness to vasopressin results in polyuria, with increased thirst and hypernatremia.
What is the most important action of vasopressin?
The most important action of vasopressin is its antidiuretic action on the collecting ducts of the kidney.
How to identify AVP neurons?
Methods of identifying AVP-producing neurons include immunocytochemical localization of AVP, AV P-associated neurophysin (neurophysin II), or the carboxyl-terminal glycopeptide of propressorphysin (copeptin) Box 9.1. 6 Further techniques involve the in situ detection of AVP mRNA. However, these methods often fail to identify nerve terminals and thin fibers with very low amounts of peptide and little if any mRNA. The problem may be overcome by expressing highly fluorescent proteins under the control of a tissue-specific AVP promoter and map AVP projections with higher sensitivity as previously accomplished for OXT neurons. 39 However, as yet, this approach has not been optimized to be sufficiently specific for the detection of AVPergic nerve fibers throughout the brain. 23,108
What is the AVP?
Vasopressin (AVP) is a polypeptide containing nine amino acids, with a disulfide bridge between cysteine residues , which has important antidiuretic and vasopressor actions and a variety of other actions including glycogenolysis.
What hormone is released in response to stress?
Vasopressin is a hormone of the posterior pituitary that is secreted in response to high serum osmolarity. Excitation of atrial stretch receptors inhibits vasopressin secretion. Vasopressin is also released in response to stress, inflammatory signals, and some medications.
Which receptor is responsible for vasopressin?
Each receptor type is responsible for a specific function of vasopressin. For example, V1a receptor activation triggers vasoconstriction, V1b receptor is responsible for modulation of mood and behavior, and V2 receptor induces water reabsorption in the kidney.
What is the net effect of reabsorption of water into the blood?
The net effect is the reabsorption of water into the blood, which, along with thirst-generated water intake, leads to normalization of plasma osmolality.
How does ADH affect kidneys?
ADH increases the water permeability of the late distal tubule (or connecting duct) and all parts of the collecting duct. It also increases the urea permeability of the inner medullary collecting duct. When the distal nephron permeability to water and urea is high, all of the mechanisms for concentrating the urine operate, and the kidneys excrete a small volume of highly concentrated urine. In the absence of ADH, the distal nephron is not permeable to either water or urea. Fluid that enters the distal tubule is hypoosmotic, about 100 mOsM. Low water permeability prevents water from leaving the hypoosmotic fluid to equilibrate with the interstitial fluid. Thus the tubular fluid stays hypoosmotic all the way through the collecting duct. Because the late distal tubule and collecting duct can pump out some Na +, the final concentration of the urine is actually lower than 100 mOsM. The kidneys can maximally dilute urine to about 70 mOsM. The low urea permeability in the absence of ADH shuts off the urea recycling, so that the concentration profile in the kidney interstitium is changed. Figure 7.5.11 shows approximate values for tubular fluid concentration along the nephron in the presence and absence of ADH.
What causes SIADH in dogs?
SIADH can be caused by cerebral disorders, pulmonary disease, or adverse effects of medications ( Box 68-1 ). 7 The cause in some cases remains idiopathic. Three cases of idiopathic SIADH have been reported in dogs. 2,3 Cerebral causes of SIADH in humans include hypothalamic tumors, head trauma, meningitis, encephalitis, cerebrovascular accidents, and hydrocephalus. Hypothalamic tumors, granulomatous meningoencephalitis, congenital hydrocephalus, suspected immune-mediated hepatitis, and probable distemper encephalitis have been reported to cause SIADH in dogs. 5-8 SIADH has been reported in a cat after the administration of anesthetic drugs to perform a laparotomy to investigate the etiology of apparent liver disease. 9 Intracranial disease may directly stimulate the supraoptic or paraventricular nuclei to secrete ADH or may alter the osmoreceptors to inappropriately stimulate ADH secretion. Other cerebral causes of SIADH are perception of nausea, pain, and psychologic stress. 10
How does ADH work?
ADH actions are achieved by the promotion of free water resorption by the kidneys , specifically the distal convoluted tubules and collecting ducts. Serum osmolality is monitored by the anterior portion of the hypothalamus. If blood pressure is normal or elevated, ADH secretion is inhibited by pressure receptors in the atria and great veins. A rise in serum osmolality is a more sensitive monitor (1% rise) and typical stimulus for ADH secretion than a decrease in blood pressure (9% decrease). 7,8 SIADH is defined as an excess of ADH in the absence of hypovolemia or hyperosmolality.
What drugs increase ADH secretion?
10,11 Drugs that are known to increase ADH secretion in humans include antidepressants (especially tricyclic antidepressants and monoamine oxidase inhibitors), anticancer drugs (intravenous cyclophosphamide and vinca alkaloids), opioids, and neuroleptics.
What is SIADH in humans?
SIADH is a PNS widely recognized in lung, head, and neck, and other tumors in humans; 1 however, it continues to be essentially unrecognized in veterinary oncology. In addition to PNS-associated SIADH, chemotherapy agents and other drugs (vincristine, cyclophosphamide, cisplatin, thiazides, morphine, and chlorpropamide), pulmonary or CNS infections, and a variety of other conditions can cause SIADH. 59,60 The initial finding in SIADH patients is hyponatremia. In addition to hyponatremia, serum hypo-osmolarity, hypernatruresis, urine hyperosmolarity, and euvolemia with normal renal, thyroid, and adrenal function are noted. 1,59 Though most human SIADH patients are asymptomatic, clinical signs can develop due to hyponatremia, which result in CNS signs such as fatigue, anorexia, confusion, and potentially seizures. The treatment of choice for PNS-associated SIADH is removal of the underlying cause. In addition, water restriction, demeclocycline (ADH antagonist), and hypertonic sodium chloride may be useful in SIADH cases. 61
What is the thirst center?
The thirst center in the hypothalamus monitors plasma osmolality and extracellular fluid volume. If the patient is conscious, psychologically normal, and has a normal thirst center, water intake will subside to compensate for the reduction in plasma osmolality and expanded extracellular fluid volume of SIADH. Patients receiving fluid therapy, under sedation or anesthesia, that are psychologically deranged, or with central nervous system disease affecting the thirst center have an impaired ability to compensate for SIADH.
How does antidiuretic hormone affect the collecting tubules?
Antidiuretic hormone exerts its activity on the collecting tubules by activating adenyl cyclase ; this results in the generation of cyclic adenosine monophosphate (cyclic AMP) and protein kinases, which in turn alter the permeability of the tubules to water ( Rose, 1984 ).
What is the syndrome of excessive vasopressin secretion?
Excess vasopressin secretion results in the syndrome of inappropriate anti-diuretic hormone (SIADH), where the water retention has such a diluting effect on plasma that it results in low plasma sodium levels (hyponatraemia) with a normal plasma volume. The syndrome is called ‘inappropriate ADH secretion’ because the appropriate physiological response would be to reduce AVP (ADH) secretion and increase diuresis. The problem in SIADH is that the AVP levels are inappropriately high.
What is VP release?
VP release is exquisitely sensitive to changes in osmolality of the plasma or ECF. A 1% increase in osmolality increases plasma VP to a level sufficient to elicit detectable antidiuresis. This is the result of an elaborate series of osmoreceptive elements strategically situated to monitor ECF osmolality at different sites in the body. Osmoreceptive elements are located in the mesenteric and hepatic portal vasculature. These receptors monitor osmolality of the blood draining the intestine and provide an early indication of water, electrolyte, and nutrient intake. Osmoreceptors are also located in several regions of the brain that lack a blood–brain barrier (i.e., subfornical organ (SFO) and organum vasculosum of lamina terminalis (OVLT)). These receptors are positioned to monitor plasma osmolality unaffected by transport limitations of the blood–brain barrier. Finally, the magnocellular neurons of the SON and PVN are also osmosensitive, providing a final site for regulation of VP secretion by osmolality.
What are the modulators of AVP release?
Modulators of AVP Release#N#AVP secretion is under the control of selective osmoreceptors that increase AVP concentration when plasma osmolality increases by 1–2%, and baroreceptors which modulate AVP release in response to changes in arterial volume. AVP is also released in response to physiological stress and perhaps psychological stress, although the mechanism is less clear. AVP release may be caused by osmotic-mediated changes in the shape of vasopressinergic neurons, possibly involving Trpv1 channels [116]. Opiates inhibit CRH- and AVP-stimulated ACTH release, and different opiate agonists differentially affect CRH- versus AVP-stimulated release. In humans, morphine blunts CRH-stimulated ACTH release without decreasing AVP or catecholamine levels.
How to treat SIADH?
The treatment of SIADH varies based on the time course. Acute SIADH (<48 h) is generally treated with fluid restriction and, if severe, HTS and/or furosemide. Chronic SIADH (>48 h) is treated with long-term fluid restriction (1200–1800 cc/day), demeclocycline, furosemide, and occasionally phenytoin (which is thought to inhibit ADH release). Care should be taken not to correct hyponatremia too rapidly: there have been reports of profound neurologic deficits following rapid correction of hyponatremia in patients with chronic disease. This phenomena, termed ‘osmotic demyelination syndrome’ can usually be avoided if the rate and degree of sodium repletion is carefully monitored – do not exceed a correction rate of 1.3 mEq/L per h, limit corrective measures if sodium increases >10 mEq/L in 24 h, and stop corrective measures if sodium increases >125 mEq/L.
Where is ADH synthesized?
ADH is synthesized in the supraoptic and paraventricular nuclei of the hypothalamus. After initial synthesis, the precursor hormone is transported down the pituitary stalk into the posterior lobe of the pituitary where ADH undergoes final maturation to active hormone and is stored in vesicles for future release.
How to treat dehydration?
The first principle of treatment is to prevent dehydration by ensuring an adequate water intake. The second is to replace arginine vasopressin with a synthetic analogue called desmopressin. Desmopressin acts like arginine vasopressin and reduces urinary water excretion and increases urine osmolality.
Where are osmoreceptive elements located?
Osmoreceptive elements are located in the mesenteric and hepatic portal vasculature. These receptors monitor osmolality of the blood draining the intestine and provide an early indication of water, electrolyte, and nutrient intake.
What is the substance that is similar to vasopressin?
It has several neurological effects on the brain. A substance that is similar to vasopressin is lysine vasopressin (LVP); it is also known as lypressin which is found in pigs that perform the same function. The synthetic version of LVP is used in humans in ...
What is the name of the hormone that is synthesized by the hypothalamus?
Vincent du Vigneaud synthesized the vasopressin for the first time. It is also known as ADH (Antidiuretic hormone), Arginine Vasopressin (AVP), or argipressin. Vasopressin is a hormone that is produced by the neurons present in the hypothalamus location, it is first synthesized as peptide prohormone, and then it is converted into AVP. The converted AVP is then transported to the axon region of the neurons. From the axon, it is terminated in the posterior pituitary, and in response to the circulation of extracellular fluid hypertonicity. It induces the differentiation of the stem cells to form cardiomyocytes, this promotes the homeostasis of the heart muscle. It is said that the life span of vasopressin is half-life as it can live in between 16 to 35 minutes.
Why is vasopressin used in diabetes?
Vasopressin is used in the treatment of diabetes insipidus, which is caused due to the absence of naturally occurring pituitary hormone in the body.
Where does ADH come from?
The ADH that is measured in the peripheral blood is derived from the secretion of vasopressin from the region of the posterior pituitary gland. Cortisol can inhibit the release of ADH. Let us see how it functions, what are its uses, side effects, and much more about this substance.
Which hormone regulates the tonicity of the fluids situated in the body?
Arginine Vasopressin. Vasopressin can regulate the tonicity of the fluids situated in the body. These are released in the posterior pituitary region in response to the hypertonicity, this makes the kidneys reabsorb the solute-free water and then the tonicity is returned from the nephron via tubules to circulation.
How long does vasopressin live?
It is said that the life span of vasopressin is half-life as it can live in between 16 to 35 minutes. The decrease in the arteriole volume of blood stimulates the secretion of the vasopressin.
Why is urine excreted?
This occurs due to the insertion of water channels in the apical membrane that is found in the collecting tubules and in the collecting duct epithelial cells, due to increased transcription.
What is the consequence of lack of vasopressin?
False. 77. A consequence of lack of vasopressin is excretion of sugar in the urine, diabetes mellitus.
What does the kidney do when water intake is decreased?
False. 69. If water intake (ingestion) is decreased, the kidney can decrease the amount of water excreted in urine by increasing the amount of water reabsorbed at the renal corpuscle.
Which tubule allows for secondary active cotransport of hydrogen ion and counter transport of glucose and amino acids?
C. in the proximal tubule allows for secondary active cotransport of hydrogen ion and counter transport of glucose and amino acids.
Which type of transport involves the movement of substances from the peritubular capillaries into the tubular fluid?
E. Both involves the movement of substances from the peritubular capillaries into the tubular fluid and involves transepithelial transport that may have a combination of active and passive components are correct
Where is the most of the body's water located?
Most of the body's water is located inside cells, whereas most of the body's sodium ion is in the interstitial fluid and plasma. True. 75. The concentration of water in the extracellular fluid is dependent upon the concentration of sodium in the extracellular fluid.
Which pressure in Bowman's space opposes filtration?
A. The hydrostatic pressure in Bowman's space opposes filtration
Does sweat increase ADH?
The loss of 0.5 L of sweat would stimulate a greater increase in ADH secretion than the loss of an equal amount of blood plasma over the same period of time. True. 88. Atrial distension is a stimulus for the secretion of atrial natriuretic factor, which stimulates the reabsorption of sodium by the kidneys.
What happens 2 hours before surgery?
Two hours before major surgery, the patient is stressed, with increased heart rate and blood pressure. These symptoms are the result of. A) decreased levels of epinephrine in the blood. B) decreased activity of sympathetic centers in the hypothalamus.
What happens when venous return is increased?
When venous return is increased, stretch receptors in the atria of the heart are activated. This results in
What is the response of osmoreceptors to plasma osmolarity?
Osmoreceptors fire after they in response to _ plasma osmolarity.
Which gland secretes B?
B) is secreted by the anterior pituitary gland in response to changes in blood osmolarity.
Which is the primary route for ion loss from the body?
The primary route for ion loss from the body is the system .
How does ECF affect thirst?
Differences between the extracellular fluid (ECF) osmolality and the desired set point induce proportional homeostatic responses according to the principle of negative feedback 21, 27 (Fig. 5 a). ECF hyperosmolality stimulates the sensation of thirst 28 to promote water intake and the release of vasopressin 29 that will enhance water reabsorption in the kidney. By contrast, ECF hypo-osmolality suppresses basal vasopressin secretion in rats and humans 30. Thirst and vasopressin release appear thus far as a purely homeostatic response to deviations in intero-sensory stimuli: blood osmolality, pressure or volume. The techniques used in the 1960s and 1970s to describe these intero-sensory stimuli lacked the ability to track thirst neurones of the lamina terminalis and vasopressin neurones projecting to the posterior pituitary in real time in behaving animals and so could not assess extero-sensory information regulating these processes 31 .
What is the role of Vasopressin in the kidney?
Vasopressin (AVP) plays a major role in the regulation of water and sodium homeostasis by its antidiuretic action on the kidney, mediated by V2 receptors. AVP secretion is stimulated by a rise in plasma osmolality, a decline in blood volume or stress. V1a receptors are expressed in vascular smooth muscle cells, ...
What is the function of vasopressin?
The primary function of vasopressin is to maintain body fluid balance by keeping plasma osmolality within narrow limits and allowing the kidneys to adapt water excretion to the body's needs, in conjunction with thirst. No other hormone can replace vasopressin: the lack of vasopressin results in diabetes insipidus (10–15 L urine·day −1 ). Vasopressin has a very short biological half-life (about 3 min), and it is cleared mostly by filtration in the kidneys. Accordingly, its effects are very prompt and promptly reversible.
What is thirst regulated by?
It is now understood that thirst and arginine vasopressin (AVP) release are regulated not only by the classical homeostatic, intero-sensory plasma osmolality negative feedback, but also by novel, extero-sensory, anticipatory signals .
How does V1AR affect V2R?
Luminal V1aR, exposed to urinary vasopressin, induces the production of prostaglandins by CD cells that , indirectly , attenuate the adenylate cyclase response to V2R stimulation. Secondly, V1aR are also abundantly expressed in interstitial cells of the medulla where they stimula te the production of prostaglandins that vaso dilate the medulla ry vasculature. This opposes the possible vasoconstrictive effect of V1aR in the descending vasa recta and induces an increase in medullary blood flow that compromises the osmotic gradient of the medulla. Thus, there is a subtle balance between V2R and V1aR effects in the kidney, as demonstrated in rats 8, 11 .
Why does eating increase thirst?
Eating increases the need for water for two reasons: (i) there is a need to replace the fluid utilized for swallowing (saliva) and digestion (water diverted from the circulation into the gastrointestinal tract) and (ii) to counteract the increase in blood osmolality caused by the absorption of salts and other osmoles from food. As described recently in a review on thirst 32, anticipatory signals about ongoing food ingestion are communicated to the lamina terminalis by multiple mechanisms. (i) Somatosensory signals from the oral cavity that report on food swallowing or its effects on the saliva. (ii) Several hormones associated with eating and satiety have been proposed to modulate thirst neurones and vasopressin release, including amylin, cholecystokinin, ghrelin, histamines, insulin and leptin. Some of these hormones might be increased in patients with diabetes mellitus and may explain their high vasopressin plasma concentration 39 .
Where is V1A expressed?
V1a receptors are expressed in vascular smooth muscle cells , but the role of vasopressin in blood pressure regulation is still a matter of debate. AVP may also play a role in some metabolic pathways, including gluconeogenesis, through its action on V1a receptors expressed in the liver.
