
Both alpha (α) and beta (β) glucose are optically active organic compounds. Alpha (α)-D glucose is an isomer of dextrose (D) glucose with the identification of having hydroxyl (-OH) groups on 1 and 4 positions on the same sides of the plane.
How many isomers of glucose are there?
Glucose has two isomers (types), the alpha and the beta. When it comes to alpha vs. beta glucose, what are the differences and similarities between the two?
What is the difference between alpha and beta glucose isomers?
Alpha and beta are both glucose isomers, but they differ only in the position of their -OH (hydroxyl) and -H (hydrogen) groups on carbon 1. Beta glucose has its -OH group above the ring. On the other hand, Alpha glucose has its -OH attached below the ring.
What are the anomers of glucose 6 phosphate?
There are two anomers of glucose 6 phosphate, the alpha anomer and the beta anomer. Specifically, beta-D-Glucose 6-phosphate is glucose sugar phosphorylated on carbon 6. It is a very common metabolite in cells as the vast majority of glucose entering a cell will become phosphorylated in this way.
What is the difference between alpha-D and beta-D glucose?
Digestion of alpha-D glucose vs beta-D glucose differs, though, as starch is easily digested, resulting in alpha glucose chains, while cellulose is not, resulting in beta glucose. The small intestine’s villi will absorb alpha glucose and convert it to its inactive form – glycogen.
See more
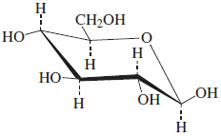
Are alpha D-glucose and beta D-glucose enantiomers?
Now, the $\alpha - D - $ glucose and $\beta - D - $glucose are non-superimposable as they do differ in the position of the hydroxyl group at carbon 1 but they are not the exact mirror image of each other. Thus, they are not enantiomers.
Are alpha and beta glucose optical isomers?
beta-D-glucose. The alpha and beta isomers differ by the position of an hydroxy group on carbon number 1. In the alpha the hydroxy group is below the plane of the molecule where as in the beta isomer the hydroxy group is above the plane of the molecule.
Are alpha glucose and beta glucose diastereomers?
α−D−(+)-glucose and β−D−(+)-glucose are those diastereomers that differ in configuration at C- 1 atom.
What is the relationship between alpha and beta D-glucose?
The D-glucose can exist in two forms alpha-D-glucose and beta-D-glucose. They differ only in the direction that -H and -OH groups point on carbon 1 (See the jmol images below). When alpha-glucose molecules are joined chemically to form a polymer starch is formed.
How do you identify an optical isomer?
The mirror images method uses a mirror image of the molecule to determined whether optical isomers exist or not. If the mirror image can be rotated in such a way that it looks identical to the original molecule, then the molecule is said to be superimposable and has no optical isomers.
What are the optical isomers of glucose?
Note: The 16 optical isomers of glucose are allose, altrose, galactose, glucose, gulose, idose, mannose, and tallose.
Are D-glucose and L-glucose enantiomers or diastereomers?
There are two enantiomers of glucose, called D-glucose and L-glucose. The D-enantiomer is the common sugar that our bodies use for energy. It has n = 4 stereocenters, so therefore there are 2n = 24 = 16 possible stereoisomers (including D-glucose itself).
What are diastereomers with examples?
Diastereomers may often include compounds which are ring structures. Imagine, for example, two compounds with a six-membered ring, each with two substituents, a chlorine atom and an ethyl group. They are also not mirror images of each other, just like our previous example, which defines them as diastereomers.
What is the difference between alpha D-glucose and beta D-glucose?
Solution : The isomer having the hydroxyl group (-OH) on the right is called `alpha-`D-glucose and one having the hydroxyl group (-OH) on the left is called `beta-`D-glucose. Such pairs of optical isomers which differ in the configuration only around `C_(1)` atom are called anomers.
What are alpha and beta stereoisomers?
3:306:56Alpha, beta isomers - YouTubeYouTubeStart of suggested clipEnd of suggested clipThey are isomers of one another we talked about isomers back with halo alkanes. Seen same molecularMoreThey are isomers of one another we talked about isomers back with halo alkanes. Seen same molecular formula. But different structure. Okay now here they have almost exactly the same structure.
How alpha and beta anomers of D-glucose are formed?
Each of these two forms can be synthesized and isolated as pure compounds. The alpha (α) anomer of D-glucose has a specific rotation of +112 degrees in water. The beta (β) anomer of D-glucose has a specific rotation of +19 degrees. (18.7 actually, but rounding up to 19).
What is the difference between alpha and beta glucose quizlet?
The difference between alpha glucose and beta glucose is in their structures. In alpha glucose, the hydroxyl group is attached facing down and away from the main structure, while in the beta glucose, the hydroxyl group is attached above the ring and on the first carbon.
What is the difference between α glucose and β glucose?
The main difference between alpha and beta glucose is that the –OH group attached to the first carbon atom in alpha glucose is located on the same side as the –CH2OH group whereas the –OH group attached to the first carbon atom of in beta glucose is located on the opposite side from the –CH2OH group.
Are alpha and beta stereoisomers?
α-D-glucose and β-D-glucose are stereoisomers - they differ in the 3-dimensional configuration of atoms/groups at one or more positions. Note that the structures are almost identical, except that in the α form, the OH group on the far right is down, and, in the β form, the OH group on the far right is up.
How do the α and β forms of glucose differ?
How do the α and β forms of glucose differ? Their linear structures differ in the location of a hydroxyl group. Their ring structures differ in the location of a hydroxyl group. The α form can be involved in 1,4- and 1,6-glycosidic linkages; the β form can participate only in 1,4 linkages.
What is the difference between the alpha and beta anomers of glucose?
The key difference between alpha and beta anomers is that in alpha anomer the hydroxyl group at the anomeric carbon is cis to the exocyclic oxygen at the anomeric centre, whereas in beta anomer the hydroxyl group is trans to the exocyclic oxygen. An anomer is a geometric variation among carbohydrates.
How many isomers are there in D-glucose?
In our recent post on ring-chain tautomerism , we said that there are two isomers of D-glucose in its 6-membered ring (pyranose) form. These two diastereomers which, to make matters more confusing, are called anomers in the context of sugar chemistry differ in the orientation of the hydroxyl group on C-1. (Note that C-1 is a hemiacetal . ) In the alpha () anomer, the OH group on C-1 is on theopposite side of the ring as the chain on C-5. In the beta () anomer, the OH group on C-1 is on the sameside of the ring as the C-5 substituent. Each of these two forms can be synthesized and isolated as pure compounds. The alpha () anomer of D-glucose has a specific rotation of +112 degrees in water. The beta () anomer of D-glucose has a specific rotation of +19 degrees. (18.7 actually, but rounding up to 19). Heres the interesting thing.When either anomer is dissolved in water, the value of the specific rotation changes over time, eventually reaching the same value of +52.5. The specific rotation of -D-glucopyranose decreases from +112 to +52.5. The specific rotation of-D-glucopyranose increases from +19 to +52.5. This behaviour is calledmutarotation (literally, change in rotation). Hold on. Isnt specific rotation of a molecule supposed to remain the same? Yes if it is indeed the same molecule! And therein lies the answer to the puzzle. For when the solutions whose specific rotations have changed to +52.5 are analyzed, they are found to no longer consist of 100% alpha () or 100% beta ()anomers, but instead a ratio of alpha () (36%) and beta () (64% ) isomers. Wait. What happened here? How did the alpha convert to the beta, and vice-versa? You may recall how we said in the last post on ring-chain tautomerism that the cyclic hemiacetal forms of sugars are in equilibrium with the str Continue reading >>
What is the D form of glucose?
This article is about the naturally occurring D-form of glucose. For the L-form, see L-Glucose. Glucose is a simple sugar with the molecular formula C6H12O6, which means that it is a molecule that is made of six carbon atoms, twelve hydrogen atoms, and six oxygen atoms. Glucose circulates in the blood of animals as blood sugar. It is made during photosynthesis from water and carbon dioxide, using energy from sunlight. It is the most important source of energy for cellular respiration. Glucose is stored as a polymer, in plants as starch and in animals as glycogen. With six carbon atoms, it is classed as a hexose, a subcategory of the monosaccharides. D-Glucose is one of the sixteen aldohexose stereoisomers. The D-isomer, D-glucose, also known as dextrose, occurs widely in nature, but the L-isomer, L- glucose, does not. Glucose can be obtained by hydrolysis of carbohydrates such as milk sugar (lactose), cane sugar (sucrose), maltose, cellulose, glycogen, etc. It is commonly commercially manufactured from cornstarch by hydrolysis via pressurized steaming at controlled pH in a jet followed by further enzymatic depolymerization. [3] In 1747, Andreas Marggraf was the first to isolate glucose. [4] Glucose is on the World Health Organization's List of Essential Medicines, the most important medications needed in a basic health system. [5] The name glucose derives through the French from the Greek γλυκός, which means "sweet," in reference to must, the sweet, first press of grapes in the making of wine. [6] [7] The suffix "-ose" is a chemical classifier, denoting a carbohydrate. Function in biology Glucose is the most widely used aldohexose in living organisms. One possible explanation for this is that glucose has a lower tendency than other aldohexoses to react nonspecific Continue reading >>
What happens when glucose is dissolved in water?
When dissolved in water, these glucose tablets will yield a mixture of alpha and beta glucose.Photo Credit: YakubovAlim/iStock/Getty Images Based in San Diego, John Brennan has been writing about science and the environment since 2006. His articles have appeared in "Plenty," "San Diego Reader," "Santa Barbara Independent" and "East Bay Monthly." Brennan holds a Bachelor of Science in biology from the University of California, San Diego. Glucose is one of the "simple sugars" -- an ironic name, because the chemistry of these compounds is rather complex. The naming system for sugars reflects this complexity. Chemists use prefixes like alpha and beta to denote different versions of glucose and other sugar molecules. To the uninitiated, these prefixes may seem mysterious, but once you understand sugar structure, their nature and purpose will become clearer. Each molecule of glucose has a carbon backbone with -OH groups and hydrogen atoms attached to it. At the top of the chain, an oxygen atom is double-bonded to a carbon atom; collectively, these two atoms are called a carbonyl group. The carbon backbone of the glucose molecule can coil up so that an -OH group near the bottom end of the chain attacks the carbonyl carbon and the glucose molecule forms a ring. This ring-shaped structure is the cyclic form of glucose, while the straight chain structure is the linear form. In solution, the cyclic form is by far the more common. Glucose can form either five-membered or six-membered rings. The six-membered ring is much more common, and in solution the vast majority of glucose molecules are found to have six-membered rings. Since linear and cyclic forms can inter-convert, however, no glucose molecule is ever fixed in the six-membered ring form; it can go back and forth. It does sp Continue reading >>
Which anomer is more stable?
Anomers (contd)The anomer, which occurs in greater percentage, is more stable. The pyranose ring is not planar, but rather tends to be in either of two chair conformations (Fig 7-8). The anomer should be more stable because its C-1 OH group is equatorial. In contrast, the C-1 OH group of the anomer is axial. In general, substituents in the equatorial positions are less hindered sterically by neighboring substituents. Monosaccharides: Reducing AgentsMonosaccharides can be oxidized by relatively a mild reducing agent (Cu 2+ )as shown in Fig. 7-10. The carbonyl group is oxidized to a carboxyl group. Glucose and others that reduce these metal ions are called reducing sugars.Fehlings reaction-measures amount of oxidizing agent reduced by the sugar. Can estimate the conc. of glucose in blood and urine (diabetes test). Currently, the enzyme, glucose oxidase, is used as a more sensitive measure for blood glucose levels. This is the end of the preview. Sign up to access the rest of the document. Continue reading >>
Why is glucose called a simple sugar?
Glucose is one of the "simple sugars" -- an ironic name, because the chemistry of these compounds is rather complex. The naming system for sugars reflects this complexity. Chemists use prefixes like alpha and beta to denote different versions of glucose and other sugar molecules.
Is postprandial blood glucose a predictor of cardiovascular events?
Postprandial Blood Glucose Is a Stronger Predictor of Cardiovascular Events Than Fasting Blood Glucose in Type 2 Diabetes Mellitus, Particularly in Women: Lessons from the San Luigi Gonzaga Diabetes Study
Where does beta glucose come from?
Alpha glucose has the -OH attached below the ring, while beta glucose has its -OH group above the ring. Alpha and beta glucose come from the carbohydrate meals you eat, such as starch, rice, potatoes, and pasta. These meals are digested to simple sugars in your small intestines and metabolized and stored in your liver.
Why is beta glucose more stable than other glucose?
Beta glucose is more stable because the body cannot easily take it apart. This stability is due to the solidly packed binding of its molecules.
What is the difference between starch and beta glucose?
On the other hand, beta glucose chains are composed of cellulose, and the body cannot break it down easily.
Why is alpha glucose important?
Alpha glucose has more vital functions in the body than beta glucose. It’s because the body cannot fully metabolize beta glucose for utilization.
What are the components of a glucose chain?
Composition. Alpha glucose chains are composed of starch, while beta glucose chains are made of cellulose. Bread, pasta, potatoes, and similar foods come from starch. Typically, the tastier parts of plants are made of starch, while the hard parts of plants, such as the stem, are made of cellulose. 3.
How many atoms are in glucose?
The most predominant simple sugar is glucose (C 6 H 12 O 6 ), meaning it has 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms. Glucose turns into glycogen and is stored in the liver when you don’t need energy, and the body converts it back to glucose plus ATP when you need energy.
Which type of glucose has the same stereogenic center?
Alpha and beta glucose have the same stereogenic center – carbon 1. Both types of glucose attach the -OH to carbon 1 – only in different positions.
How many isomers are there in D-glucose?
In our recent post on ring-chain tautomerism , we said that there are two isomers of D-glucose in its 6-membered ring (pyranose) form. These two diastereomers which, to make matters more confusing, are called anomers in the context of sugar chemistry differ in the orientation of the hydroxyl group on C-1. (Note that C-1 is a hemiacetal . ) In the alpha () anomer, the OH group on C-1 is on theopposite side of the ring as the chain on C-5. In the beta () anomer, the OH group on C-1 is on the sameside of the ring as the C-5 substituent. Each of these two forms can be synthesized and isolated as pure compounds. The alpha () anomer of D-glucose has a specific rotation of +112 degrees in water. The beta () anomer of D-glucose has a specific rotation of +19 degrees. (18.7 actually, but rounding up to 19). Heres the interesting thing.When either anomer is dissolved in water, the value of the specific rotation changes over time, eventually reaching the same value of +52.5. The specific rotation of -D-glucopyranose decreases from +112 to +52.5. The specific rotation of-D-glucopyranose increases from +19 to +52.5. This behaviour is calledmutarotation (literally, change in rotation). Hold on. Isnt specific rotation of a molecule supposed to remain the same? Yes if it is indeed the same molecule! And therein lies the answer to the puzzle. For when the solutions whose specific rotations have changed to +52.5 are analyzed, they are found to no longer consist of 100% alpha () or 100% beta ()anomers, but instead a ratio of alpha () (36%) and beta () (64% ) isomers. Wait. What happened here? How did the alpha convert to the beta, and vice-versa? You may recall how we said in the last post on ring-chain tautomerism that the cyclic hemiacetal forms of sugars are in equilibrium with the str Continue reading >>
What is the anomeric centre of a sugar?
If you would like to suggest an addition or correction, please contact the page's Responsible Curator directly by e-mail, or using this form . The anomeric centre of a sugar is a stereocentre created from the intramolecular formation of an acetal (or ketal) of a sugar hydroxyl group and an aldehyde (or ketone) group. The two stereoisomers formed from the two possible stereochemistries at the anomeric centre are called anomers. They are diastereoisomers of one another. The configuration at the anomeric centre (that derived from the carbonyl carbon) is denoted alpha- (-) or beta- (-) by reference to the stereocentre that determines the absolute configuration. In a Fischer projection, if the substituent off the anomeric centre is on the same side as the oxygen of the configurational (D- or L-) carbon, then it is the --anomer. If it is directed in the opposite direction it is the -anomer. Example 1. Fischer projections and Haworth conformational projections of L-arabinose. Example 2. Fischer projections and Haworth conformational projections of D-fructose. In the case of D-hexopyranoses drawn in the 'usual' Haworth projection, the -D-anomer is the isomer with the anomeric substituent on the opposite face to the C5 (hydroxymethyl) substitutent, ie directed down; the -D-anomer is that with the anomeric substituent being on the same face as the C5 hydroxymethyl substitutent, ie directed up. For L-hexoses the -L-anomer has the anomeric group pointing up; the -L-anomer has this group pointing down. Example 3. Fischer projections and Haworth conformational projections of D-glucose. Continue reading >>
What happens when glucose is dissolved in water?
When dissolved in water, these glucose tablets will yield a mixture of alpha and beta glucose.Photo Credit: YakubovAlim/iStock/Getty Images Based in San Diego, John Brennan has been writing about science and the environment since 2006. His articles have appeared in "Plenty," "San Diego Reader," "Santa Barbara Independent" and "East Bay Monthly." Brennan holds a Bachelor of Science in biology from the University of California, San Diego. Glucose is one of the "simple sugars" -- an ironic name, because the chemistry of these compounds is rather complex. The naming system for sugars reflects this complexity. Chemists use prefixes like alpha and beta to denote different versions of glucose and other sugar molecules. To the uninitiated, these prefixes may seem mysterious, but once you understand sugar structure, their nature and purpose will become clearer. Each molecule of glucose has a carbon backbone with -OH groups and hydrogen atoms attached to it. At the top of the chain, an oxygen atom is double-bonded to a carbon atom; collectively, these two atoms are called a carbonyl group. The carbon backbone of the glucose molecule can coil up so that an -OH group near the bottom end of the chain attacks the carbonyl carbon and the glucose molecule forms a ring. This ring-shaped structure is the cyclic form of glucose, while the straight chain structure is the linear form. In solution, the cyclic form is by far the more common. Glucose can form either five-membered or six-membered rings. The six-membered ring is much more common, and in solution the vast majority of glucose molecules are found to have six-membered rings. Since linear and cyclic forms can inter-convert, however, no glucose molecule is ever fixed in the six-membered ring form; it can go back and forth. It does sp Continue reading >>
Is Alpha (d+) Glucose Same As Beta (d-) Glucose?
Is alpha (D+) glucose same as beta (D-) glucose? No. In the name D (+) Glucose, D represents the orientation of the hydroxyl group at the chiral carbon that is farthest from the highest oxidised carbon (Aldehyde group in this case) with respect to glyceraldehyde. D says that the hydroxyl group is on the right side (In fischer projection). L says the opposite. Where as (+) and (-) represent the direction of rotation of plane polarised light {Optical rotation} (Determined experimentally) by the solution as a whole. When a water molecule adds to the glucose molecule, the aldehyde group turns into hemiacetal (Ring is formed between 1st and 5th carbon), making the first carbon (previously aldehyde) chiral. Alpha and Beta represent the orientation of hydroxide group (R and S) at that new chiral carbon (Anomeric carbon). Hence those forms are called anomers. Pure Alpha glucose has a positive optical rotation and the Beta form has the opposite rotation (but of different magnitude). The compounds which you have mentioned in the question are pure alpha and pure beta forms of glucose. If you get some random D glucose from somewhere and you some how determined that it has a positive optical rotation,it doesnt mean that it is alpha D glucose. It may also contain beta D glucose each cancelling the effect of other and ultimately resulting a positive optical rotation (Overall dominated by alpha D glucose). Continue reading >>
Difference Between Alpha(α) and Beta(Β) Glucose
- Similarities among alpha (α) and beta (β) glucose
1. Both alpha(α) and beta(β) glucose structures are simple sugar monomers. 2. They can be crystallized from their aqueous solutions, although, alpha(α) glucose cannot be crystallized as α-glucopyranose hydrate. 3. They both have the same number of chiral carbon centers i.e. 4. 4. Bo…
Alpha(Α)-D Glucose
- Alpha(α)-D glucose is an isomer of dextrose(D) glucose with the identification of having hydroxyl (-OH) groups on 1 and 4 positions on the same sides of the plane. It can also be referred to as an orientation in which the hydroxyl group of 1 position is on the opposite side to that of (-CH2OH). This isomer of glucose is less stable than the beta(β) glucose because of the steric hindrance o…
Key Differences Between α and β Glucose
- Alpha and Beta glucose are both aldose structures, and neither of them is from the ketose family. Aldose and Ketoseterms are really important in knowing about sugar molecules especially, glucose and fructose.
Concepts Berg
- Chiral centers in Alpha and Beta Glucose Alpha and beta glucose, both contain similar chiral centers, although cyclic and open chain forms have different numbers of chiral centers. Cyclic glucose (α and β) structures have 5 chiral centers while open chains have 4. Read more at, Chiral vs Achiral vs Meso compounds What is beta glucose? Beta glucose is an isomer of dextrose(D) …