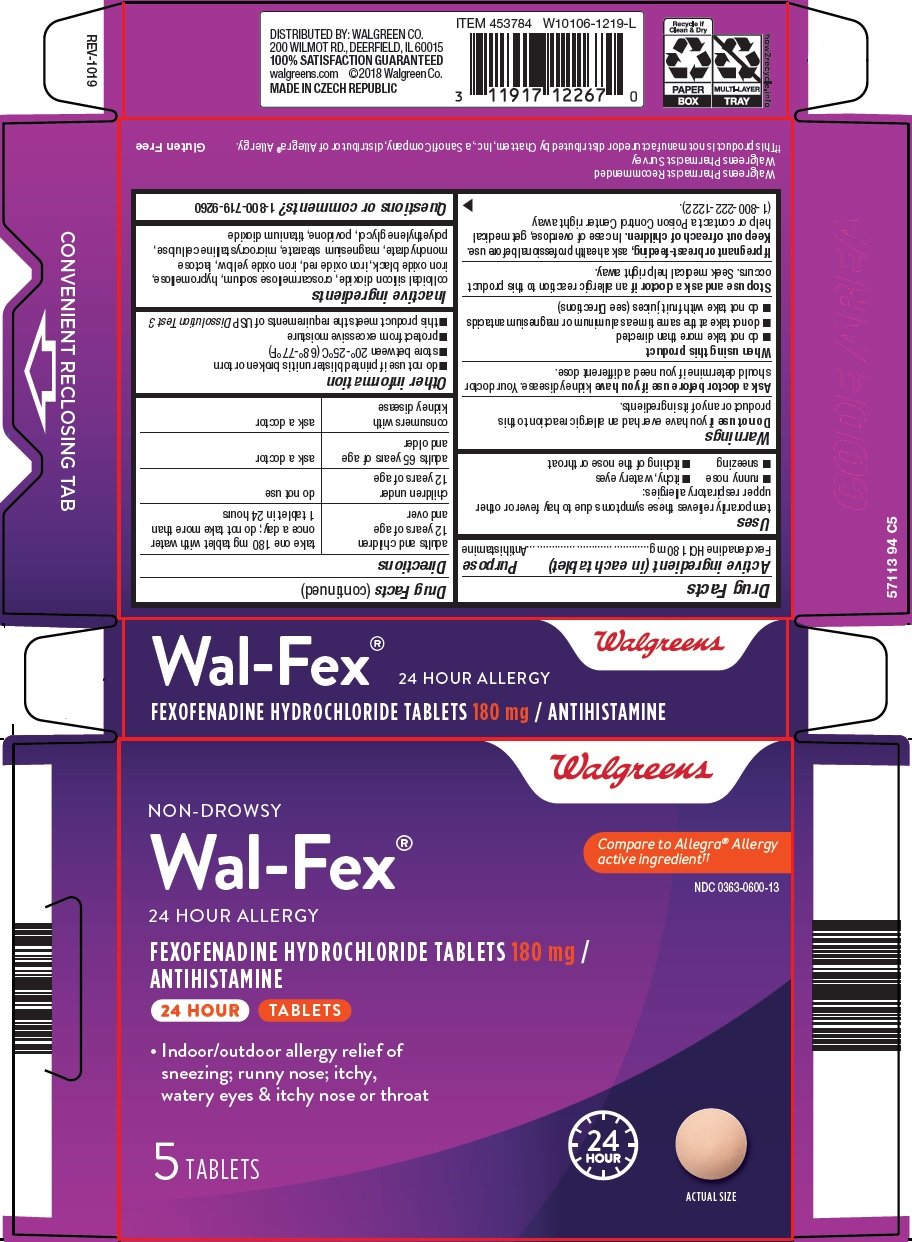
Precautions
This is a flu-like illness with symptoms of metallic taste in the mouth, headache, fever and chills, aches, chest tightness and cough. The symptoms may be delayed for several hours after exposure and usually last for a day or two. IDENTIFICATION Magnesium Oxideis a white powder. When finely divided
Is magnesium oxide good or bad?
coma. dizziness or fainting. drowsiness (severe) increased or decreased urination. slow heartbeat. troubled breathing. Some side effects may occur that usually do not need medical attention. These side effects may go away during treatment as your body adjusts to the medicine.
What does magnesium oxide taste like?
What does magnesium oxide mean? Definition of magnesium oxide . : a white highly infusible compound MgO used especially in refractories, cements, insulation, and fertilizers, in rubber manufacture, and in medicine as an antacid and mild laxative.
What are the adverse effects of magnesium oxide?
What does magnesium oxide mean?
How much improvement does magnesium oxide give?
How does it compare with other forms of magnesium?
What is magnesium used for?
Which magnesium supplement has the lowest bioavailability?
Which is more effective for magnesium absorption?
Which magnesium is better for migraines?
Does magnesium oxide help with headaches?
See 4 more
About this website

Is magnesium oxide a base oxide?
Magnesium oxide is another simple basic oxide, which also contains oxide ions. However, it is not as strongly basic as sodium oxide because the oxide ions are not as weakly-bound.
What type of oxide is MgO?
Magnesium oxide (MgO). An inorganic compound that occurs in nature as the mineral periclase. In aqueous media combines quickly with water to form magnesium hydroxide. It is used as an antacid and mild laxative and has many nonmedicinal uses.
Is magnesium oxide acidic or basic?
Hence, magnesium oxide is basic in nature.
Is magnesium oxide an ionic oxide?
Magnesium Oxide is an ionic compound. In this magnesium is metal and oxygen is a nonmetal. Metal and nonmetal bond together to form an ionic compound.
Why is MgO a basic oxide?
Magnesium is a metal. Metals react with oxygen to form metallic oxides. Hence, Magnesium oxides are basic in nature. On the other hand, nonmetals react with oxygen to form nonmetallic oxides.
What are the 4 types of oxide?
Classification Of OxidesAcidic oxides.Basic oxides.Amphoteric oxides.Neutral oxides.
Is magnesium oxide amphoteric oxide?
Magnesium oxide is an amphoteric oxide.
Is Lithium oxide an acidic oxide or a basic oxide?
Yes, Lithium oxide is basic as it dissolves in pure water, the resulting solution has a pH value greater than 7.0. It forms Lithium hydroxide when added to water, which is alkaline.
Is magnesium oxide a strong base?
Magnesium oxide is again a simple basic oxide, because it also contains oxide ions. However, it isn't as strongly basic as sodium oxide because the oxide ions aren't so free. In the sodium oxide case, the solid is held together by attractions between 1+ and 2- ions.
Is magnesium oxide covalent or ionic?
In there is a large difference in electronegativity between magnesium and oxygen. So they form an ionic bond in place of covalent bonds.
Is MgO ionic covalent or metallic?
MgO is an ionic compound. An ionic bond is formed by the transfer of electrons from a metal (Mg) to a non-metal (O). Mg loses two valence electrons to form Mg2+, which are gained by O to form O2-. Thus, an electrostatic interaction arises between two oppositely charged ions, and an ionic bond is formed.
What is an ionic oxide?
The alkali metals and the alkaline earth metals, as well as the transition metals and the posttransition metals (in their lower oxidation states), form ionic oxides—i.e., compounds that contain the O2− anion. Metals with high oxidation states form oxides whose bonds have a more covalent nature.
Does magnesium oxide increase pH?
Feeding of heavy magnesium oxide or magnesium carbonate increased rumen fluid pH as compared to the control diet.
What is the nature of magnesium oxide?
Magnesium oxide is basic in nature as it reacts with water to form magnesium hydroxide which is a base.
Before Taking This Medicine
You should not use magnesium oxide if you are allergic to it.Ask a doctor or pharmacist if it is safe for you to use this medicine if you have othe...
How Should I Take Magnesium Oxide?
Use exactly as directed on the label, or as prescribed by your doctor. Do not use in larger or smaller amounts or for longer than recommended.Take...
What Happens If I Miss A Dose?
Since magnesium oxide is sometimes used when needed, you may not be on a dosing schedule. If you are on a schedule, take the missed dose as soon as...
What Happens If I Overdose?
Seek emergency medical attention or call the Poison Help line at 1-800-222-1222.Overdose symptoms may include nausea, vomiting, weakness, breathing...
What Should I Avoid While Taking Magnesium Oxide?
Magnesium oxide can make it harder for your body to absorb other medicines you take by mouth. Avoid taking other medicines within 2 hours before or...
Magnesium Oxide Side Effects
Get emergency medical help if you have signs of an allergic reaction: hives; difficult breathing; swelling of your face, lips, tongue, or throat.St...
What Other Drugs Will Affect Magnesium Oxide?
Ask a doctor or pharmacist if it is safe for you to take magnesium oxide if you are also using any of the following drugs: 1. an antibiotic; 2. a d...
Pros and cons of taking a magnesium supplement - Mayo Clinic
Many Americans don't get enough magnesium in their diets. Before you reach for a supplement, though, you should know that just a few servings of magnesium-rich foods a day can meet your need for this important nutrient.
Health Benefits of Magnesium Oxide - WebMD
Continued Health Risks. Magnesium oxide is widely used and generally recognized as safe, but it can come with some uncomfortable side effects, such as:
Magnesium oxide Side Effects: Common, Severe, Long Term - Drugs.com
Drugs.com provides accurate and independent information on more than 24,000 prescription drugs, over-the-counter medicines and natural products. This material is provided for educational purposes only and is not intended for medical advice, diagnosis or treatment. Data sources include IBM Watson Micromedex (updated 1 Nov 2022), Cerner Multum™ (updated 25 Oct 2022), ASHP (updated 12 Oct 2022 ...
Magnesium oxide Uses, Side Effects & Warnings - Drugs.com
Generic name: magnesium oxide [ mag-NEE-see-um-OCK-side ] Brand names: MagGel, Phillips' Cramp-free, Uro-Mag, Mag-200, Mag-Ox 400, UroMag Dosage forms: oral capsule (140 mg; 600 mg); oral tablet (250 mg; 400 mg; 420 mg; 500 mg; base 500 mg) Drug class: Minerals and electrolytes Medically reviewed by Drugs.com on Apr 18, 2022. Written by Cerner Multum.
What is magnesium oxide?
Magnesium oxide (MgO). An inorganic compound that occurs in nature as the mineral periclase. In aqueous media combines quickly with water to form magnesium hydroxide. It is used as an antacid and mild laxative and has many nonmedicinal uses.
Where does magnesium oxide occur?
Magnesium oxide occurs in nature as the mineral periclase (1).
What is the concentration of magnesium in sediment?
SEDIMENT: Sediment from three Antarctic lakes (collected in 1993/94) contained between 3,350 to 5,240 mg/kg dry wt magnesium (1). The mean concentration of magnesium (as MgO) in surficial sediment from Lake Ontario ranged from 2.79 to 2.85% dry weight (2). In Po river (Italy) suspended solids collected in 1989, the concentration of magnesium was 1.8% (3).
What are magnesium compounds used for?
About 69% of the magnesium compounds used in the United States were used for refractories (e.g., olivine) (1). The remaining 31% of magnesium compounds were used in agriculture as fertilizer or animal feed (e.g, magnesium oxide, magnesium sulfate ), as chemical intermediates (e.g., magnesium chloride, magnesium hydroxide, magnesium carbonate, magnesium oxide) , construction materials (e.g., magnesium oxide), environmental (e.g., magnesium oxide, magnesium hydroxide ), and industrial applications (e.g., magnesium oxide) (1,2). Other uses include road dust and ice control (e.g., magnesium chloride ), pulp and paper applications (e.g., magnesium sulfate ), pharmaceuticals (e.g., magnesium sulfate, magnesium carbonate, magnesium oxide), and cosmetics (e.g., magnesium carbonate ) (1,2).
How many people are exposed to magnesium oxide?
According to the 2006 TSCA Inventory Update Reporting data, the number of persons reasonably likely to be exposed in the industrial manufacturing, processing, and use of magnesium oxide is 1000 or greater; the data may be greatly underestimated (1).
Can magnesium cause fever?
Irritates the eyes and respiratory tract. Magnesium in the form of nascent magnesium oxide can cause metal fume fever with cough, chest pain, and flu like fever, if inhaled in sufficient quantity. The symptoms of metal fume fever may be delayed for 4-12 hr following exposure.
Can magnesium containing antacids cause diarrhea?
Magnesium -containing antacids commonly cause a laxative effect and frequent administration of these antacids alone often cannot be tolerated; repeated doses cause diarrhea which may cause fluid and electrolyte imbalances. ... / Magnesium containing antacids/
What is magnesium oxide?
Magnesium oxide was historically known as magnesia alba (literally, the white mineral from Magnesia – other sources give magnesia alba as MgCO 3 ), to differentiate it from magnesia negra, a black mineral containing what is now known as manganese .
How is magnesium oxide made?
Magnesium oxide is produced by the calcination of magnesium carbonate or magnesium hydroxide. The latter is obtained by the treatment of magnesium chloride MgCl#N#2 solutions, typically seawater, with limewater or milk of lime.
What is MgO used for?
It has two useful attributes: high thermal conductivity and low electrical conductivity. Filling the spiral Calrod range top heating elements on kitchen electric stoves is a major use. "By far the largest consumer of magnesia worldwide is the refractory industry, which consumed about 56 % of the magnesia in the United States in 2004, the remaining 44 % being used in agricultural, chemical, construction, environmental, and other industrial applications." MgO is used as a basic refractory material for crucibles .
What is MgO cement?
MgO is one of the components in Portland cement in dry process plants .
Why is MgO important?
MgO has an important place as a commercial plant fertilizer and as animal feed.
What is magnesium oxide wallboard?
It is a principal fireproofing ingredient in construction materials. As a construction material, magnesium oxide wallboards have several attractive characteristics: fire resistance, termite resistance, moisture resistance, mold and mildew resistance, and strength.
What is the compound of magnesium?
Chemical compound. Magnesium oxide ( Mg O ), or magnesia, is a white hygroscopic solid mineral that occurs naturally as periclase and is a source of magnesium (see also oxide ). It has an empirical formula of Mg O and consists of a lattice of Mg 2+ ions and O 2− ions held together by ionic bonding.
What is magnesium oxide?
Magnesium is a naturally occurring mineral. Magnesium is important for many systems in the body especially the muscles and nerves.
What other drugs will affect magnesium oxide?
Ask a doctor or pharmacist if it is safe for you to take magnesium oxide if you are also using any of the following drugs:
What should I avoid while taking magnesium oxide?
Magnesium oxide can make it harder for your body to absorb other medicines you take by mouth. Avoid taking other medicines within 2 hours before or 2 hours after you take magnesium oxide. You may need to wait 4 hours to take your other medicines after taking magnesium oxide. Ask your doctor how to best schedule your medications.
Can you take magnesium oxide while pregnant?
Also make sure your doctor knows if you are pregnant or breast-feeding. In some cases, you may not be able to take magnesium oxide, or you may need a dose adjustment or special precautions.
Can magnesium oxide cause bowel movement?
Stop using magnesium oxide and call your doctor at once if you have: no bowel movement after using magnesium oxide as a laxative. Common side effects may include: upset stomach. This is not a complete list of side effects and others may occur. Call your doctor for medical advice about side effects.
Can you use magnesium oxide if you are allergic to it?
You should not use magnesium oxide if you are allergic to it. Ask a doctor or pharmacist if it is safe for you to use magnesium oxide if you have other medical conditions, especially: a sudden change in bowel habits for 2 weeks or longer. It is not known whether magnesium oxide will harm an unborn baby.
Can magnesium oxide harm a baby?
It is not known whether magnesium oxide will harm an unborn baby. Ask a doctor before using this medicine if you are pregnant. It is not known whether magnesium oxide passes into breast milk or if it could affect a nursing baby. Ask a doctor before using this medicine if you are breast-feeding.
What Is Magnesium Oxide Used For?
Magnesium oxide offers some impressive health benefits and can help to relieve the symptoms of a variety of conditions.
How is magnesium oxide obtained?
Magnesium oxide is acquired by burning magnesium with pure oxygen , but this method is quite costly. The cheaper method is to expose oxygen to magnesium salts using underground deposits, seawater, or salt beds. With this method, the source of the magnesium salt determines the quality of the magnesium oxide that is produced.
How to get magnesium?
The best way to get magnesium is through diet and eating a variety of magnesium-rich foods, such as salmon and mackerel, quinoa, black beans, pumpkin seeds, and dark chocolate. 1 Supplements are an option for maintaining magnesium levels in people whose magnesium levels remain low despite diet changes.
What are the symptoms of low magnesium?
The most common symptoms of deficiency are muscle cramping, fatigue, depression, and irritability.
Can you take magnesium oxide while pregnant?
In addition, pregnant and breastfeeding mothers should avoid magnesium oxide as risks are unknown. It is crucial to consult with a doctor before starting or continuing supplements.
Is it safe to take magnesium oxide?
Typically, yes, it is safe to take magnesium oxide daily. However, you should check with your doctor before taking any supplement on a long-term basis.
Can magnesium oxide cause diarrhea?
Upset stomach and diarrhea are the most common side effects of magnesium oxide. Taking magnesium oxide with food can reduce stomach troubles. If side effects are persistent or if they worsen, it is best to talk to your doctor. Most people using magnesium oxide supplements do not have serious side effects. Serious allergic reactions ...
What is magnesium oxide used for?
Doctors may use magnesium oxide to treat constipation or as an antacid for heartburn or indigestion.
What is the role of magnesium in the body?
Magnesium plays a crucial role in the body, regulating blood pressure, blood sugar levels, neurotransmitters, and more. For those who do not get enough magnesium, taking more may.
What is magnesium citrate?
Magnesium citrate is a popular form of magnesium. It is often an ingredient in supplements and appears to be easier for the body to absorb than some other forms.
How much magnesium does the body absorb?
However, the body only absorbs around 30–40% of the dietary magnesium a person consumes. This, combined with the relatively small amount of foods that contain high amounts of magnesium, may make it challenging for some people to get enough of this nutrient from their diet.
Which magnesium is the fastest to absorb?
A 2019 animal study found that out of several types of magnesium, magnesium malate was the fastest to absorb after a single dose. This may also be true of humans, but human trials are necessary to confirm this.
What is the most common mineral in the body?
Magnesium is one of the most common minerals in the body. It plays a role in over 600. Trusted Source. metabolic reactions, including energy production, protein formation, and blood pressure regulation. However, some people need extra help getting enough magnesium.
Is magnesium a mineral?
For example, some types of magnesium are easier to absorb than others. Magnesium is one of the most common minerals in the body. It plays a role in over 600.
What is magnesium oxide?
Magnesium oxide is a salt that combines magnesium and oxygen. It naturally forms a white, powdery substance and may be sold in powder or capsule form. It’s also the main active ingredient in milk of magnesia, a popular over-the-counter medication for constipation relief ( 6 ).
How is magnesium sulfate formed?
Magnesium sulfate is formed by combining magnesium, sulfur, and oxygen. It’s commonly referred to as Epsom salt.
What is magnesium glycinate made of?
Magnesium glycinate is formed from elemental magnesium and the amino acid glycine.
Which is better for high blood sugar: magnesium taurate or magnesium L-threonate?
Magnesium taurate may be the best form for managing high blood sugar and high blood pressure, though more studies are necessary. 7. Magnesium L-threonate. Magnesium L-threonate is the salt formed from mixing magnesium and threonic acid, a water-soluble substance derived from the metabolic breakdown of vitamin C ( 21 ).
Why do we dissolve magnesium sulfate in water?
Magnesium sulfate is frequently dissolved in bathwater to soothe sore, achy muscles and relieve stress. It’s also sometimes included in skin care products, such as lotion or body oil.
Does magnesium malate help with digestion?
Research suggests that magnesium malate is very well absorbed in your digestive tract, making it a great option for replenishing your magnesium levels ( 14 ).
Is magnesium lactate a preservative?
This acid is not only produced by your muscle and blood cells but also manufactured for use as a preservative and flavoring agent ( 11 ). Indeed, magnesium lactate is utilized as a food additive to regulate acidity and fortify foods and beverages. It’s less popular as an over-the-counter dietary supplement.
How much improvement does magnesium oxide give?
In fact, over 70% of those treated with magnesium oxide reported overall symptom improvement, compared with only 25% of those in the placebo group ( 19 ).
How does it compare with other forms of magnesium?
Magnesium oxide and other inorganic salts of magnesium like magnesium carbonate are high in elemental magnesium, which is the total amount of magnesium in a supplement ( 3 ).
What is magnesium used for?
Magnesium is a mineral that’s needed for many bodily processes, including blood sugar regulation, nerve function, energy production, and DNA synthesis ( 1. Trusted Source. ). It’s found in a number of foods but can also be taken as a dietary supplement. These supplements contain different forms of magnesium, including magnesium citrate, ...
Which magnesium supplement has the lowest bioavailability?
A 2019 study confirmed this by testing 15 magnesium supplements and finding that a supplement containing only magnesium oxide had the lowest bioavailability ( 3 ).
Which is more effective for magnesium absorption?
In contrast, magnesium citrate, magnesium acetyl taurate, magnesium malate, and magnesium glycinate all have high absorption rates and are more effective at increasing magnesium levels in the body ( 6, 7, 8, 9, 10 ).
Which magnesium is better for migraines?
However, other forms of magnesium like magnesium sulfate and magnesium citrate may be more effective at treating migraine, as they are typically better absorbed ( 14, 15, 16 ).
Does magnesium oxide help with headaches?
Studies show that magnesium oxide may reduce headache symptoms. It may even be as effective as some migraine headache medications ( 11 ).

Overview
This medication is a mineral supplement used to prevent and treat low amounts of magnesium in the blood. Some brands are also used to treat symptoms of too much stomach acid such as stomach upset, heartburn, and acid indigestion.
May Treat: Hypomagnesemia
Brand Names: Uro-Mag · MagOx · Maox · Laxative Dietary Supplement · MagGel and more
Drug Class: Antacid - Magnesium · Minerals and Electrolytes - Magnesium
Availability: Prescription sometimes needed
Lactation: Does not adversely affect lactation
Related oxides
Electric properties
Production
Applications
Magnesium oxide (MgO), or magnesia, is a white hygroscopic solid mineral that occurs naturally as periclase and is a source of magnesium (see also oxide). It has an empirical formula of MgO and consists of a lattice of Mg ions and O ions held together by ionic bonding. Magnesium hydroxide forms in the presence of water (MgO + H2O → Mg(OH)2), but it can be reversed by heating it to remove moist…
Precautions
While "magnesium oxide" normally refers to MgO, magnesium peroxide MgO2 is also known as a compound. According to evolutionary crystal structure prediction, MgO2 is thermodynamically stable at pressures above 116 GPa (gigapascals), and a semiconducting suboxide Mg3O2 is thermodynamically stable above 500 GPa. Because of its stability, MgO is used as a model system for investigating vibrational properties of crystals.
See also
Pure MgO is not conductive and has a high resistance to electric current at room temperature. The pure powder of MgO has a relative permettivity inbetween 3.2 to 9.9 with an approximate dielectric loss of tan(δ) > 2.16x10 at 1kHz.
External links
Magnesium oxide is produced by the calcination of magnesium carbonate or magnesium hydroxide. The latter is obtained by the treatment of magnesium chloride MgCl 2 solutions, typically seawater, with limewater or milk of lime.
Mg + Ca(OH)2 → Mg(OH)2 + Ca
Calcining at different temperatures produces magnesium oxide of different reactivity. High temp…