
What type of proteoglycans are found in bones?
Bones contain small leucine-rich proteoglycans (SLRPs). Decorin and biglycan are predominantly present in bones and contain either chondroitin sufate or dermatan sulfate. These proteoglycans are required in all phases of bone development such as proliferation of cells, deposition of matrix, and mineral deposition of cells.
What is the function of proteoglycans?
Function of proteoglycans in the extracellular matrix Proteoglycans are glycosylated proteins which have covalently attached highly anionic glycosaminoglycans.
Are proteoglycans present in extracellular matrix?
Many forms of proteoglycans are present in virtually all extracellular matrices of connective tissues.
Are proteoglycans highly glycosylated?
Proteoglycans are highly glycosylated and, in many cases, 90–95% of the molecular weight of the typical proteoglycan is due to the carbohydrate components. The carbohydrate components of proteoglycans are known as glycosaminoglycans.
Where are proteoglycans most abundant?
connective tissuesThey are found in all connective tissues, extracellular matrix (ECM) and on the surfaces of many cell types. Proteoglycans are remarkable for their diversity (different cores, different numbers of GAGs with various lenghts and compositions).
What is the function of proteoglycans in the body?
The major biological function of proteoglycans derives from the physicochemical characteristics of the glycosaminoglycan component of the molecule, which provides hydration and swelling pressure to the tissue enabling it to withstand compressional forces.
What are proteoglycans examples?
Proteoglycans are a type of glycoproteins present in the body, especially in connective tissues, bone and cartilage, and cell surfaces. Examples of proteoglycans are versican (a large chondroitin sulfate proteoglycan), perlecan, neurocan, aggrecan, brevican, fibromodulin, and lumican.
Are proteoglycans in cartilage?
The predominant proteoglycan present in cartilage is the large chondroitin sulfate proteoglycan 'aggrecan'. Following its secretion, aggrecan self-assembles into a supramolecular structure with as many as 50 monomers bound to a filament of hyaluronan.
Are proteoglycans in bone?
Small Leucine Rich Proteoglycans (SLRPs) are an important family of PGs described in bones. SLRPs are localized in most skeletal regions and play a major role during all phases of bone formation, including cell proliferation, organic matrix deposition, remodeling, and mineral deposition.
Where do proteoglycans attach to?
extracellular matrixAll core proteins contain a glycosaminoglycan substitution domain and most of the proteoglycans are attached to macromolecules present in the extracellular matrix through domains that are present in the core proteins.
What is the most abundant proteoglycan?
hyaluronic acidThe largest in size and most abundant by weight is aggrecan, a proteoglycan that possesses over 100 chondroitin sulfate and keratan sulfate chains. Aggrecan is also characterized by its ability to interact with hyaluronic acid to form large proteoglycan aggregates.
Is proteoglycan a carbohydrate?
Proteoglycans are a subclass of glycoproteins in which the carbohydrate units are polysaccharides that contain amino sugars. Such polysaccharides are also known as glycosaminoglycans.
What is proteoglycan made of?
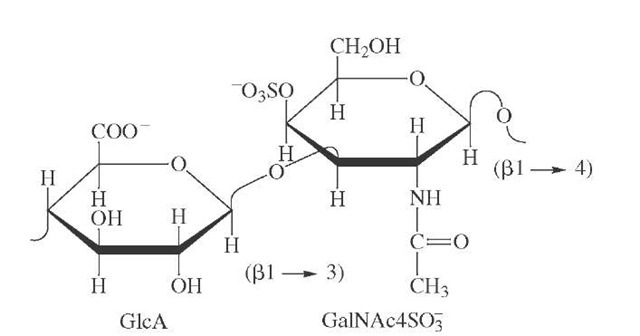
Proteoglycans consist of a core protein and one or more covalently attached GAG chains (Figure 11.2). GAGs are linear polysaccharides, whose building blocks (disaccharides) consist of an amino sugar (either GlcNAc or GalNAc) and an uronic acid (GlcA and IdoA).
What are proteoglycans in joints?
The proteoglycans are large macromolecules consisting of a protein core to which are attached multiple chains of glycosaminoglycans and oligosaccharides. During normal and pathological turnover, degradation products are released to the synovial fluid and to the circulation.
What is the difference between proteoglycan and glycoprotein?
Definition. A proteoglycan refers to a compound consisting of a protein bonded to glycosaminoglycan groups, present especially in connective tissue while a glycoprotein refers to any class of proteins which have carbohydrate groups attached to the polypeptide chain.
Which of the following statement is correct about is proteoglycans?
The correct answer is c. Proteoglycans are composed of carbohydrates and proteins. Unbranched heteropolysaccharides that are known as glycosaminoglycans, covalently attach to proteins forming proteoglycan.
What do you mean by proteoglycans?
Proteoglycans are proteins that are heavily glycosylated. The basic proteoglycan unit consists of a "core protein" with one or more covalently attached glycosaminoglycan (GAG) chain(s).
What are proteoglycans quizlet?
a class of glycoproteins composed of many long, linear chains of glycosaminoglycans (GAGs) covalently linked to a core protein.
What is the difference between proteoglycans and glycoproteins?
Glycoproteins are proteins to which carbohydrates are covalently linked through glycosidic bonds. Proteoglycans are a subclass of glycoproteins with distinctive features of carbohydrate structure.
What produces proteoglycans?
Proteoglycans are cartilaginous tissues produced from marine by-products that include glycosaminoglycans, namely chondroitin-4-sulphate and chondroitin-6-sulphate (Garnjanagoonchorn et al., 2007; Iozzo and Schaefer, 2015).
Where are proteoglycans found?
Proteoglycans are mostly found at the cell surface or in the extracellular matrix. In embryonic kidneys, sulfated proteoglycans are concentrated around the tip of the ureteric bud, and perturbation of their synthesis by β-D-xyloside results in the inhibition of ureteric bud branching morphogenesis. 217,218 This perturbation also abolishes the expression of Wnt11 at the tip of the ureteric bud. 219 As described previously, loss of Wnt11 expression at the tip of the ureteric bud can lead to loss of Gdnf expression in the metanephric mesenchyme. However, the linking mechanism between inhibition of sulfated proteoglycan and the loss of Wnt11 expression is unclear. Along the same lines, genetic inactivation of an enzyme, heparan sulfate 2-O-sulfotransferase, involved in heparan sulfate proteoglycan synthesis results in kidney agenesis. 53 In this case, the ureteric bud forms from the Wolffian duct, but subsequent invasion of the metanephric mesenchyme is perturbed. As for the mesenchyme, initial specification of the metanephric mesenchyme appears intact, but subsequent mesenchymal condensation is affected. Consistent with the in vitro result described above, heparan sulfate biosynthesis perturbation results in loss of Wnt11 expression and reduced Gdnf expression. Given the fact that many growth factors that regulate branching morphogenesis of the ureteric bud are heparin (a heavily sulfated form of GAG chain)-binding, it is likely that global inhibition of GAG chain synthesis or its sulfation could compromise actions of these heparin-binding growth factors.
What are proteoglycans?
Proteoglycans also are commonly referred to in the older literature as connective tissue mucins or mucopolysaccharides. These molecules are large glycoconjugate complexes that are found in high concentrations within the extracellular matrix of connective tissues. Proteoglycans are highly glycosylated and, in many cases, 90–95% of the molecular weight of the typical proteoglycan is due to the carbohydrate components.
Which sarcomas express glycosaminoglycans?
Glycosaminoglycans and proteoglycans are expressed by a number of different sarcomas. Hyaluronic acid and chondroitin sulfates in particular may be found in high concentrations in myxoid chondrosarcomas as well as the myxoid variants of liposarcoma and malignant fibrous histiocytoma ( Tighe 1963; Kindblom & Angervall 1975; Weiss & Goldblum 2001 ). In addition, proteoglycans may be observed in the stromal components of sarcomas as well as some carcinomas.
Which amino acids are covalently bound to the protein core of the proteoglycan?
The glycosaminoglycan chains are covalently bound to a protein core of the proteoglycan via the side chain of the amino acids serine or threonine ( O -glycosidic linkage) and to a lesser extent to asparagine ( N -glycosidic linkage). The number of glycosaminoglycan chains varies greatly among different proteoglycans. While some proteoglycans may contain only one or two glycosaminoglycans per protein core, other proteoglycans may contain as many as 100 glycosaminoglycans ( Varki et al 1999 ). An exception to this structural motif is hyaluronic acid which does not contain a covalently bound protein core ( Mason et al 1982 ).
How do PGs form?
PGs occur if GAGs are bound to a protein chain. The PG gains a characteristic stretched form as a result of the strong negative loading of the GAG, because of which the molecules repel each other and seek the greatest possible distance from each other. This can be compared with the shape of a toilet brush. For PG aggregate to occur, many PGs must be bound to a central hyaluronic acid chain. Binding or link proteins are required for this.
Where does hyaluronic acid take place?
The synthesis of hyaluronic acid chains, protein chains and some oligosaccharides takes place in the endoplasmic reticulum of all connective tissue cells. Other oligosaccharides and GAGs are formed in the Golgi apparatus of the cell. The turnover of hyaluronic acid is about 2 to 4 days; for the other sulfated GAGs it is about 7 to 10 days. This means that the cells must always remain synthetically active, otherwise there is the risk of a reduction in the quantity of ground substance. The residual products of GAGs, which can be released outside the cells, have a feedback effect on the cells and this controls synthesis. Even the mechanical distortion of the cells itself represents a stimulus to synthesis.
What are the proteins in the ECM?
A proteoglycan consists of a core protein having a variable number of GAG side chains [21] ( Table 14.1). The ECM proteoglycan superfamily includes the small leucine-rich repeat (LRR) family, the lectican family, the collagen family, and others. Members of these families present in the brain ECM include phosphacan, agrin, versican (also known as PG-M), neurocan, and brain-enriched hyaluronan-binding (BEHAB [22]; also known as brevican), and the vascular basement membrane proteoglycan perlecan. In brain tumors, changes in the expression of several ECM proteoglycans are observed within the tumor-brain adjacent ECM and the tumor vasculature.
Where are proteoglycans produced?
As such, a proteoglycan is a long polysaccharide (sugar) chain covalently attached to a protein and produced in the endoplasmic reticulum (ER), a specialized organelle. PGs are more than 95% carbohydrate (sugar) by weight. But what does that mean? Well, imagine picking up a handful of sand and pebbles.
What is a proteoglycan?
Proteoglycans (PGs) are protein molecules that exist between cells in the human body, providing aid in healing , protection of organs, and other underappreciated functions . Learn the chemical composition, structure, and locations of proteoglycans. Updated: 11/02/2021
Why are PGs so heavy?
Because PGs often have lots and lots of chains of sugar chains, they are frequently very big and heavy molecules. All PGs have a particular sugar chain known as glycosaminoglycan (GAG). A GAG is a sequence of two sugars, where one of the sugars is a sugar acid, and the other contains an amine group.
What is the difference between proteoglycan and glycan?
The word proteoglycan helps to provide us with clues to the molecule's chemical composition. For example, proteo refers to protein, while glycan means sugar or a group of sugars. As such, a proteoglycan is a long polysaccharide (sugar) chain covalently attached to a protein and produced in the endoplasmic reticulum (ER), a specialized organelle.
What do PGs do?
PGs help protect cells from injury by outside forces.
Where are proteoglycans stored?
They are either secreted in the extracellular matrix, inserted into the plasma membrane, or stored in various secretory granules. Proteoglycans are basically a class of proteins that are heavily glycosylated. Here, the glycosaminoglycans are covalently attached to certain proteins called core proteins.
Why do different proteoglycans arise?
These glycosaminoglycans impart a negative charge on proteoglycans. Different proteoglycans arise due to the different glycosaminoglycans that are attached to it. Proteoglycans can be classified on the basis of glycosaminoglycans they possess.
What are the two proteins that are found in bones?
Decorin and biglycan are predominantly present in bones and contain either chondroitin sufate or dermatan sulfate. These proteoglycans are required in all phases of bone development such as proliferation of cells, deposition of matrix, and mineral deposition of cells. Biglycan is required for proper formation of collagen fibers and bone production ...
How many proteoglycans are in cartilage?
The extracellular matrix of cartilage contains five well-characterized proteoglycans. Out of all the proteoglycans present, aggrecan is the most abundant. It has more than hundred chondroitin sulfates and keratan sufaltes. They interact with molecules of hyaluronan to form large aggregates.
What percentage of proteoglycans are glycosaminoglycans?
Glycosaminoglycans make about 95% of the weight of proteoglycans; due to this, proteoglycans resemble more of polysaccharides rather than proteins.
What are glycosaminoglycans?
Glycosaminoglycans are long, unbranched molecules that contain repeating disaccharide units of a uronic acid such as glucuronic acid (GlcA) or iduronic acid. and an amino sugar (either N-acetylglucosamine, or N-acetylgalactosamine). These glycosaminoglycans impart a negative charge on proteoglycans. Different proteoglycans arise due ...
What are the two basic molecules of proteoglycans?
Proteoglycans are composed of two basic molecules, core protein and glycosaminoglycans. The core protein may contain serine residues; these residues act as a point of attachment to which different glycosaminoglycans attach. The glycosaminoglycans attach to the core proteins perpendicularly and give rise to a brush-like structure. Their attachment in most but not all proteoglycans is through a tetrasaccharide linker that consist of glucuronic acid (GlcA), two galactose (Gal) and a xylose (Xyl) residue via glycosidic bonds. And some glycosaminoglycans are linked to the protein core of proteoglycans through a trisaccharide linkage that lacks the GlcA residue.
What are proteoglycans made of?
These proteins are composed of glycosaminoglycan chains that are covalently attached to a protein core.
What is the function of proteoglycans?
Proteoglycans act as polysaccharides rather than proteins as 95% of their weight is composed of glycosaminoglycan. The glycosaminoglycan chains consist of alternating hexosamine and hexuronic acid or galactose units. There are also glycopeptide linkage regions that connect the polysaccharide chains to the core proteins that contain N- and/or O-linked oligosaccharides.
What are the two major families of cell surface heparan sulfate proteoglycans?
The two major families of cell surface heparan sulfate proteoglycans are syndecans and glypicans. These two families bind to several growth factors and matrix proteins and are involved in various signal transduction pathways implicated in the proliferation of cells and cell shape changes.
What are the proteins in the proteoglycan family?
These small core proteins include decorin, biglycan, fibromodulin, etc. Heparan sulfate proteoglycans are present in the extracellular or basement membrane and are related to the heparan sulfate proteoglycan secreted by ...
What is the elongation of glycosaminoglycan chains?
The elongation of the chain is initiated by xylosylation of specific serine residues. The serine units that are susceptible to xylosylation occur in the specific tetrapeptide sequence that is preceded by a few acidic residues. Synthetic peptides that contain this sequence have been shown to be adequate substrates for xylosylation in vitro.
What are syndecans?
Syndecans are transmembrane proteins that are linked to the cell membrane using glycosylphosphatidylinositol (GPI) lipid anchors. There are four known mammalian syndecan proteins. While the structure of these proteins is quite similar to several shared cytoplasmic, juxtamembrane and transmembrane domains, they also have distinct regions and distributions inside cells. Both conserved and divergent protein partners have roles in the cellular and developmental functions of proteoglycans.
Which domain is found in core proteins?
All core proteins contain a glycosaminoglycan substitution domain and most of the proteoglycans are attached to macromolecules present in the extracellular matrix through domains that are present in the core proteins.
Which proteoglycans are most closely associated with the surface of many cell types?
This group of four proteoglycans is closely associated with the surface of many cell types anchored via integrins or other receptors, but they can also be a part of most basement membranes. Pericellular proteoglycans are mostly HSPGs and include perlecan and agrin, which share homology especially at their C-termini, and collagens XVIII and XV, which share homology at their N- and C-terminal noncollagenous domains (Fig. 1).
What are the four major classes of proteoglycans?
These three signatures were utilized to design four major classes of proteoglycans with distinct forms and functions: the intracellular, cell-surface, pericellular and extracellular proteoglycans . The proposed nomenclature encompasses forty-three distinct proteoglycan-encoding genes and many alternatively-spliced variants. The biological functions of these four proteoglycan families are critically assessed in development, cancer and angiogenesis, and in various acquired and genetic diseases where their expression is aberrant.
What is the function of betaglycan?
Betaglycan is a ubiquitously-expressed cell surface proteoglycan that acts as a co-receptor for members of the TGFβ superfamily of Cys knot growth factors which also include activins, inhibins, GDFs and BMPs [69,70]. For example, betaglycan enhances the binding of all the TGFβ isoforms to the signaling TGFβ complex [71] and is needed for TGFβ2 high-affinity interaction with the receptor complex. Betaglycan also blocks the aggressiveness of ovarian granulosa cell tumors by suppressing NF-κB-evoked MMP2 expression [72]. Betaglycan, together with other TGFβ-binding SLRPs, i.e. decorin and biglycan (see below), can be cleaved by granzyme B, thereby releasing an active form of TGFβ [73]. Ectodomain shedding of betaglycan is indeed necessary for betaglycan-mediated suppression of TGFβ signaling and breast cancer migration and invasion [74]. The ability of betaglycan to affect epithelial mesenchymal transformation [70], together with genetic evidence of embryonic lethality in Tgfbr3−/−mice, suggests that betaglycan may play a unique and non-redundant function during development.
How are glypicans processed?
Second, glypicans are dually processed via partial proteases and lipases. In the former case, the ectodomain of glypicans is processed via endoproteolytic cleavage by a furin-like convertase. This processing generates two subunits that are then bound via disulfide bonds, in a way similar to the Met receptor. In the latter case, the entire glypican proteoglycan is released from the plasma membrane via an extracellular lipase (Notum) that cleaves the GPI anchor. Drosophilastudies have shown that the Notum-mediated release of glypican can regulate morphogen gradients including Wnt, BMP and Hh gradients [84].
What is the drosophilaortholog of CSPG4?
Perdido/Kon-tiki, the Drosophilaortholog of mammalian CSPG4, genetically interacts with integrins during Drosophilaembryogenesis, and its loss is embryonic lethal [62]. RNAi-mediated suppression of Perdido/Kon-tikiin the muscles, just before adult myogenesis starts, induces misorientation and detachment of Drosophilaadult abdominal muscle, generating a phenotype similar to the embryonic lethal ones [63]. Thus, it is possible that, based on its high conservation through species, mammalian CSPG4could also play a role in myogenesis and function as well.
What is the role of syndecan-1 in the cell?
An emerging new role for syndecan-1 is linked to its ability to reach the nuclei in a variety of cells. Initial observations showed that myeloma and mesothelioma cells contain syndecan-1 in their nuclei [46,47] and this nuclear translocation is also regulated by heparanase [46], indicating that there must be a cellular receptor for shed syndecan-1 that could mediate its nuclear targeting and transport. In support of these studies are previous observations that exogenous HS can translocate to the nuclei and modulate the activity of DNA Topoisomerase I [48] and histone acetyl transferase (HAT) [49]. N-terminal acetylation of histones by HAT is linked to transcriptional activation, and this process is finely tuned by its counteracting enzyme, histone deacetylase (HDAC). Heparanase-evoked loss of nuclear syndecan-1 causes an increase in HAT enzymatic activity and enhances transcription of pro-tumorigenic genes [50]. Syndecan-1 that is shed from myeloma tumor cells is uptaken by bone marrow stromal cells and is transported to the nuclei by a mechanism that requires its HS chains, as this process is inhibited by heparin and chlorate [51]. Once nuclear, soluble syndecan-1 binds to HAT p300 and inhibits its activity, thereby providing a new mechanism for tumor–host cell interaction and cross-talk [52].
What are syndecans involved in?
Syndecans are involved in a wide variety of biological functions, too vast to be reviewed here, but reviewed recently [5,30,34]. Briefly, syndecans bind numerous growth factors, especially through their HS chains, and dictate morphogen gradients during development. In concert with other cell-surface HSPGs, syndecans can act as endocytosis receptors and are also involved in the uptake of exosomes [35]. Syndecans play key roles as co-receptors for many RTKs and can also function as receptors for atherogenic lipoproteins [36]. Indeed, there is strong genetic evidence that syndecan-1 is the main HSPG mediating clearance of triglyceride-rich lipoproteins derived from either the liver or from intestinal absorption [37].
Where are proteoglycans found?
Many forms of proteoglycans are present in virtually all extracellular matrices of connective tissues. The major biological function of proteoglycans derives from the physicochemical characteristics of the glycosaminoglycan component of the molecule, which provides hydration and swelling pressure to the tissue enabling it to withstand compressional ...
What is the function of proteoglycans?
Function of proteoglycans in the extracellular matrix. Proteoglycans are glycosylated proteins which have covalently attached highly anionic glycosaminoglycans. Many forms of proteoglycans are present in virtually all extracellular matrices of connective tissues.
How much proteoglycans should I take daily?
Very low oral doses of proteoglycans, ranging from 5 to 10 mg daily, have evidence in these regards.
How do proteoglycans help with OA?
In addition to holding moisture in the tissue matrix, proteoglycans help structurally protect the cartilage from deterioration. As OA progresses, however, the proteoglycans are lost from the superficial articular cartilage surface to which the type II collagen runs parallel and the highest tensile forces, due to shear stress, exist. As the body dynamically attempts to remodel and repair the tissue in the early stages of OA, an increase in proteoglycans is seen deeper within the tissue, as if the body is attempting to counteract the surface changes. [18] The concentration of proteoglycans decreases in the later stages of disease, [19] however, with proteoglycan fragments in addition to the enzymes contributing to their degradation being observed in the joint synovial fluid. [20], [21] The degradation of aggrecans via a family of enzymes collectively known as aggrecanases has been recognized as a major mechanism contributing to the histopathological changes associated with OA, and medications directed at inhibiting this process are in an ongoing process of investigation. [22]
What is the link protein?
Interestingly, protein fragments (known as link proteins [33]) of proteoglycan aggregates found in cartilage have been observed to stimulate proteoglycan and collagen synthesis in in vitro studies. [34], [35] The link protein N-terminal peptide has also been shown to stimulate cartilage regeneration by increasing the proliferation, migration, and chondrogenic differentiation of cartilage stem/progenitor cells. [36] Additionally, link protein N-terminal peptide counteracts degenerative changes induced by IL-1α. [37]
Does salmon nasal cartilage contain proteoglycans?
Salmon nasal cartilage primarily contains the proteoglycan aggrecan, which has an amino acid composition and functional domains very similar to the aggrecan found in the articular cartilage of mammals. [50] In cell cultures, salmon nasal cartilage proteoglycans (SNCPs) have been shown to reduce matrix metalloproteinase expression, [51] a primary contributor to cartilage degradation. [52] Early studies performed with SNCPs in macrophages stimulated with heat-killed Escherichia coli suggest these proteoglycans have immunomodulatory and anti-inflammatory effects [53] —treatment with the SNCP solution reduced levels of the pro-inflammatory cytokine TNF-α and nitric oxide synthase (also a contributor to the joint destruction in OA [54] ), and increased IL-10.
Is salmon cartilage a proteoglycan?
Salmon nasal cartilage is one source of proteoglycans that has been highly researched with positive findings in a myriad of settings, including several animal models of autoimmunity, [38], [39], [40], [41] allergies, [42] infection, [43] wound healing, [44] diet-induced inflammation, [45] and ultraviolet (UV) light–associated skin damage, [46] as well as clinical studies of joint pain, [47] arthritis, [48] and skin aging. [49]
Do chondrocytes replicate?
Unfortunately, chondrocytes have minimal ability to replicate, and their ability to synthesise cartilage and proteoglycans declines with age, which is one issue that contributes to diminished articular tissue integrity with age. [11] Proteoglycans are also essential to the health of the skin. Similar to cartilage, proteoglycans affect ...
How many members are in the proteoglycan family?
The syndecan family consists of four members, syndecan-1, -2, -3, and -4.
What is the secretory vesicle proteoglycan?
Serglycin is also known as hematopoietic proteoglycan. The core protein of serglycin is encoded by the SRGN gene. Serglycin is the major proteoglycan found in cytoplasmic secretory granules within endothelial, endocrine, and hematopoietic cells. Serglycin contains a variable number of GAG attachment sites to which either heparin chains or chondroitin sulfate chains are attached. Heparin, a more highly sulfated form of heparan sulfate, is made exclusively on serglycin that is produced in mast cells associated with connective tissue. Although there are other secretory proteoglycans (e.g. chromogranin A) these have only been shown to be part-time proteoglycans.
Where is brevican found?
belongs to the lectican family; a chondroitin sulfate proteoglycan (CSPG); protein core encoded by the BCAN gene; predominantly expressed in the central nervous system; brevican protein devoid of glycosaminoglycan chains is also found within the brain
What are the four members of the lectican family?
The lectican family (also referred to as the aggrecan family) of interstitial proteoglycans consists of aggrecan, brevican, neurocan, and versican. All four members of this proteoglycan family contain a unique protein core and each core protein contains an amino-terminal domain that can bind hyaluronans, a central domain to which chondroitin sulfates and dermatan sulfates are attached, and a carboxy-terminal domain that contains a C-type lectin domain. The core protein of aggrecan is encoded by the ACAN gene. The core protein of brevican is encoded by the BCAN gene. The core protein of neurocan is encoded by the NCAN gene. The core protein of versican is encoded by the VCAN gene. Aggrecan itself is the most well characterized member of aggrecan family and is the major proteoglycan in cartilage. Aggrecan can contain upwards of 100 chondroitin sulfate chains and can also contain keratan sulfate chains dependent upon the tissue of synthesis. Neurocan is produced in the late embryonic central nervous system (CNS) and can inhibit neurite outgrowth. Brevican is produced in the terminally differentiated CNS. Versican is produced predominantly by connective tissue cells. The protein core of versican is variable due to alternative splicing of the VCAN mRNA resulting in a family of protein cores in the versicans. Versicans have major roles in neural crest cell and axonal migration.

Structure of Proteoglycans
Diverse Functions of Proteoglycans
- Proteoglycans in Cartilage
The extracellular matrix of cartilage contains five well-characterized proteoglycans. Out of all the proteoglycans present, aggrecan is the most abundant. It has more than hundred chondroitin sulfates and keratan sufaltes. They interact with molecules of hyaluronan to form large aggrega… - Proteoglycans in the Bone
The bone matrix has a lower proteoglycan content than those in the cartilage. This is the reason bones take up less quantity of water and are, thus, more brittle. Bones contain small leucine-rich proteoglycans (SLRPs). Decorin and biglycan are predominantly present in bones and contain eit…
Other Functions of Proteoglycans
- ◆ Proteoglycans, many a time, act as cell surface receptors for different enzymes or their inhibitors. Some proteoglycans that are present in the extracellular matrix may bind to different cytokines, growth factors, and other compounds and prevent their degeneration by proteases. ◆ They can act as endocytic receptors for the clearance of bound ligands. ◆ They cooperate with …