
Which family does manganese belong to on the periodic table?
The highlighted elements of this periodic table belong to the alkaline earth element family. Todd Helmenstine. The alkaline earth metals or simply alkaline earths are recognized as an important group and family of elements. These elements are metals. Examples include calcium and magnesium.
What is the most dangerous element of the periodic table?
When you're looking at reactive elements on the periodic table, one of the most violent is francium. But... it's kind of hard to tell. According to ThoughtCo., we're not entirely sure just what francium is capable of, because there's only ever been very, very small quantities produced. But that might actually be a good thing, because it doesn't just react, it reacts by exploding.
What are the man made elements on the periodic table?
What are the man-made elements in the periodic table? Technetium, promethium, and the transuranic elements: neptunium through oganesson.
What is the formula for manganese?
Manganese;manganese(2+);oxygen(2-) | Mn2O | CID 10313135 - structure, chemical names, physical and chemical properties, classification, patents, literature ...

What state of matter is manganese commonly found in?
solidManganese is a chemical element with symbol Mn and atomic number 25. Classified as a transition metal, Manganese is a solid at room temperature.
What is manganese known for?
Manganese is used to produce a variety of important alloys and to deoxidize steel and desulfurize. It is also used in dry cell batteries. Manganese is used as a black-brown pigment in paint. It is an essential trace element for living creatures.
What type of metal is manganese?
manganese (Mn), chemical element, one of the silvery white, hard, brittle metals of Group 7 (VIIb) of the periodic table.
What metals contain manganese?
Manganese is also used to make alloys with metals other than iron or steel. For example, the alloy known as manganin is 84 percent copper, 12 percent manganese, and 4 percent nickel. Manganin is used in electrical instruments.
What makes manganese unique?
Manganese is a very hard, brittle, gray-white transition metal that is naturally found in a variety of minerals, but never on its own. Manganese is one of the most common elements in Earth's crust and is widely distributed across the planet's surface. Manganese is vital to human and animal life in metabolic functions.
What is manganese used for in industry?
As much as 90 percent of manganese consumption, both in the United States and globally, is accounted for by the steel industry. Manganese removes oxygen and sulfur when iron ore (an iron and oxygen compound) is converted into iron. It also is an essential alloy that helps convert iron into steel.
Is manganese harmful to humans?
Workers may be harmed from exposure to manganese through the breathing of manganese fumes or dusts. Continued exposure can damage the lungs, liver, and kidneys. Exposure to manganese dust or fumes can also lead to a neurological condition called manganism.
Is manganese toxic?
Manganese toxicity can result in a permanent neurological disorder known as manganism with symptoms that include tremors, difficulty walking, and facial muscle spasms. These symptoms are often preceded by other lesser symptoms, including irritability, aggressiveness, and hallucinations.
Where is manganese found?
The main mining areas for manganese are in China, Africa, Australia and Gabon.
How is manganese obtained?
The metal is obtained by reducing the oxide with sodium, magnesium or aluminium, or by the electrolysis of manganese sulfate. Manganese nodules have been found on the floor of the oceans. These nodules contain about 24% manganese, along with smaller amounts of many other elements.
What enzymes contain manganese?
Many types of enzymes contain manganese. For example, the enzyme responsible for converting water molecules to oxygen during photosynthesis contains four atoms of manganese. Some soils have low levels of manganese and so it is added to some fertilisers and given as a food supplement to grazing animals.
What is manganese steel used for?
Manganese steel contains about 13% manganese. This is extremely strong and is used for railway tracks, safes, rifle barrels and prison bars.
How much manganese is in the human body?
The average human body contains about 12 milligrams of manganese. We take in about 4 milligrams each day from such foods as nuts, bran, wholegrain cereals, tea and parsley. Without it, bones grow spongier and break more easily. It is also essential for utilisation of vitamin B1.
How are elements organized into blocks?
Elements are organised into blocks by the orbital type in which the outer electrons are found. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The number of protons in an atom.
What is a vertical column in the periodic table?
A vertical column in the periodic table. Members of a group typically have similar properties and electron configurations in their outer shell. A horizontal row in the periodic table.
Where is manganese found in the body?
Manganese is found in the enzymes necessary to metabolize fats and carbohydrates. Manganese is found in the bones, liver, kidneys, and pancreas. Manganese is important in the processes that form bones, clots blood, and regulates blood sugar. As important as manganese is to our health, the body does not store manganese.
What is the melting point of manganese?
Properties: Manganese has a melting point of 1244+/-3°C, boiling point of 1962°C, specific gravity of 7.21 to 7.44 (depending on allotropic form ), and valence of 1, 2, 3, 4, 6, or 7. Ordinary manganese is a hard and brittle gray-white metal. It is chemically reactive and slowly decomposes in cold water. Manganese metal is ferromagnetic (only) ...
What is manganese dioxide used for?
Manganese dioxide is used as a depolarizer in dry cells and as a decolorizing agent for glass that has been colored green due to iron impurities. The dioxide is also used in drying black paints and in the preparation of oxygen and chlorine.
What is the difference between silica glass and manganese dioxide?
Manganese dioxide is used to make clear glass. Normal silica glass is tinted green and the manganese oxides add a purple tint to the glass that cancels out the green. Because of this property, glassmakers called it 'glassmaker's soap'. Manganese is found in the enzymes necessary to metabolize fats and carbohydrates.
Why is manganese used in steel?
Manganese is used in steel production to fix the sulfur found in iron ores. It also strengthens steel and prevents oxidation.
How many isotopes are there in manganese?
Isotopes: There are known 25 isotopes of manganese ranging from Mn-44 to Mn-67 and Mn-69. The only stable isotope is Mn-55. The next most stable isotope is Mn-53 with a half-life of 3.74 x 10 6 years. Density (g/cc): 7.21
How did Gahn get manganese?
Sources: In 1774, Gahn isolated manganese by reducing its dioxide with carbon. The metal may also be obtained by electrolysis or by reducing the oxide with sodium, magnesium, or aluminum. Manganese-containing minerals are widely distributed.
Where is manganese found?
It is a critical component in dozens of proteins and enzymes. It is found mostly in the bones, but also the liver, kidneys, and brain. In the human brain, the manganese is bound to manganese metalloproteins, most notably glutamine synthetase in astrocytes . Manganese was first isolated in 1774.
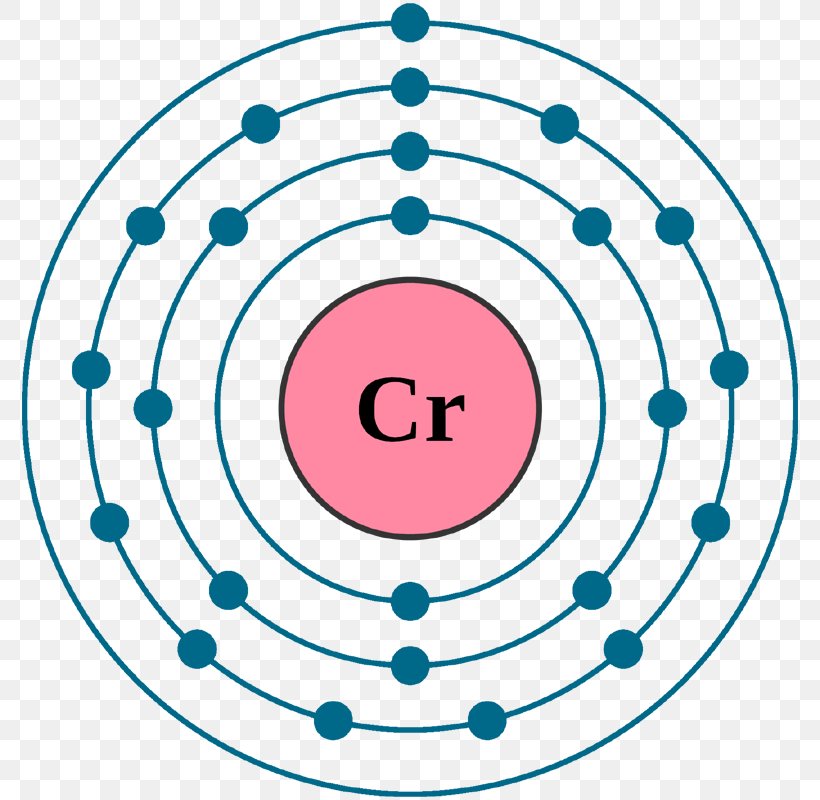
What is the atomic number of manganese?
Manganese is a chemical element with the symbol Mn and atomic number 25. It is a hard brittle silvery metal, often found in minerals in combination with iron. Manganese is a transition metal with a multifaceted array of industrial alloy uses, particularly in stainless steels.
What is the color of manganese?
Manganese (II) chloride crystals – the pale pink color of Mn (II) salts is due to a spin-forbidden 3d transition. The most common oxidation states of manganese are +2, +3, +4, +6, and +7, though all oxidation states from −3 to +7 have been observed. Mn 2+ often competes with Mg 2+ in biological systems.
What is the half life of 53 Mn?
53 Mn decays to 53 Cr with a half-life of 3.7 million years. Because of its relatively short half-life, 53 Mn is relatively rare, produced by cosmic rays impact on iron.
How many isotopes are in manganese?
Naturally occurring manganese is composed of one stable isotope, 55 Mn. Several radioisotopes have been isolated and described, ranging in atomic weight from 44 u ( 44 Mn) to 69 u ( 69 Mn). The most stable are 53 Mn with a half-life of 3.7 million years, 54 Mn with a half-life of 312.2 days, and 52 Mn with a half-life of 5.591 days.
Where did the name manganese come from?
The origin of the name manganese is complex. In ancient times, two black minerals were identified from the regions of the Magnetes (either Magnesia, located within modern Greece, or Magnesia ad Sipylum, located within modern Turkey). They were both called magnes from their place of origin, but were considered to differ in sex. The male magnes attracted iron, and was the iron ore now known as lodestone or magnetite, and which probably gave us the term magnet. The female magnes ore did not attract iron, but was used to decolorize glass. This female magnes was later called magnesia, known now in modern times as pyrolusite or manganese dioxide. Neither this mineral nor elemental manganese is magnetic. In the 16th century, manganese dioxide was called manganesum (note the two Ns instead of one) by glassmakers, possibly as a corruption and concatenation of two words, since alchemists and glassmakers eventually had to differentiate a magnesia nigra (the black ore) from magnesia alba (a white ore, also from Magnesia, also useful in glassmaking). Michele Mercati called magnesia nigra manganesa, and finally the metal isolated from it became known as manganese (German: Mangan ). The name magnesia eventually was then used to refer only to the white magnesia alba (magnesium oxide), which provided the name magnesium for the free element when it was isolated much later.
When was manganese first discovered?
Manganese was first isolated in 1774 . It is familiar in the laboratory in the form of the deep violet salt potassium permanganate. It occurs at the active sites in some enzymes. Of particular interest is the use of a Mn-O cluster, the oxygen-evolving complex, in the production of oxygen by plants.
What foods contain manganese?
Includes a variety of protein foods such as lean meats; poultry; eggs; seafood; beans, peas, and lentils; nuts and seeds; and soy products. Nuts, legumes, and mollusks contain manganese. Limits foods and beverages higher in added sugars, saturated fat, and sodium.
What is manganese involved in?
Through the action of these enzymes, manganese is involved in amino acid, cholesterol, glucose, and carbohydrate metabolism; reactive oxygen species scavenging; bone formation; reproduction; and immune response [ 3-7 ]. Manganese also plays a role in blood clotting and hemostasis in conjunction with vitamin K [ 5 ].
What is the DRI for manganese?
DRI is the general term for a set of reference values used for planning and assessing nutrient intakes of healthy people. These values, which vary by age and sex, include:
How much manganese is in the human body?
The human body contains about 10 to 20 mg manganese, of which 25% to 40% is in bone [ 1, 2 ]. The liver, pancreas, kidney, and brain also contain manganese. The body maintains stable tissue manganese concentrations through regulatory control of manganese absorption and excretion [ 5 ].
Does manganese increase the risk of illness?
Because of the role of manganese as a cofactor for several enzymes, low intakes might increase the risk of illness . This section focuses on two health areas in which manganese might be involved: bone health and diabetes.
Is manganese deficiency rare?
Manganese deficiency is very rare in humans, and signs and symptoms of deficiency have not been firmly established [ 1, 2 ]. The very limited evidence in humans suggests that manganese deficiency might cause bone demineralization and poor growth in children; skin rashes, hair depigmentation, decreased serum cholesterol, and increased alkaline phosphatase activity in men; and altered mood and increased premenstrual pain in women [ 2, 4 ]. Manganese deficiency might also alter lipid and carbohydrate metabolism and cause abnormal glucose tolerance [ 3 ].
Does the National Health and Nutrition Examination Survey include manganese?
The National Health and Nutrition Examination Survey, which provides dietary intake data for most nutrients, does not include manganese [ 19 ]. The Total Diet Study (TDS) is an FDA program that monitors the nutrient content of typical foods consumed by the U.S population [ 20 ].
Where are metals located on the periodic table?
The metals are located on the left side of the Periodic Table.
What are metals in Periodic table?
Metals are the elements which have the tendency to donate or lose electrons to form positive ions.
How many rare earth metals are there?
There are total 17 Rare Earth metals on the Periodic table. Rare Earth Metals includes all the 15 Lanthanides as well as scandium (Sc) and yttrium (Y). So total 15 + 2 = 17 Rare Earth metals.
Which group of metals is the most reactive?
They are the Alkali metals of group 1. In 1st group, as we move down from top to bottom, the reactive of metals increases. Thus the bottom most element of group 1 (i.e francium) is the most reactive metal on the Periodic table. ( Note: Francium is a laboratory made element.
What are the elements in group 3 to group 12?
The elements lying in group 3 to group 12 are known as Transition metals (or transition elements). Transition metals form a bridge between the chemically active metals of s-block elements and the less active elements of Groups 13 and 14. Thus these metals are known as “Transition metals”.
What are the two bottom rows of the periodic table called?
The two bottom rows in the Periodic table are called inner transition metals.
Which side of the periodic table has more atomic size?
And if the atomic size is more, then it has less tendency to attract the electron pair (means bigger the size, lesser is the electronegativity.) Now the elements on the left side of Periodic table have more atomic size. So they will have less electronegativity. Hence, they are more metallic in nature.
Occurrence
- Manganese is an abundant element. It is found in about 1000 ppm in the Earth’s crust. And it is ranked the 12th most abundant element on Earth, with considerable amounts present in water and soil. Manganese does not exist in its free elemental form. It is present in mineral, mostly in combination with iron. The most common minerals of manganese inc...
Physical Characteristics
- Manganese is a greyish-pink metal. It resembles iron in its physical appearance. It is brittle and hard. Manganese has a paramagnetic nature. when exposed to air and moisture, it undergoes slow oxidation that leads to rusting. Manganese is a dense metal, with a density of 7. 43 g.cm-3 at room temperature.
Chemical Characteristics
- Manganese is a reactive metal in its pure elemental form. It reacts with water and is burned in the presence of oxygen. Manganese dissolves in dilute acids. In compounds, the common oxidation states of manganese are +2, +3, +4 and +6, while the most stable oxidation state is +2. These compounds include manganese chloride, manganese carbonate and manganese sulphate.
Significance and Uses
- Manganese is widely used in making various industrially important alloys such as stainless steel.
- Manganese in its ionic form is used as pigments in various industries. For instance, manganese oxide is used to make violet colored glass.
- Manganese is used in alkaline and zinc-carbon batteries as cathode.
- Manganese is widely used in making various industrially important alloys such as stainless steel.
- Manganese in its ionic form is used as pigments in various industries. For instance, manganese oxide is used to make violet colored glass.
- Manganese is used in alkaline and zinc-carbon batteries as cathode.
- Manganese oxide is used in making fertilizers.
Health Hazards
- Manganese is a neurotoxic element. It can lead to damage to neurons and nervous system if inhaled in large amount. It has been found to linked with Parkinson’s disease in miners working with manganese. Manganese contamination in water has been a source of various intellectual impairments in humans, especially children. In trace amounts, however, manganese is an essen…
Isotopes of Manganese
- There is one naturally occurring isotope of manganese, manganese-55. There are eighteen radioactive isotopes of manganese, with atomic masses ranging from 46 to 65. Among the radioactive isotopes, manganese-53 is the most stable and have a half-life of around 4 million years.