How do you find the atomic mass on a periodic table?
0:552:23Understanding Atomic Number and Atomic Mass - YouTubeYouTubeStart of suggested clipEnd of suggested clipThe number that appears below the element symbol is called the atomic mass the mass of an atomMoreThe number that appears below the element symbol is called the atomic mass the mass of an atom depends on the number of protons neutrons.
What number is the atomic mass on the periodic table?
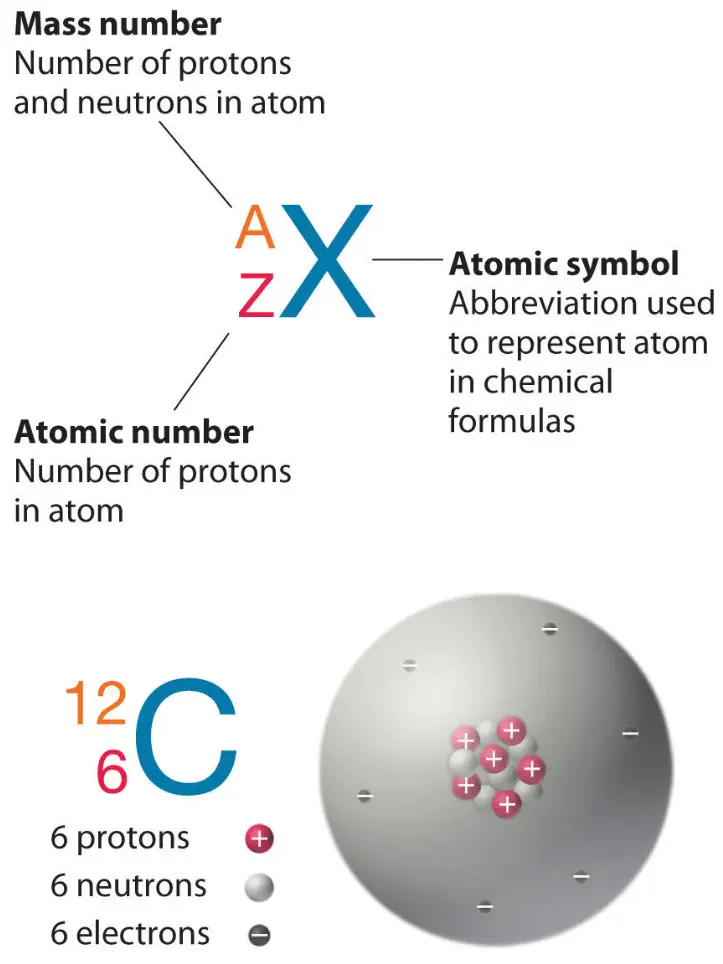
The mass number (represented by the letter A) is defined as the total number of protons and neutrons in an atom.
Is atomic mass included in the periodic table?
This is a periodic table with atomic mass, element name, element symbol, and atomic number. The atomic mass is the average number of protons and neutrons in atoms of a chemical elements, allowing for the natural abundances of the element's isotopes.
What is the atomic mass and where is it found?
Atomic mass can be defined as the total mass of one atom of any given element. The unit of atomic mass is called the unified atomic mass unit (denoted by 'u'). Most of the atomic mass of a substance is made up of protons and neutrons. Therefore, it is almost equal to its mass number.
How do you read the periodic table?
On the periodic table, elements are listed in order of increasing atomic number.Elements in the same row are in the same period. ... Elements in the same column are in the same group. ... Here's a close-up look at the carbon square from the Periodic Table.
How are atomic number of elements arranged in the periodic table?
In the modern periodic table, the elements are listed in order of increasing atomic number. The atomic number is the number of protons in the nucleus of an atom. The number of protons define the identity of an element (i.e., an element with 6 protons is a carbon atom, no matter how many neutrons may be present).
Why are elements on the periodic table not arranged by mass?
Assuming there were errors in atomic masses, Mendeleev placed certain elements not in order of increasing atomic mass so that they could fit into the proper groups (similar elements have similar properties) of his periodic table.
How is the periodic table arranged?
The periodic table is a tabular array of the chemical elements organized by atomic number, from the element with the lowest atomic number, hydrogen, to the element with the highest atomic number, oganesson. The atomic number of an element is the number of protons in the nucleus of an atom of that element.
What is the symbol of atomic mass?
u'Atomic mass is the average mass of the protons, neutrons, and electrons in an atom. Its unit is a unified atomic mass and is denoted by the symbol 'u'.
How do you find atomic mass without neutrons?
Subtract the atomic number from the atomic weight and you will get the atomic mass or the number of neutrons of that element.
What is atomic mass example?
An atomic mass unit is defined as a mass equal to one twelfth the mass of an atom of carbon-12. The mass of any isotope of any element is expressed in relation to the carbon-12 standard. For example, one atom of helium-4 has a mass of 4.0026amu. An atom of sulfur-32 has a mass of 31.972amu.
Is mass number and atomic mass same?
Mass number is a whole number because it is the sum of number of proton and number of neutrons whereas atomic mass is fractional because it is the average relative mass of its atoms as compared with mass an atom of C-12 isotope taken as 12.
Is mass number and atomic number same?
The major difference between atomic number and mass number is that the atomic number states the number of protons present in an atom whereas, the mass number indicates the total number of protons and the number of neutrons present in an atom.
Is mass number and atomic mass same?
Mass number is a whole number because it is the sum of number of proton and number of neutrons whereas atomic mass is fractional because it is the average relative mass of its atoms as compared with mass an atom of C-12 isotope taken as 12.
What are the elements 1 to 30?
Atomic Mass of First 30 ElementsATOMIC NUMBERELEMENTATOMIC MASS1Hydrogen1.0082Helium4.00263Lithium6.944Beryllium9.012226 more rows
What is atomic number equal to?
The number of protons in a nucleus is called the atomic number and always equals the number of electrons in orbit about that nucleus (in a nonionized atom).
Does the Modern Periodic Table Change? If So, How and Who Does That?
The periodic table as we know it today is managed by the International Union of Pure and Applied Chemistry, or IUPAC (eye-you-pack).
What is the CIAAW?
Since 1899 the IUPAC Commission on Isotopic Abundances and Atomic Weights ( CIAAW) has been evaluating atomic weights and abundances. For example, Carbon had an atomic weight of 12.00 in 1902 but today it is [12.0096, 12.0116]! Times sure have changed as the source of the sample will determine the value.
What is PubChem working with?
PubChem is working with IUPAC to help make information about the elements and the periodic table machine-readable.
Who is responsible for deciding what needs to be changed?
While much of what is in the periodic table is stable and unlikely to change, the IUPAC organization is responsible for deciding what needs to be changed. They have created criteria for what constitutes the discovery of a new element.
Can periodic table games be used for grade?
The periodic table game available on this page is for entertainment purposes only, and should not be used to grade students on their knowledge of chemical elements.
Who created the periodic table?
The creator of the periodic table, Dmitri Mendeleev, in 1869 began collecting and sorting known properties of elements, like he was playing a game, while traveling by train.
Did Mendeleev's predictions get dismissed?
There were plenty of skeptics and it took years to gain international acceptance, but once newly-discovered elements matched the ones that Mendeleev predicted, his patterns could not be dismissed. In addition, some of the properties that he "fudged" were later recalculated and found to be much closer to his predictions.
What is the atomic mass of a periodic table?
The atomic mass (atomic weight) values cited on a periodic table are weighted averages of naturally occurring isotopes. From one year to the next, the values may change slightly (usually only in the last significant digit) as more information about the source of each element becomes available.
What is the black and white periodic table?
This black and white periodic table contains the accepted atomic weights of each element as accepted by the IUPAC.
Where do the accepted values apply to chemistry?
For most chemistry calculations, the most recent table of accepted values should be used. These values do not apply to elements collected anywhere except the Earth's crust. The weighted atomic mass for an element from the Earth's core, the Moon, the Sun, etc., would not be the same as the accepted value.
What is the atomic number of an element?
The atomic number of an element is equal to the total number of protons in the nucleus of the atoms of that element. The atomic number can provide insight into the electronic configuration of the element. For example, carbon has an electron configuration of [He] 2s 2 2p 2, since its atomic number is 6.
What is the number of protons in the nucleus called?
The number of protons in the nucleus is called the atomic number. The atomic number of each element is unique.
Why is the atomic number of each element unique?
While the atomic number always stays the same some elements have atoms with different atomic mass numbers. This is because some elements have a different number of neutrons in the nucleus.
How to find the mass of an element?
The number of protons and the number of neutrons shall determine the mass number of an element. Since the isotopes of an element have slightly different mass numbers, it calculates the atomic mass by obtaining the mean of the mass numbers for its isotopes.
How can periodic trends be observed?
Periodic trends in the properties of the elements can be observed down the groups and across the periods of the modern periodic table. Every chemical element has a specific atomic number, which provides insight into the number of protons present within its nucleus.
Why is the atomic number important?
This number is very important, because it is unique to a given element’s atoms. An element’s atoms all have the same number of protons and each element has a different number of protons in its atoms. Test your knowledge on periodic table elements.
What is the name of the tabular arrangement of all the elements on the basis of their respective atomic numbers?
The periodic table is the tabular arrangement of all the chemical elements on the basis of their respective atomic numbers. In the periodic table , the vertical columns are called ‘groups’ and the horizontal rows are called ‘periods’.
What Is Atomic Mass?
Atomic mass is the sum of the masses of the protons, neutrons, and electrons in an atom, or the average mass, in a group of atoms. However, electrons have so much less mass than protons and neutrons that they don't factor into the calculation. So, the atomic mass is the sum of the masses of protons and neutrons. There are three ways to find atomic mass, depending on your situation. Which one to use depends on whether you have a single atom, a natural sample of the element, or simply need to know the standard value.
How to find the mass of an element?
To calculate the atomic mass of a single atom of an element, add up the mass of protons and neutrons.
Why do the atomic mass values on the periodic table change?
This happens when scientists revise the estimated isotope ratio in the crust. In modern periodic tables, sometimes a range of values is cited rather than a single atomic mass.
How to find the mass of an isotope?
Multiply each isotope's mass by its abundance. If your abundance is a percent, divide your answer by 100. Add these values together. The answer is the total atomic mass or atomic weight of the element.
How to convert percentages to decimals?
First, convert the percentages to decimal values by dividing each percentage by 100. The sample becomes 0.98 carbon-12 and 0.02 carbon-13. (Tip: You can check your math by making certain the decimals add up to 1. 0.98 + 0.02 = 1.00).
Is the atomic mass of carbon higher than the periodic table?
Advanced Note: This atomic mass is slightly higher than the value given in the periodic table for the element carbon. What does this tell you? The sample you were given to analyze contained more carbon-13 than average. You know this because your relative atomic mass is higher than the periodic table value, even though the periodic table number includes heavier isotopes, such as carbon-14. Also, note the numbers given on the periodic table apply to the Earth's crust/atmosphere and may have little bearing on the expected isotope ratio in the mantle or core or on other worlds.
Is electron mass factored in chemistry?
Electrons are much smaller than protons and neutrons, so their mass isn't factored into the calculation. Dr. Helmenstine holds a Ph.D. in biomedical sciences and is a science writer, educator, and consultant. She has taught science courses at the high school, college, and graduate levels. You may be asked to calculate atomic mass in chemistry ...