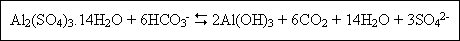Which acid is present in Fitkari?
Potash alum is also known as potassium alum or potassium aluminium sulfate. The chemical formula of potash alum is K2SO4.Al2(SO4)3. 24H2O. It is also commonly referred to as 'fitkari....Observations.The shape of the crystalOctahedralSolubility in waterSoluble2 more rows
Does alum have acid?
Most alums have an astringent and acid taste. They are colourless, odourless, and exist as a white crystalline powder. Alums are generally soluble in hot water, and they can be readily precipitated from aqueous solutions to form large octahedral crystals.
What is chemical formula of alum?
KAl(SO₄)₂·12H₂OPotassium alum / Formula
Is alum an acid salt?
Acidic salt has an ionizable proton present in it. , where X is a monovalent cation such as potassium or ammonium. “The salts that contain more than one cation or anion are known as double salt”. Therefore, alum is a double salt.
What is another name for alum?
noun Chemistry. Also called potash alum, potassium alum. a crystalline solid, aluminum potassium sulfate, K2SO4⋅Al2(SO4)3⋅24H2O, used in medicine as an astringent and styptic, in dyeing and tanning, and in many technical processes.
What alum is used for?
Alum (aluminum sulfate) is a nontoxic liquid that is commonly used in water treatment plants to clarify drinking water. It's use in lakes began in the early 1970's and is used to reduce the amount of phosphorus in the water.
Is alum basic or acidic?
acidicAqueous solution of Potash alum is acidic in nature because it is a double salt of strong acid (H2SO4) & weak base(Al(OH)3) Q. The aqueous solution of potash alum [K2SO4.
Can I drink alum water?
a variety of possible causes have been considered, however, no link between aluminium intake and the disease has been established. RESEARCH FINDINGS the csIro has conducted extensive research in this matter and in late 1998 found convincing evidence that the use of alum to treat drinking water is safe.
Is alum good for skin?
Yes, Alum, a naturally occurring ingredient is considered to be good for tightening the skin. It has an astringent property that causes the cells to shrink and removes excess oil from the skin. Thus, Alum is used in the form of gels or creams for skin care[8]. Alum is an effective remedy for skin problems.
Can we eat alum?
Yes, alum is edible. Potassium alum is an active ingredient in baking powder.
What is alum made out of?
Alum is a natural, crystalline stone that has been in use for thousands of years for its healing properties, as it is both astringent and antiseptic. More technically, it is a naturally occurring chemical compound called potassium aluminium sulfate, which is a double sulfate of potassium and aluminium.
Which salt is alum?
hydrated double sulfate saltReason : An alum is a type of chemical compound, usually a hydrated double sulfate salt of aluminium with the general formula XAl(SO4)2. 12H2O, where X is a monovalent cation such as potassium or ammonium.
Is alum basic or acidic?
acidicAqueous solution of Potash alum is acidic in nature because it is a double salt of strong acid (H2SO4) & weak base(Al(OH)3) Q. The aqueous solution of potash alum [K2SO4.
Is alum poisonous to humans?
Alum's toxicity to humans Aluminum sulfate is fairly non-toxic, with acute and chronic oral LD50 both greater than 5,000mg/kg (5). However, alum can still cause irritation, burns, and respiratory issues. If inhaled, it may cause headaches, nausea, and respiratory irritations.
Is alum good for skin?
Yes, Alum, a naturally occurring ingredient is considered to be good for tightening the skin. It has an astringent property that causes the cells to shrink and removes excess oil from the skin. Thus, Alum is used in the form of gels or creams for skin care[8]. Alum is an effective remedy for skin problems.
How often can I put alum on canker sore?
Make a paste by mixing a small amount of alum powder and a drop of water. Apply it directly to the canker sore and leave it on for one minute. Then, rinse your mouth out. Do this once per day until the canker sore is gone.
How is alum obtained?
The specific process used to obtain the alum depends on the original mineral. When alum is obtained from alunite, the alunite is calcined. The resulting material is kept moist and exposed to air until it turns to a powder, which is lixiviated with sulfuric acid and hot water.
What is potassium alum?
Potassium Alum: Potassium alum is also known as potash alum or tawas. It is aluminum potassium sulfate. This is the type of alum that you find in the grocery store for pickling and in baking powder. It is also used in leather tanning, as a flocculant in water purification, as an ingredient in aftershave and as a treatment to fireproof textiles.
What is the compound used to grow lavender?
This deep violet compound is used in tanning and can be added to other alum to grow lavender or purple crystals. Selenate Alums: Selenate alums occur when selenium takes the place of sulfur so that instead of a sulfate you get a selenate, (SeO 42- ).
What is the formula for ammonium alum?
It is used in baking powder and as an acidulant in food. Ammonium Alum: Ammonium alum has the formula N 4 Al (SO 4) 2 ·12H 2 O. Ammonium alum is used for many of the same purposes as potassium alum and soda alum. Ammonium alum finds applications in tanning, dyeing textiles, making textiles flame retardant, in the manufacture ...
What are some uses for alum?
Uses of Alum. Alum has several household and industrial uses. Potassium alum is used most often, although ammonium alum, ferric alum, and soda alum may be used for many of the same purposes. purification of drinking water as a chemical flocculant. in styptic pencil to stop bleeding from minor cuts.
What is the formula for alum?
Usually, when you hear about alum it is in reference to potassium alum, which is the hydrated form of potassium aluminum sulfate and has the chemical formula KAl (SO 4) 2 ·12H 2 O. However, any of the compounds with the empirical formula AB (SO 4) 2 ·12H 2 O are considered to be an alum.
Is alum a crystal?
Sometimes alum is seen in its crystalline form, although it is most often sold as a powder. Potassium alum is a fine white powder that you can find sold with kitchen spices or pickling ingredients. It is also sold as a large crystal as a "deodorant rock" for underarm use.
What is the first step in alum synthesis?
The first step in this synthesis, which you will perform during Week 1, is to react metallic aluminum with a concentrated solution of potassium hydroxide (KOH) to form the potassium salt of the tetrahydroxoaluminate complex ion,
What will you do in week 2 of alum?
In Week 2 you will perform qualitative tests to demonstrat e the presence of K+, and sulfate ion (SO42-) in the alum. You will also perform a quantitative determination to determine the percent water by mass in alum.
Is alum a hydrate or a hydrate?
Alum is a hydrate , which means that it is a compound that has water molecules trapped within the solid. Hydrates will release some, or all, of their “waters of hydration” upon heating. If the chemical reaction between Al and KOH does produce alum as a product, we would expect that heating the product should result in a decrease in the sample’s weight corresponding to the loss of 12 water molecules per formula unit of alum. Thus, if one knows the starting mass of alum, and the amount (mass, and therefore number of moles) of anhydrous alum remaining after all of the water has been driven off, one can calculate the amount of water that was present in the alum (by the Law of Conservation of Mass). A comparison of the experimentally determined waters of hydration and the number expected from the chemical formula can then be used as evidence for the formation of the desired product. The process by which the waters of hydration are driven off is described by the chemical equation shown below, where the “Δ” written above the arrow indicates that heat was applied to the reactant (s).
Can you use a filter paper to filter aluminum?
When the aluminum has completely dissolved (do not be concerned if the solution appears cloudy or contains black specks), gravity filter the reaction mixture into a 50-mL beaker through fluted filter paper (the instructor will demonstrate). Dispose of the used filter paper in the laboratory garbage. CAUTION! The filter paper will be wet with the corrosive KOH solution. So, wash your hands after handling the wet filter paper.
Is alum an ionic compound?
The second and third weeks of this exercise will be devoted to characterizing the alum. Alum is an ionic compound, which means its melting and boiling points are likely to be too high to be measured conveniently. Also, most spectroscopic methods would not yield useful information. Therefore, we will rely on chemical means to show that we did, in fact, form alum in our reaction. This procedure duplicates how chemists characterized chemical reactions until the late 20th century, and in some cases chemical means of characterization are still the only methods available.
Acid Alum
Acid alum can be used in municipal and industrial water treatment applications to remove turbidity, color, suspended solids and oil (emulsion breaking). USALCO ® Acid Alum is especially effective in water treatment applications where TOC (total organic carbon) removal is important.
Acid Alum Solution Specs
USALCO ® Acid Alum, Aluminum Sulfate acidized with 7% free acid, is a high quality, iron-free, clear solution that meets or exceeds the specifications of the American Water Works Association Standard B403-16. The solution also complies with the requirements of NSF/ANSI/CAN Standard 60 at a maximum dosage of 150 mg/L.
Benefit from Reduced Operating Costs
USALCO ® Aluminum Sulfate is widely used because it is a cost-effective, clear solution that mixes well in water treatment. It does not add color to the water treatment process. Another benefit of our acid alum product is a marked improvement in the settled water filterability as less sludge is typically produced.
How is alum made?
Alum can be created through one of several chemical processes. One is by adding aluminum hydroxide Al (OH) 3 to sulfuric acid H 2 SO 4. The chemical reaction is: Translated to plain english, adding 2 molecules of aluminum hydroxide to 3 molecules of sulfuric acid results in 1 molecule of aluminum sulfate and 6 molecules of water.
What is alum used for?
As we have learned from our posts about aluminum sulfate so far, alum is a chemical compound mainly used as a coagulating agent in the purification of drinking water and wastewater treatment plants , among several other applications. This has all been related to the final product, alum.
How many molecules of water are in aluminum sulfate?
The process creates hydrated aluminum sulfate per the following reactions: Adding 3 molecules of sulfuric acid to 2 molecules of aluminum hydroxide and 8 molecules of water results in 1 molecule of aluminum sulfate and 14 molecules of water. Adding 3 molecules of sulfuric acid to 1 molecule of hydrated aluminum and 8 molecules ...
What is bauxite made of?
It involves reacting liquid sulfuric acid with either bauxite ore containing aluminum hydroxide (Al (OH) 3) and hydrated aluminum (Al 2 O 3 ∙ 3H 2 O), or synthetic hydrated aluminum previously refined from bauxite. Bauxite ore is the main source of aluminum for the world and contains various aluminum minerals and two iron minerals). The process creates hydrated aluminum sulfate per the following reactions:
What is alum schist?
Alum schist, also known as alum slate or alum shale, an argillaceous sedimentary rock, often a variety of black shale, containing pyrite or marcasite. Decomposition of pyrite and marcasite by weathering forms sulfuric acid which reacts with aluminous minerals in the rock to form alum and other aluminum sulfates (2).
How long does alum sit in a scrubber?
The steam that is generated is processed through a scrubber and the batch is allowed to sit for 12 hours, then tested to ensure product specifications are met. A single batch contains approximately 20,000 gallons of liquid alum, enough for five truck loads.
What is the specific gravity of aluminium sulfate?
The mass is now systematically extracted with water, and a solution of aluminium sulfate of specific gravity 1.16 is prepared. This solution is allowed to stand for some time (so that any calcium sulfate and basic ferric sulfate may separate), and is then evaporated until ferrous sulfate crystallizes on cooling.
Why is aluminum used in cooking?
In addition to its use in cookware, aluminum is also used for food processing and storage because it is easily cleaned with steam, is resistant to fatty acids and is splinter-proof. The food industry uses food processing equipment such as vats and piping that are made of aluminum.
What are chromatic acids?
Chromic or organic acids may include acetic and oxalic acids. These products may also contain chelating agents, detergents and small amounts of water-miscible solvents. Acidic aqueous solutions are sometimes used to clean or remove oxide, rust or scale from aluminum. Advertisement.
How to remove dark stains from aluminum pan?
Michigan State University recommends making an aluminum acid formula by mixing 1 tbsp. of vinegar in 1 qt. of water and adding the liquid to the discolored aluminum pan. After boiling for 10 to 15 minutes, remove the pan from the heat, and discard the hot liquid.
How to clean aluminum pans with lime scale?
Scour the pan with a steel wool pad, wash in soapy water, rinse and allow the pan to dry. To remove lime scale with vinegar, add equal parts of vinegar and water to the pan and bring it to a boil. Lemon juice also cleans discoloration from aluminum. Another aluminum cleaner, cream of tartar or potassium hydrogen tartrate, ...
What is the best way to brighten aluminum?
Another aluminum cleaner, cream of tartar or potassium hydrogen tartrate, is useful for as an aluminum brightener. Soak the aluminum pot, pan or utensil in a quart of hot water mixed with 2 tbsp. cream of tartar.
What is aluminum foil used for?
Aluminum foil is used to prevent partially prepared or frozen foods from going rancid or spoiling. Aluminum is widely used in medicine such as antacids and elsewhere in the pharmaceutical industry. Elsewhere in food processing, aluminum is used in everything from sugar, beer, flour, gelatin, meats, frozen foods and dairy products, ...
Is aluminum safe to clean?
It is an ideal metal for pots and pans because it heats fast and evenly and is lightweight. Aluminum is easy to clean and brighten with a mild aluminum acid wash using ingredients often found in pantries or kitchen cupboards. Other acidic aqueous cleaning solutions are also effective, but they are much harsher and not as safe.
Which acid doesn't react with Al2O3?
As far as I know, there is only one acid that doesn’t react with Al, it is concentrated nitric acid (HNO3), the upper layer of Al gets oxidized to form Al2O3 which renders the metal passive for further reaction with acid, also Al2O3 is a very stable metal oxide
What is the chemical formula for aluminum oxide?
Aluminium. Aluminium oxide ( British English) or aluminum oxide ( American English) is a chemical compound of aluminium and oxygen with the chemical formula Al 2 O 3. Aluminium oxide - Wikipedia. Aluminium oxide is responsible for the resistance of metallic aluminium to weathering.
How does aluminum oxidize?
Aluminum will corrode or oxidize just as all other metals in varying degrees when in contact with moisture or water. But Aluminum has one different characteristic is that it can be artificially oxidized on the surface in a electrochemical process called anodizing. Once anodized (oxidized) and sealed the surface will resist further corrosion for a long period of time 10–30 years. That is why it is found in home windows and curtain walls of tall and large buildings. Anodizing is not a paint but a process which converts a very thin layer the surface from aluminum to aluminum oxide. The tough hard surface will resist corrosion especially weathering, in fact aluminum oxide in powdered form is used on sandpaper as an abrasive.
Why is aluminum corrosion resistant?
Corrosion resistance can be excellent because a thin surface layer of aluminium oxide forms when the bare metal is exposed to air, effectively preventing further oxidation , [22] in a process termed passivation. The strongest aluminium alloys are less corrosion resistant due to galvanic reactions with alloyed copper.
What is the layer of metal that protects the metal from oxidation?
Metallic aluminium is very reactive with atmospheric oxygen, and a thin passivation layer of aluminium oxide (4 nm thickness) forms on any exposed aluminium surface. [8] . This layer protects the metal from further oxidation.
How to remove oxide layer from aluminium?
When you put a piece of aluminium in acid to remove the oxide layer. Take it out and put in acetone and then ether. You will for a very short time see pure Aluminium. It is highly electropositive and will react with oxygen very quickly to form Aluminium oxide. Al2O3.
What is the strongest metal?
The strongest aluminium alloy. Aluminium or aluminum is a chemical element with symbol Al and atomic number 13. It is a silvery-white, soft, nonmagnetic, ductile metal in the boron group. Aluminium metal is so chemically reactive that native specimens are rare and limited to extreme reducing environments. Instead, it is found combined in ...
What are the acids in soda?
Image Credit: scanrail/iStock/Getty Images. Most soft drinks contain one or more of three common acids: citric acid, carbonic acid and phosphoric acid. Soft drinks with specific flavor ingredients may also contain other less common acids. The level of acidity in sodas can be quite high, and many dentists deplore its long-term effect on your teeth.
Where does citric acid come from?
Citric acid comes from citrus products and any soft drink with citrus flavoring will contain it. The chemical formula is H (3)C (6)H (5)O (7). Carbonic acid forms from dissolved carbon dioxide, which is present in almost all soft drinks.
What are the three acids in soft drinks?
The three most common acids in soft drinks are citric, carbonic and phosphoric acids . Citric acid comes from citrus products and any soft drink with citrus flavoring will contain it. The chemical formula is H (3)C (6)H (5)O (7). Carbonic acid forms from dissolved carbon dioxide, which is present in almost all soft drinks. The chemical formula is simply a combination of water's H (2)O and the CO (2) of carbon dioxide, which makes H (2)CO (3). Soft-drink makers often add phosphoric acid to colas to produce a sharp flavor and to slow the growth of mold and bacteria. Most of the acidity in colas comes from phosphoric acid, whose chemical formula is H (3)PO (4).
Why do soft drinks have phosphoric acid?
Soft-drink makers often add phosphoric acid to colas to produce a sharp flavor and to slow the growth of mold and bacteria. Most of the acidity in colas comes from phosphoric acid, whose chemical formula is H (3)PO (4). Advertisement.
Does acidity affect teeth?
Effects. The effects of acidity in soft drinks may be more damaging to teeth than the sugar they contain. Industrial users of phosphoric acid apply it to steel parts in order to strip them to bare metal before being painted.
Does soda have malic acid?
Others. Some sodas contain acids in addition to the three most common ones. Malic acid is present in some fruits and their juices and makers of noncarbonated drinks sometimes add quantities of it for flavor and as a preservative. For similar reasons, some fruit drinks contain fumaric acid as an additive.