
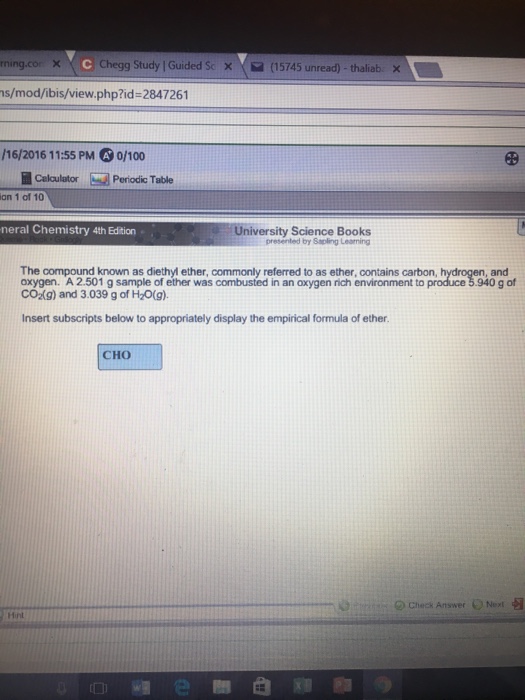
What is the general formula for ether?
Preparation Of Ethers
- Dehydration of Alcohols. Alcohol undergoes dehydration in the presence of protic acids (sulphuric acid, phosphoric acid) to produce alkenes and ethers under different conditions.
- Williamson Synthesis. This is an important method for the preparation of symmetrical and asymmetrical ethers in laboratories.
- Alkyl Halide With Dry Silver Oxide. ...
What is the molecular formula of ether?
Ethers are a class of organic compounds that contain an ether group —an oxygen atom connected to two alkyl or aryl groups. They have the general formula R–O–R′, where R and R′ represent the alkyl or aryl groups.
What type of bond does ether have?
Ether molecules have no hydrogen atom on the oxygen atom (that is, no OH group). … Ether molecules do have an oxygen atom, however, and engage in hydrogen bonding with water molecules. Consequently, an ether has about the same solubility in water as the alcohol that is isomeric with it.
What are the chemical properties of ether?
Chemical properties of ethers. 1. Ethers are quite stable chemical compounds which do not react with bases, active metals, dilute acids, oxidising agents and reducing agents. Generally, they are of low chemical reactivity, but they are more reactive than alkanes (epoxides, ketals, and acetals are unrepresentative classes of ethers and are ...
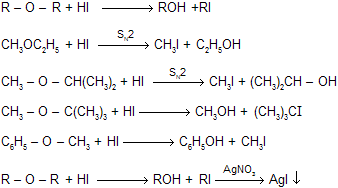
Which is an example of an ether?
Thus, in IUPAC systems, ethers are alkoxy alkanes. Example: ethoxyethane (diethyl ether), methoxy ethane (methyl ethyl ether), 2-methoxy-2-methylpropane (methyl tert-butyl ether), phenoxy benzene (diphenyl ether).
Is CH3OCH3 an ether?
An ether in which the oxygen atom is connected to two methyl groups. This entity has been manually annotated by the ChEBI Team. Dimethyl ether (DME; also known as methoxymethane) is the organic compound with the formula CH3OCH3, simplified to C2H6O.
Which molecule shown is an ether?
Which molecule shown is an ether? ethanol.
How do you know if a compound is an ether?
An ether is an organic compound in which two hydrocarbon groups are bonded to the same atom of oxygen. An ether is represented by the general formula R−O−R'. The R' in the formula means that the hydrocarbon group can be the same as R or it can be different.
Is diethyl an ether?
ethyl ether, also called diethyl ether, well-known anesthetic, commonly called simply ether, an organic compound belonging to a large group of compounds called ethers; its molecular structure consists of two ethyl groups linked through an oxygen atom, as in C2H5OC2H5.
What are ethers in chemistry?
ether, any of a class of organic compounds characterized by an oxygen atom bonded to two alkyl or aryl groups. Ethers are similar in structure to alcohols, and both ethers and alcohols are similar in structure to water.
Is ethanol an ether?
Ethanol is an alcohol having the chemical formula C2H5OH. Dimethyl ether is an ether compound having the chemical formula C2H6O. Ethanol is an alcohol. Dimethyl ether is an ether.
How do you name ether compounds?
Common nomenclature of ethers follows the rule of naming different alkyl/aryl groups attached to the oxygen atom on either side in alphabetical order and finally adding the word ether to it. For example, CH3OC6H5 is named as Methyl phenyl ether.
How do you name an ether group?
Simple ethers are given common names in which the alkyl groups bonded to the oxygen are named in alphabetical order followed by the word "ether". The top left example shows the common name in blue under the IUPAC name. Many simple ethers are symmetrical, in that the two alkyl substituents are the same.
What is the most common ether?
Important ethersEthylene oxideA cyclic ether. Also the simplest epoxide.Tetrahydrofuran (THF)A cyclic ether, one of the most polar simple ethers that is used as a solvent.Anisole (methoxybenzene)An aryl ether and a major constituent of the essential oil of anise seed.8 more rows
What is ether in the 5 elements?
According to ancient and medieval science, aether (/ˈiːθər/, alternative spellings include æther, aither, and ether), also known as the fifth element or quintessence, is the material that fills the region of the universe beyond the terrestrial sphere.
Is acetone an ether?
Acetone, propanone or dimethyl ketone, is an organic compound with the formula (CH3)2CO. It is the simplest and smallest ketone. It is a colourless, highly volatile and flammable liquid with a characteristic pungent odour....Acetone.NamesSolubilityMiscible in benzene, diethyl ether, methanol, chloroform, ethanollog P−0.1673 more rows
What functional group is CH3OCH3?
Functional GroupCondensed FormulaNamealcoholCH3CH2OHethyl alcoholetherH3COCH3dimethyl etheretherCH3CH2OCH2CH3diethyl ethercarboxylic acidHCOOHformic acid7 more rows
What is an ether functional group?
The ether functional group consists of an oxygen atom that forms single bonds with two carbon atoms. Figure 4.4. 4: Ethers. Ethers are good solvents for other organic compounds because of their low reactivity. They readily dissolve nonpolar molecules.
What type of bond is CH3OCH3?
If we look at organic compounds like CH3OCH3, we have covalent bonds formed between carbon and hydrogen and also carbon and oxygen.
What is the functional isomer of CH3OCH3?
The correct answer is Option 1) i.e Ethanol.
What is an ether?
Full Article. Ether, any of a class of organic compounds characterized by an oxygen atom bonded to two alkyl or aryl groups. Ethers are similar in structure to alcohols, and both ethers and alcohols are similar in structure to water. In an alcohol one hydrogen atom of a water molecule is replaced by an alkyl group, ...
What are the unique properties of ethers?
The unique properties of ethers (i.e., that they are strongly polar, with nonbonding electron pairs but no hydroxyl group) enhance the formation and use of many reagents. For example, Grignard reagents cannot form unless an ether is present to share its lone pair of electrons with the magnesium atom.
What is the name of the alkyl group bonded to oxygen?
Common names of ethers simply give the names of the two alkyl groups bonded to oxygen and add the word ether. The current practice is to list the alkyl groups in alphabetical order ( t -butyl methyl ether), but older names often list the alkyl groups in increasing order of size (methyl t -butyl ether). If just one alkyl group is described in the name, it implies two identical groups, as in ethyl ether for diethyl ether.
What is the difference between ether and alcohol?
At room temperature, ethers are pleasant-smelling colourless liquids. Relative to alcohols, ethers are generally less dense , are less soluble in water, and have lower boiling points.
How are crown ethers named?
They are named by using the parent name crown preceded by a number describing the size of the ring and followed by the number of oxygen atoms in the ring. In the crown-ether complex, the metal ion fits into the cavity of the crown ether and is solvated by the oxygen atoms. The exterior of the complex is nonpolar, ...
What are ethers used for?
Vapours of certain ethers are used as insecticides, miticides, and fumigants for soil. Ethers are also important in medicine and pharmacology, especially for use as anesthetics.
Is ether a solvent?
Ethyl ether is an excellent solvent for extractions and for a wide variety of chemical reactions. It is also used as a volatile starting fluid for diesel engines and gasoline engines in cold weather. Dimethyl ether is used as a spray propellant and refrigerant.
What Is Ether?
- “A molecule with an oxygen atom linked to two alkyl groups is known as ether.” Ethers are colourless liquids with a sweet odour that are liquid at room temperature. Diethyl etheris the most common form of ether, which is highly flammable and was one of the first anaesthetics used in surgery.
Table of Contents
Preparation of Ethers
- Ether is a functional group generated when an alkoxy (R–O) or aryloxy (Ar–O) group replaces a hydrogen atom in a hydrocarbon. They are denoted by the letters R–O–R′. The word ‘Ether’ comes from the Latin word ‘Aether,’ which literally means “pure high, light air.” Ethers are an extremely light, volatile, and flammable liquid that may be used in a variety of applications. This type of che…
Physical and Chemical Properties of Ether
- Asymmetrical ether (when two identical groups are connected to the oxygen atom) and symmetrical ether (when two different groups are attached to the oxygen atom) are the two types of ethers (when two different groups are attached to the oxygen atom). Ethers have a diverse set of physical and chemical characteristics.
Nomenclature of Ethers
- The word ether is added to the names of the two alkyl groups linked to oxygen in common ether names. Currently, the alkyl groups are listed in alphabetical order (t-butyl methyl ether), although older names frequently mention the alkyl groups in increasing size order (methyl t-butyl ether). If the term just mentions one alkyl group, it implies two similar groups, as in ethyl ether for diethyl …
Uses of Ethers
- Ethers are chemical substances that are extremely flammable. They’re employed in medicine, laboratories, our daily lives, and a variety of other applications. Let’s take a closer look at how they’r...
- They’re usually used in surgeries as an anaesthetic. Because ethers are volatile liquids, they release vapours that patients inhale or are given intravenously. They cause individuals to los…
- Ethers are chemical substances that are extremely flammable. They’re employed in medicine, laboratories, our daily lives, and a variety of other applications. Let’s take a closer look at how they’r...
- They’re usually used in surgeries as an anaesthetic. Because ethers are volatile liquids, they release vapours that patients inhale or are given intravenously. They cause individuals to lose consci...
- Antiseptics were also made from ethers. Ether was used to sanitise patients’ wounds and save them from dangerous illnesses during World War II. They are still used as antiseptics.
- Because of their olfactory and organoleptic qualities, ethers are frequently employed in the perfumery and scent business. Fruit-based meals and beverages, sweets, fruit jams, yoghurts…