What mesoderm gives rise to the heart?
Splanchnic mesodermSplanchnic mesoderm: The ventral layer of the lateral mesoderm, which lays adjacent to the endoderm. This tissue layer forms when lateral mesoderm separates into distinct dorsal and ventral layers: the somatic and splanchnic mesoderm. It is the splanchnic mesoderm that will give rise to the heart.
What tissue is formed by endocardial tubes?
For the heart to grow and continue developing, a mesenchymal tissue called cardiac jelly forms around the endocardial tube. This allows for the heart to separate from the foregut.
What layer of the embryo gives rise to the embryonic heart tubes?
The primitive heart tube is composed of three layers, which are analogous to the adult human heart. The endocardium forms the endothelial lining of the embryonic heart. The myocardium forms the muscular bulk of the embryonic heart while the visceral pericardium forms the embryonic heart tube's external surface.
Where do the endocardial tubes come from?
The endocardial heart tubes derive from the visceral mesoderm and initially are formed by a confluence of angioblastic blood vessels on either side of the embryonic midline. The endocardial tubes have an intimate proximity to the foregut or pharyngeal endoderm.
What develops from the Hypoblast?
The hypoblast gives rise to the yolk sac, which in turn gives rise to the chorion. The hypoblast is a layer of cells in fish and amniote embryos.
What do the 3 germ layers give rise to?
The ectoderm gives rise to the nervous system and the epidermal skin cells, the mesoderm gives rise to the muscle cells and connective tissue in the body, and the endoderm gives rise to the digestive system and other internal organs. Organogenesis is the formation of organs from the germ layers.
How many endocardial tubes give rise to the embryonic heart?
Two endocardial heart tubesTwo endocardial heart tubes arise from cardiogenic mesoderm. As lateral folding occurs, these fuse to form the primitive heart tube, which develops into the endocardium.
What layer gives rise to neural tube?
ectodermThe ectoderm is also sub-specialized to form the (2) neural ectoderm, which gives rise to the neural tube and neural crest, which subsequently give rise to the brain, spinal cord, and peripheral nerves. The endoderm gives rise to the lining of the gastrointestinal and respiratory systems.
What is endocardial tissue?
The endocardium is the innermost layer of tissue that lines the chambers of the heart. Its cells are embryologically and biologically similar to the endothelial cells that line blood vessels. The endocardium also provides protection to the valves and heart chambers.
What is produced when the endocardial tubes fuse together?
As the cardiogenic cords develop, a lumen rapidly develops within them. At this point, they are referred to as endocardial tubes. The two tubes migrate together and fuse to form a single primitive heart tube.
What two types of tissue make up the endocardium?
The endocardium has two layers. The inner layer lines the heart chambers and is made of endothelial cells. Superiorly, is the second layer: a subendocardial connective tissue which is continuous with the connective tissue of the myocardium.
What do the endocardial cushions form?
The endocardial cushions are two thicker areas that develop into the walls (septum) that divide the four chambers of the heart. They also form the mitral and tricuspid valves. These are the valves that separate the atria (top collecting chambers) from the ventricles (bottom pumping chambers).
What are the blood islands in YS?
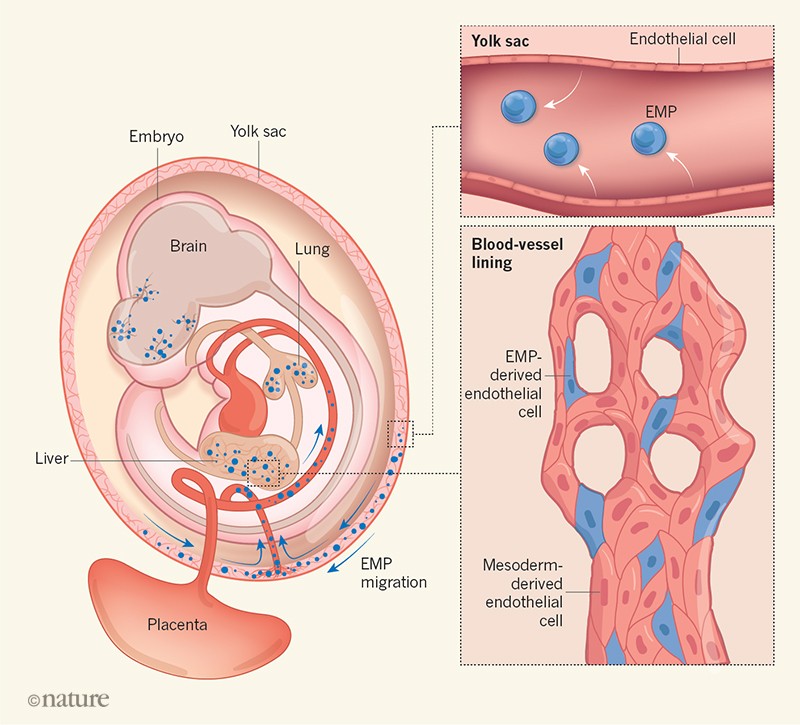
Shortly after gastrulation, mesodermal cells in the YS form clusters of immature cells (blood islands/belt) (Ferkowicz and Yoder, 2005) that differentiate into erythroid and surrounding endothelial cells. This led to postulating a common bipotent endothelial-hematopoietic precursor, the hemangioblast. Sharing gene expression programmes between endothelial and blood cells supported this idea. However, it took a long time before the hemangioblast was experimentally identified, initially in differentiating embryonic stem (ES) cells ( Huber et al., 2004 ). The relationship between endothelial and hematopoietic cells appears to be more complex than initially thought and the concept has undergone significant elaboration. Hemangioblasts localize mainly to the primitive streak, which on arrival in the YS blood islands/belt segregate into endothelial and hematopoietic lineages ( Huber et al., 2004 ). Furthermore, YS blood and endothelial lineages originate mostly from independent epiblast populations, specified before gastrulation ( Padron-Barthe et al., 2014 ). Hemangioblast progression later involves an intermediate precursor, the hematogenic (or ‘hemogenic’) endothelial (HE) cell, which subsequently differentiates into blood ( Eilken et al., 2009; Lancrin et al., 2009; Nishikawa et al., 1998 ). Hemangioblastic populations have also been identified in anamniotic vertebrates. However, only a small fraction of embryonic endothelial cells are linked to blood production.
What are the two markers of the mesenchyme?
NC-derived and mesodermal cells both make major contributions to the craniofacial mesenchyme. Two permanent cell-lineage markers have transformed our understanding of these tissue lineages. Wnt1 is transiently expressed in NC cells, as they leave the neural tube, and in some parts of the cranial neuroepithelium. Mesp1 is transiently expressed in the nascent mesoderm, that is, mesoderm cells as they leave the primitive streak. The expression of both genes has been made permanent by constructing Wnt1-Cre or Mesp1-Cre transgenic mice, which, when mated with mice carrying the Rosa26 reporter gene in all cells, produce embryos carrying both constructs ( Wnt1-Cre/R26R or Msp1-Cre/R26R ); these embryos, as either whole (at early stages) or sectioned material, can be stained with X-gal to stain blue all cells that have ever expressed Wnt1 or Mesp2 ( Jiang et al., 2000; Yoshida et al., 2008 ). For Wnt1-Cre/R26R embryos, all neural crest-derived tissues are stained, as well as parts of the neural tube (midbrain plus parts of the diencephalon and upper hindbrain) from which some of the cells are derived. For Mesp1/R26R embryos, all mesoderm-derived tissues are X-gal positive, that is, stained blue. These two transgenic constructs have clarified the relative contributions of neural crest cells and the mesoderm to the developing head, pharynx, heart, and great vessels; analysis of sequential stages of mouse development has also revealed information on the relative movements of tissues of these respective origins as development progresses. Figure 16.1D shows an X-gal-stained Wnt1-Cre/R26R E9.5 embryo when neural crest cell migration into the frontonasal and pharyngeal regions is complete. The caudal boundary of the frontonasal neural crest cell domain is well defined. Further migration of neural crest cells from the lower pharyngeal arches into the heart will form the septum dividing the outflow tract into aortic and pulmonary trunks ( Jiang et al., 2000 ).
How does microscopy reveal fibronectin?
Electron microscopy reveals a potential directional cue in the micro-structure of fibronectin in both living embryos and ‘blotted’ matrices in culture. Its molecules are aligned to produce fibres that lie along the direction of normal cell migration. These fibres are the result of activity in the cells that secrete the fibronectin and seem to rely on mechanical force exerted by the actin/myosin cytoskeleton acting via integrins. Interference either with fibronectin-binding integrins using the competitor peptide sequence, GRGDSP, or interference with the actin cytoskeleton using cytochalasin B, blocks the formation of fibronectin fibres. The matrix laid down by a blastocoel roof that is cultured in the presence of either of these drugs is rich in fibronectin but has no aligned fibres. Significantly, mesodermal cells plated on to it still adhere normally and still move, but their movement is now undirected and random. 49 Clearly, the alignment of fibres is an important directional cue for mesodermal cells. The mechanism by which cells can be guided by aligned fibres has not been investigated in detail. Simple arguments based on adhesion, being more stable on fibres than between them, may explain a preference for following fibres but cannot in itself explain a preference for going one way rather than the other (unless the fibres converge, in which case the overall density of adhesive fibres will be highest where they are coming together—adhesion measurements in the blastocoel roof suggest that adhesion gradients are not present in amphibian gastrulation).
What are the two waves of mesodermal cell migration?
Two waves of mesodermal cell migration can be distinguished during heart development: the first mobilizes cells from the primitive streak toward the anterior and anteriorlateral regions of the gastrula where cardiac induction occurs; the second directs convergence of cardiac progenitors to the ventral midline during heart tube formation. Involvement of the FGF signaling system in cell migration within mesoderm is a recurrent theme in diverse systems (Blelloch, et al., 1999; Gisselbrecht, et al., 1996; Zelzer and Shilo, 2000 ). Genetic experiments in flies show a role for the FGF receptor gene. heartless, acting through Ras, in dorsal migration of nascent mesoderm ( Beiman, et al., 1996; Gisselbrecht, et al., 1996 ). Mutant mesoderm fails to contact the dorsal ectodermal zone of dpp expression, an essential requirement for cardiac induction ( Frasch, 1995 ). A number of mouse mutations, including knockouts in Fgf8 and the bHLH Mesp genes, form mesoderm that is blocked or delayed in its migration away from the streak ( Kitajima, et al., 2000; Sun, et al., 1999 ).
What is the difference between the somatic and splanchnic layers?
This process potentially involves polar expression of a Na − K − -ATPase ( Linask and Lash, 1998 ). The splanchnic layer forms the cuboidal epithelium that gives rise to myocardial and endocardial cells, while the somatic layer gives rise to pericardial mesoderm and ceases to express cardiac markers. The timing and direction of movement of cardiac progenitors toward the midline ( DeHaan, 1963) depends on the graded distribution of fibronectin in extracellular matrix, deposited at the mesodermal/endodermal interface. Inhibition of the interaction between fibronectin and its integrin receptor in the chick using blocking antibodies or RGD peptide ( Linask and Lash, 1998 ), or in mice using gene targeting ( George, et al., 1997 ), leads to varying degrees of cardia bifida. In a subset of mice mutant for the fibronectin gene (Fn1) on a 129Sv genetic background, cardiac cells reach the anterior and express myosins normally, but never move from the crescent ( George, et al., 1997 ). In chick embryos, cavitation, epithelialization, and even differentiation of cardiac progenitors also depends on the calcium-dependent adhesion molecule N-cadherin ( Linask, et al., 1997; Linask and Lash, 1998 ). In mice mutant for the N-cadherin gene (Cdh2), cavitation and cardiac differentiation proceed normally, but the adhesive integrity of the myocardial epithelium is not maintained and the heart tube essentially disintegrates ( Radice, et al., 1997 ).
Where do HSCs bud?
It is broadly accepted that HSCs bud from the endothelium of the dorsal aorta which culminates in intra-aortic cluster formation ( Figure 3 ). Similar cell clusters are formed in large extraembryonic vessels. The first HSCs in the mouse are labeled by the Sca1 reporter and localize to the aortic endothelium ( de Bruijn et al., 2002 ). They also inherit an endothelial marker VE-cadherin which gradually disappears during their further development ( North et al., 2002; Taoudi et al., 2005 ). In agreement, cell fate experiments using Cre-mediated genetic tagging of embryonic endothelial cells results in labeling of the adult hematopoietic system ( Chen et al., 2009; Zovein et al., 2008 ). Committed progenitors and HSCs were reported to arise from distinct subsets of HE ( Chen et al., 2011 ), correlated at least partly with YS and AGM region localizations. While in mammals, HSCs can be distinguished functionally, in other vertebrate models, transient multipotent progenitors (MPPs) and HSCs are difficult to discriminate and both populations are sometimes cumulatively termed hematopoietic stem and progenitor cells (HSPCs). Formation of HSPCs from the aortic endothelium has been clearly visualized in live zebrafish ( Bertrand et al., 2010; Kissa and Herbomel, 2010; Lam et al., 2010 ). During this process, called the endothelial-hematopoietic transition (EHT), endothelial cells round up and dive under the dorsal aorta to enter circulation through the cardinal vein. In mice, HSPCs are thought to enter aortic circulation by budding directly into the lumen ( Boisset et al., 2010; Smith and Glomski, 1982 ).
How long does endothelial cell turnover take?
In different vascular beds endothelial cell turnover has been estimated to vary from 2 months to 3 years. 14 The disposal of damaged endothelial cells occurs as a result of patrolling function of non-invading subset of Ly6C low monocytes. Intravital microscopy studies demonstrated that these monocytes crawl on the luminal surface of glomerular and peritubular capillaries and scavenge microparticles. 15 In response to nucleic acid “danger” signaling, Ly6C low monocytes exhibit prolonged dwell times in glomerular and peritubular capillaries, more complex patrolling routes, attachment to damaged endothelial cells, and recruitment of neutrophils, which induce focal necrosis and disposal of cellular debris. Cells that escape this in situ disposal mechanism detach from their basement membranes and appear in the circulation.
What are the paired regions of the heart?
Endocardial tubes. The endocardial tubes are paired regions in the embryo that appear in its ventral pole by the middle of the third week of gestation and consist of precursor cells for the development of the embryonic heart. The endocardial heart tubes derive from the visceral mesoderm and initially are formed by a confluence ...
Where do endocardial tubes come from?
The endocardial heart tubes derive from the visceral mesoderm and initially are formed by a confluence of angioblastic blood vessels on either side of the embryonic midline. The endocardial tubes have an intimate proximity to the foregut or pharyngeal endoderm.
What is the first step in cardiac development?
4.1; Kodo and Yamagishi, 2011; Srivastava, 2006; Vincent and Buckingham, 2010). Formation of the precardiac mesoderm is considered to be the first step in cardiac development. In this process, a specific subset of mesodermal cells, generated during embryonic gastrulation, migrate anterolaterally to form a crescent shape on approximately embryonic day (E)7.5 in the mouse embryo, corresponding roughly to week 2 of human gestation. By E8.0 in mice, or week 3 in humans, a pair of endocardial tubes appears in the cardiac crescent and fuses along the ventral midline to form a primitive heart tube, consisting of an interior layer of endocardial cells and an exterior layer of myocardial cells, separated by the extracellular matrix, or “cardiac jelly,” that mediates reciprocal signaling between the two layers. The crescent-shaped pool of cardiogenic progenitor cells is referred to as the “first heart field”.
How does the foregut form?
As the foregut forms, the left and right precardiac cells are moved into the midline and each begins to condense into an endocardial tube. The developing foregut loses contact with the yolk sac and the two tubes of the heart fuse in the midline to form a single tube, starting anteriorly and progressing posteriorly. The process can best be seen by studying a sequence of sections from progressively older embryos ( Text-Figure 35). Fusion of the two endocardial tubes begins, anterior and ventral to the anterior intestinal portal, at about stage 8 (Coffin and Poole, 1988). At the same time, the two sides of the foregut join medially, forming the characteristically V-shaped pharynx (Plate 64 ). The left and right sheets of splanchnic mesoderm ventral to the pharynx combine to form the dorsal and ventral mesocardia ( Text-Figure 35; Plate 32 ). The latter is so transient, however, as to be seldom visible, rupturing at about stage 10. The simple tube is initially suspended into the coelom beneath the pharynx by the dorsal mesocardium, but this structure is also transient, persisting only until about stages 12–13. As the lateral body folds form (see p. 45 ), the coelom surrounding the heart becomes isolated from the more posterior coelom and is henceforth known as the pericardial coelom or cavity ( Plates 35, 81Plate 35Plate 81 ).
What are the layers of the heart?
Once the heart has formed into a simple tube ( Plates 32, 33Plate 32Plate 33 ), two distinct layers are visible ( Plate 85 ): the thin inner layer, the endocardium, and the thicker outer layer, commonly known as the epimyocardium. Subsequently, the heart becomes three-layered, these layers being the endocardium, the myocardium and the thin enveloping epicardium. The traditional view has been that the precardiac mesoderm gives rise to all three layers of the heart, but several authors using transmission or scanning electron microscopy (e.g. Shimade and Ho, 1980 ), or experimental techniques ( Männer, 1993 ), have concluded that the epicardium arises from the pericardium (i.e. from the splanchnic mesoderm of the pericardial wall), and the traditional view has now been modified. The relevant part of the pericardial wall, now known as the pro-epicardial serosa ( Plate 36 ), surrounds the entire region near the sinus venosus, and contributes primarily extracardiac cells to the developing heart ( Männer et al., 2001 ). As the pro-epicardial serosa starts to develop at stages 13–14, bleb-like protrusions form at the ventral wall of the left and right horns of the sinus venosus. Cells from the pro-epicardial serosa are then transferred across the pericardial cavity to the surface of the developing ventricles, either as free-floating aggregates or by the formation of bridges across the lumen. The primordial epicardium then spreads as a continuous epithelial sheet over the myocardium ( Männer et al. 2001 ). The pro-epicardial serosa also gives rise to the coronary blood vessels as well as to a small number of endocardial cells ( Männer, 2001 ). For more information on the origin of the pro-epicardial serosa in the quail, see Virágh et al. (1993) or Männer et al. (2001).
What is the septum primum?
In the atria, the septum primum, a thin membrane, grows from the dorsal roof of the common atrium toward the AV canal. Its forward edge creates an opening between the septum and the endocardial cushions, the ostium primum. As the ostium primum is being closed by fusion of the septum primum and the fused endocardial cushion tissue, perforations form in the septum primum by programmed cell death. The coalescence of these holes forms the ostium secundum. A septum secundum then forms to the right of the septum primum, growing from the roof of the atrium, covering the ostium secundum. However, the septum secundum forms an incomplete division of the atria. The opening between the two septa is the foramen ovale. Defects in closure of the ostium primum or ostium secundum result in atrial septal defects. Ostium secundum defects are relatively common and can be due to defects in formation of the septum primum or the septum secundum.
What is the AV canal?
The atrioventricular (AV) canal connects the common atrium and the early embryonic ventricle. The AV canal is divided by endocardial cushions that arise at the superior and inferior borders of the canal in the fourth week. The cushions also form the AV valves. The endocardial cushions grow across the AV canal and fuse, creating left and right AV canals. Regions of the endocardial cushions then fuse with both atrial and ventricular septa. If both atrial and ventricular septa and the medial AV valve leaflets are incomplete due to failure of fusion of the cushions, a persistent AV canal can result, as may sometimes be seen in association with Down syndrome.
How does cell death affect development?
Programmed cell death is crucial to development; it eliminates vestigial anatomy, manages the number of cells in the developing tissue, and aids in the reorganization of structures ( Vaux and Korsmeyer, 1999). Remarkably, from embryonic day 20 to embryonic day 35, the human heart develops from two endocardial tubes into a four-chambered heart (Okamoto and Satow, 1975 ). During this transformation, large amounts of cell migration, proliferation, and death systematically occur. Cell death was first observed in the developing chicken embryonic heart in the late 1960s by transmission electron microscopy (EM) ( Manasek, 1969 ). Extensive characterization of the areas of cell death deemed “zones of cell death” have been established in the chick embryo. Subsequently, research has since extensively recognized 32 zones of death from day 1 to day 10 in the developing chick heart, 21 zones of death between embryonic day 13–17 in rats, and 16 zones in humans ( Van Den Hoff et al., 2000 ). Each zone of death has a specific spatial and temporal location that plays a critical role in cardiac morphogenesis ( Van Den Hoff et al., 2000 ). For example, this cell death aids in fusion of arterial septum with the interventricular septum ( Van Den Hoff et al., 2000 ). By day 10 in the chick, which is equivalent to day 50 in the human, the zones of cell death are no longer detectable and programmed cell death in the heart is complete ( Van Den Hoff et al., 2000 ).
How to prevent posterior extension of the foregut?
The posterior extension of the foregut can be prevented experimentally by cutting through the anterior intestinal portal ( DeHaan, 1959) or by inserting a mechanical obstacle such as a lump of thick albumen. In either case the result is that the two sides fail to fuse and diplocardia results. Each side then forms a small but perfect heart which begins to beat independently and at the appropriate time.
