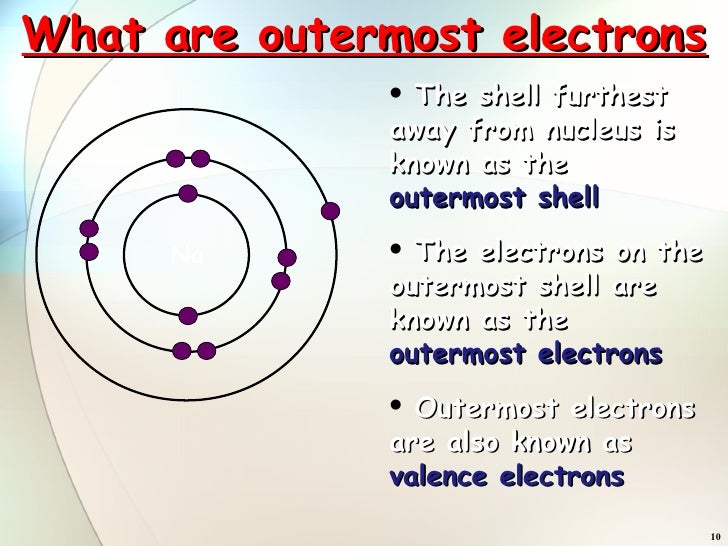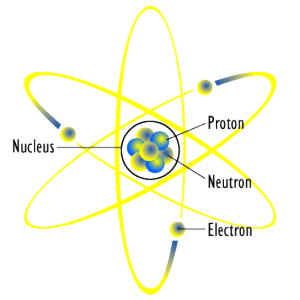
What part of the atom carries a positive charge?
lbw00. Terms in this set (15) the part of the atom that carries no electric charge. nucleus. the part of the atom that carries a positive charge. proton. the part of the atom that carries an electric charge. electron. the number of electrons that can be held in the first orbit.
What are parts of the atom have a positive charge?
The three main parts of a atom is protons,neutrons,and electrons. Protons - have a positive charge, located in the nucleus, Protons and neutrons have nearly the same mass while electrons are much less massive. Neutrons- Have a negative charge, located in the nucleus.
What are negatively charged particles forming part of an atom?
Negatively Charged Particles – Electrons within an atom is known as the negative charge particles. The negatively charged particles have more electrons as compared to positive charge particles. The atom becomes negatively charged when it gains the electrons from another atom. The mass of negative charge particle is 9.11 X 10 -31 kg.
How does an atom become positive or negatively charged?
When one or more electrons is stripped away from an atom, it becomes positively charged. Some atoms can attract additional electrons so they become negatively charged. Atoms which are not electrically neutral are called ions. One can collect electric charge by transferring electrons. Can an atom only be negatively charged?
What happens when a positively charged particle moves close to a neutral atom?
What are the subatomic particles?
Which type of charge is the same on a proton and electron?
Where are protons and neutrons in an atom?
What are the two types of electric charges?
Which part of an atom is positively charged?
Which subatomic particles are present in the nucleus?
See 2 more
About this website
Which part of the atom is positively charged and which is negatively charged?
There are three subatomic particles: protons, neutrons and electrons. Two of the subatomic particles have electrical charges: protons have a positive charge while electrons have a negative charge.
Which part of an atoms is positively charged?
The nucleusThe nucleus has an overall positive charge as it contains the protons. Every atom has no overall charge (neutral). This is because they contain equal numbers of positive protons and negative electrons. These opposite charges cancel each other out making the atom neutral.
What part of the atom has a negative charge?
electronsThe electrons have a negative electrical charge. An atom usually contains an equal number of positively charged protons and negatively charged electrons. This makes the atom itself electrically neutral. The electrons exist at different energy levels, called shells, around the nucleus.
Are neutrons positive or negative?
electrically neutralMiller, a UW physics professor, has found that the neutron has a negative charge both in its inner core and its outer edge, with a positive charge sandwiched in between to make the particle electrically neutral.
Which of the following has a negative charge?
Electrons (negative charge)
What are the 3 parts of an atom and their charges?
Structure: Our current model of the atom can be broken down into three constituents parts – protons, neutron, and electrons. Each of these parts has an associated charge, with protons carrying a positive charge, electrons having a negative charge, and neutrons possessing no net charge.
Why nucleus is positively charged?
A nucleus of an atom has protons and neutrons. we know that a proton has a charge of + 1 , while a neutron has no charge, or 0. Therefore, the nucleus of an atom will always have a positive charge.
What is positive charge and negative charge?
There are two types of electric charges and they are known as positive charge and negative charge. When an object has more electrons than protons then the object is said to be negatively charged. Similarly, when an object has more protons than electrons, the object is said to be positiely charged.
What parts of the atom are charged?
An atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. The positive charges equal the negative charges, so the atom has no overall charge; it is electrically neutral.
Why nucleus is positively charged?
A nucleus of an atom has protons and neutrons. we know that a proton has a charge of + 1 , while a neutron has no charge, or 0. Therefore, the nucleus of an atom will always have a positive charge.
What is a positively charged particle called?
positron, also called positive electron, positively charged subatomic particle having the same mass and magnitude of charge as the electron and constituting the antiparticle of a negative electron.
What is a positively charged object?
"A positively charged object is an object that has an excess of positive electrons."
What happens when a positively charged particle moves close to a neutral atom?
So what happens when a positively charged particle moves close to a neutral atoms? (This is exactly the same as a + charged comb being close to some bits of paper.) The Positive particle will attract the electrons and repel the protons in the neutral atom. The electrons can movve a bit so tend to reside on the side of the atom where the + charge is and less on the other side. The atoms is distorted or polarised y the presence of the positive charge. If we look at the sitauation now, the + charge attracts the electrons which have moved a boit closer to the + charge so the force of attraction is greater. We assume that the protons dont move / move a negligible amount further away from the + charge. Now although the size of the charge on the protons is the same magnitude as the charge on the electrons, they are further away. This means that the force of repulsion is less than the force of attraction so there is a net force of attraction. This is a very small effect compared with the forces between two charged aparticles at the same distance.
What are the subatomic particles?
Atoms can be divided into 3 sub atomic particles namely protons (positively charged),electrons (negatively charged) and neutrons (these doesn't have any charge).
Which type of charge is the same on a proton and electron?
Protons. Protons have a positive charge. Electrons have a negative charge. The charge on the proton and electron are exactly the same size but opposite. Neutrons have no charge.
Where are protons and neutrons in an atom?
The protons and neutrons are present in the nucleus of the atom and the electrons revolve around the nucleus (just like planets revolving around the sun in the solar system). So, the charge of the nucleus would be the sum of the charge of particles present in the nucleus viz, Protons and Neutrons.
What are the two types of electric charges?
There are two types of electric charges; positive and negative (commonly carried by protons and electrons respectively).
Which part of an atom is positively charged?
Thus, it is the nucleus of the atom that is positively charged.
Which subatomic particles are present in the nucleus?
An atom contains three subatomic particles → Neutron, proton, and electron. Electron is negatively charged whereas proton is positively charged whereas neutron has no charge. Protons and neutrons are present in the nucleus. So we can say that the protons have a positive charge and therefore the nucleus will have a positive charge.
What happens when a positively charged particle moves close to a neutral atom?
So what happens when a positively charged particle moves close to a neutral atoms? (This is exactly the same as a + charged comb being close to some bits of paper.) The Positive particle will attract the electrons and repel the protons in the neutral atom. The electrons can movve a bit so tend to reside on the side of the atom where the + charge is and less on the other side. The atoms is distorted or polarised y the presence of the positive charge. If we look at the sitauation now, the + charge attracts the electrons which have moved a boit closer to the + charge so the force of attraction is greater. We assume that the protons dont move / move a negligible amount further away from the + charge. Now although the size of the charge on the protons is the same magnitude as the charge on the electrons, they are further away. This means that the force of repulsion is less than the force of attraction so there is a net force of attraction. This is a very small effect compared with the forces between two charged aparticles at the same distance.
What are the subatomic particles?
Atoms can be divided into 3 sub atomic particles namely protons (positively charged),electrons (negatively charged) and neutrons (these doesn't have any charge).
Which type of charge is the same on a proton and electron?
Protons. Protons have a positive charge. Electrons have a negative charge. The charge on the proton and electron are exactly the same size but opposite. Neutrons have no charge.
Where are protons and neutrons in an atom?
The protons and neutrons are present in the nucleus of the atom and the electrons revolve around the nucleus (just like planets revolving around the sun in the solar system). So, the charge of the nucleus would be the sum of the charge of particles present in the nucleus viz, Protons and Neutrons.
What are the two types of electric charges?
There are two types of electric charges; positive and negative (commonly carried by protons and electrons respectively).
Which part of an atom is positively charged?
Thus, it is the nucleus of the atom that is positively charged.
Which subatomic particles are present in the nucleus?
An atom contains three subatomic particles → Neutron, proton, and electron. Electron is negatively charged whereas proton is positively charged whereas neutron has no charge. Protons and neutrons are present in the nucleus. So we can say that the protons have a positive charge and therefore the nucleus will have a positive charge.