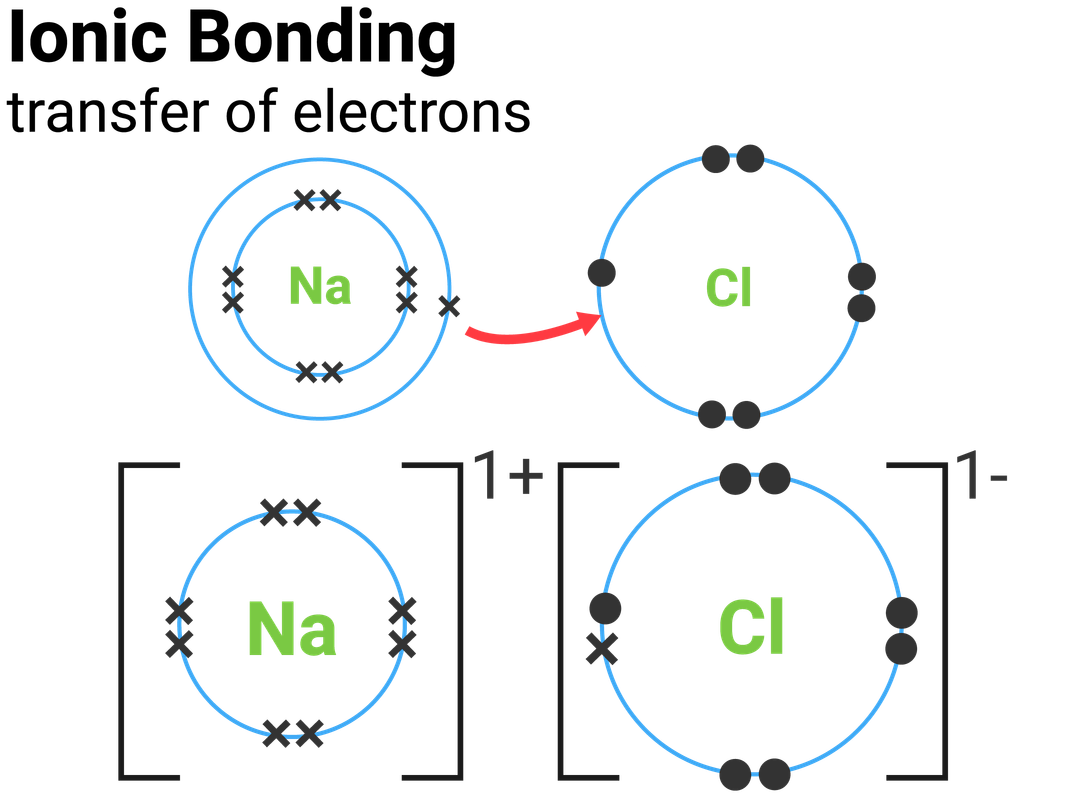
Table salt is a ionic bond since it is a compound made of sodium and chloride. In an ionic bond cations lose electrons and anions
Ion
An ion is an atom or a molecule in which the total number of electrons is not equal to the total number of protons, giving the atom or molecule a net positive or negative electrical charge. Ions can be created, by either chemical or physical means, via ionization.
Full Answer
What is the ionic bond of magnesium?
Ionic bonds are when atoms gain or lose electrons to become charged species (ions) that share an electrostatic interaction called an ionic bond. Magnesium forms a positive ion (cation) with a 2+ charge and chlorine forms a negative ion (anion) with a 1- charge.
Is MgCl2 ionic or covalent?
Magnesium forms a positive ion (cation) with a 2+ charge and chlorine forms a negative ion (anion) with a 1- charge. So two chlorine anions form an ionic bond with one magnesium cation for form MgCl2, a neutral chemical compound.
Is magnesium and bromine ionic or covalent?
An ionic bond. A typical metal - non-metal compound. Magnesium (Mg) reacts with Bromine (Br2) to form Magnesium Bromide (MgBr2) which has an ionic formula of Mg2+ (Br-)2.
Why do chlorine anions form ionic bonds with magnesium cations?
So two chlorine anions form an ionic bond with one magnesium cation for form MgCl2, a neutral chemical compound. Metals and non-metals often form ionic bonds because of the difference in their electronegativity values. Electronegativity is basically a measure of how strongly an element will attract electrons toward it.
What is the atomic number of magnesium?
How many protons does magnesium have?
Which element has the greatest electronegativity?
About this website

Which elements will form an ionic bond with magnesium?
Another example of ionic bonding takes place between magnesium (Mg) and oxygen (O2) to form magnesium oxide (MgO). Magnesium has two valence electrons and an electronegativity of 1,31, while oxygen has six valence electrons and an electronegativity of 3,44.
What would Mg bond with?
oxygenMagnesium (Mg) is able to bond with one oxygen (O) atom. The formula of the compound is MgO.
Is Mg2 O2 ionic or covalent?
1 Answer. No, its ionic.
What type of electron transfer happens between Mg and Mg2+?
An ionic bond is the attraction between oppositely charged ions. Ionic bonds are strong and substantial energy is required to break them. For example, when magnesium reacts with oxygen to form magnesium oxide, the magnesium atom loses two electrons to form the Mg2+ cation, which has electronic configuration 2,8.
Can magnesium form ionic bonds?
Magnesium typically forms single ionic bonds. Magnesium atoms lose two electrons to become Mg+2.
Will Mg and O form an ionic compound?
Magnesium forms a positive ion which attracts to the negative oxide ion. This “attraction” between this oppositely charged ions is called an ionic bond. Formula:MgO. Formed from Mg+2 and O-2, one ion of each to form a stable, neutral compound.
Does sodium and magnesium form ionic bonds?
For example, N a + ions have the 1 + charge state because sodium is a group one element. M g 2 + ions have the 2 + charge state because magnesium is a group two chemical element....Answer.SolubleInsolubleSodium carbonate, potassium carbonate, and ammonium carbonateMost other carbonates4 more rows
Is magnesium a ionic or covalent?
0:011:55Is MgCO3 (Magnesium carbonate) Ionic or Covalent? - YouTubeYouTubeStart of suggested clipEnd of suggested clipSo overall this is an ionic compound. Because of that we should think about charge. So magnesiumMoreSo overall this is an ionic compound. Because of that we should think about charge. So magnesium right here is in group two it has a two plus ionic charge.
Can Mg and O form a covalent bond?
In there is a large difference in electronegativity between magnesium and oxygen. So they form an ionic bond in place of covalent bonds.
How is Mg2+ formed?
Magnesium is in Group 2. It has two electrons in its outer shell. When these electrons are lost, a magnesium ion, Mg 2+, is formed.
What does Mg2+ mean?
magnesium ion (Mg2+) A magnesium atom that has lost two electrons. In the nervous system, a single magnesium ion blocks the opening of each of the cell's NMDA receptor, preventing the influx of calcium ions into the cell.
What is the electron configuration for an Mg2+ ion?
Magnesium atom has 12 protons and 12 electrons and when magnesium loses 2 electrons it produces magnesium 2 plus ion so magnesium 2 plus ion contains 12 minus 2 electrons. That is 10 electrons. So the electron configuration of m g 2 plus that will be 1 s 2.
How does magnesium bond with chlorine?
Mg forms ionic bond to Cl by donating its valence electrons to two Cl atoms. The electron configuration of Mg is [Ne]3s². It can achieve a complete octet by losing its two valence electrons to form Mg²⁺.
Does magnesium have a lone pair?
There are two electron pairs around magnesium and no lone pairs.
Can magnesium make covalent bond?
If, however, when these ions are surrounded by electron rich atoms, such as oxygens, electron density from the oxygens can populate the region around the magnesium ions, reducing their positive charge and leading to covalent bond formation between the ions.
What happens when sulfur combines with magnesium?
MgS is formed by the reaction of sulfur or hydrogen sulfide with magnesium. It crystallizes in the rock salt structure as its most stable phase, its zinc blende and wurtzite structures can be prepared by molecular beam epitaxy.
What is the atomic number of magnesium?
Magnesium has atomic number 12, which means it.
How many protons does magnesium have?
Magnesium has atomic number 12, which means it has 12 protons and 12 electrons. It lost two electrons to form magnesium cation (Mg²⁺) with stable electron configuration like closest noble gas neon (Ne) with 10 electrons.
Which element has the greatest electronegativity?
2) Fluorine (F) is nonmetal with greatest electronegativity, which meand it easily gain electrons.
What are ionic compounds?
ionic compounds have. high melting points. what dissolves easily in water. ionic compounds. crystal lattice. the regular pattern in which a crystal is arranged. ion. a charged particle that forms when an atom or group of atoms gains or loses one or more electrons. ionic bond.
When the number of protons and electrons are not =. atoms become charged particles called?
when the # of protons and electrons are not =. atoms become charged particles called IONs. metal atoms =. few valence electrons. metal atoms form. positive ions. for an atom to lose an electron. the attraction between the electron and the protons has to be broken. small amount of energy.
Why is MgO Ionic?
Magnesium is an alkaline earth metal that has 12 electrons and 12 protons in one atom of Mg.
What is the bond between a metal and a nonmetal called?
Thus, the bond formed between a metal and a non-metal by the transfer of electrons is called an ionic bond .
How many electrons does MgO have?
MgO is an ionic compound formed by the complete transfer of 2 electrons from the valence shell of Magnesium metal to the outermost shell of the O- atom.
How many valence electrons does Mg lose?
1. Ionisation Energy: We saw that Mg loses two valence electrons to form a divalent cation.
Why is energy released when electrons are added?
Energy is released on gaining electrons because the addition of electrons causes stress on other electrons, and consequently energy is released.
Why are ionic compounds brittle?
They are brittle as they break into small pieces by applying pressure.
What is the name of the metal oxide that is made of magnesium and oxygen?
Magnesium oxide (MgO) or magnesia is a metal oxide that is composed of Magnesium (an alkaline earth metal) and oxygen.
Why do metals and nonmetals form ionic bonds?
Metals and non-metals often form ionic bonds because of the difference in their electronegativity values. Electronegativity is basically a measure of how strongly an element will attract electrons toward it. Chlorine (a non-metal) has an electronegativity value of 3.0 and magnesium (a metal) has an electronegativity value of 1.3.
Which atoms are negatively charged?
The magnesium cations coordinate with the partially negatively charged oxygen atoms and the chlorine anions coordinate with the partially positively charged hydrogen atoms. Since numerous water molecules can surround each ion and separate them, this causes them to solvate, or dissolve in the solvent.
What are the two types of mixtures?
3) There are two basic kind of mixtures, homogeneous and heterogeneous. Heterogeneous mixtures are composed of two distinct phases, like sand in water. Homogeneous mixtures are composed of a single phase. Seawater is an example of a homogeneous mixture.
Is magnesium a cation or an anion?
Magnesium forms a positive ion (cation) with a 2+ charge and chlorine forms a negative ion (anion) with a 1- charge. So two chlorine anions form an ionic bond with one magnesium cation for form MgCl2, a neutral chemical compound. Metals and non-metals often form ionic bonds because of the difference in their electronegativity values.
Is magnesium an ionic bond?
Page Citation. Share Link. 1) Magnesium and chlorine form an ionic bond. Covalent bonds are formed when two or more atoms share electrons between them. Ionic bonds are when atoms gain or lose electrons to become charged species (ions) that share an electrostatic interaction called an ionic bond. Magnesium forms a positive ion (cation) ...
Is MgCl2 a polar compound?
In other words, polar solids tend to dissolve in polar solvents and non-polar compounds tend to dissolve in non-polar solvents. We know that MgCl2 is an ionic solid, so we know that it is a polar compound since it is made up of ions. Water (H2O) is a polar solvent.
Is seawater a homogeneous mixture?
Seawater is an example of a homogeneous mixture. The MgCl2 and other ionic solids are completely dissolved in the water, thus forming a single phase to the eye (liquid water). In fact, all ionic solutions are considered homogeneous mixtures.
What is the atomic number of magnesium?
Magnesium has atomic number 12, which means it.
How many protons does magnesium have?
Magnesium has atomic number 12, which means it has 12 protons and 12 electrons. It lost two electrons to form magnesium cation (Mg²⁺) with stable electron configuration like closest noble gas neon (Ne) with 10 electrons.
Which element has the greatest electronegativity?
2) Fluorine (F) is nonmetal with greatest electronegativity, which meand it easily gain electrons.
