Who invented nitrogen fixation?
Fritz HaberFritz Haber, (born December 9, 1868, Breslau, Silesia, Prussia [now Wroclaw, Poland]—died January 29, 1934, Basel, Switzerland), German physical chemist and winner of the 1918 Nobel Prize for Chemistry for his successful work on nitrogen fixation.
When was nitrogen fixation discovered?
Biological nitrogen fixation (BNF), discovered by Beijerinck in 1901 (Beijerinck 1901), is carried out by a specialized group of prokaryotes. These organisms utilize the enzyme nitrogenase to catalyze the conversion of atmospheric nitrogen (N2) to ammonia (NH3).
Where was nitrogen fixation discovered?
Hermann Hellriegel (1831-1895), a noted German agricultural chemist, discovered that leguminous plants took atmospheric nitrogen and replenished the ammonium in the soil through the process now known as nitrogen fixation.
Who discovered non symbiotic nitrogen fixation?
Clostridium spp. The anaerobic nitrogen fixers of significance in natural soils are chiefly the clostridia of which the first representative was found by Winogradsky (1895).
Why is it called nitrogen fixation?
Nitrogen fixation in nature Nitrogen is fixed, or combined, in nature as nitric oxide by lightning and ultraviolet rays, but more significant amounts of nitrogen are fixed as ammonia, nitrites, and nitrates by soil microorganisms.
Which enzyme was first used for nitrogen fixation?
nitrogenaseComplete answer: The enzyme involved is nitrogenase.
How is nitrogen fixation formed?
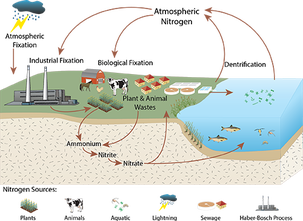
Nitrogen fixation is the process by which nitrogen is taken from its molecular form (N2) in the atmosphere and converted into nitrogen compounds useful for other biochemical processes. Fixation can occur through atmospheric (lightning), industrial, or biological processes.
What is nitrogen fixation in chemistry?
Nitrogen fixation is a chemical process that converts atmospheric nitrogen into ammonia, which is absorbed by organisms. Nitrogen fixation is essentially converting atmospheric nitrogen into a form that plants can more readily utilize.
How was nitrogen discovered by Daniel Rutherford?
Answer and Explanation: Daniel Rutherford discovered nitrogen by doing an experiment in which he turned an empty bottle over, trapping the air in the bottle. He burned a candle in the bottle, which used up the oxygen, leaving behind nitrogen.
What is the name of nitrogen-fixing bacteria?
Nitrogen-fixing bacteria examples comprise Rhizobium (formerly Agrobacterium), Frankia, Azospirillum, Azoarcus, Herbaspirillum, Cyanobacteria, Rhodobacter, Klebsiella, etc. N-fixing bacteria synthesize the unique nitrogenase enzyme responsible for N fixation.
What is nitrogen fixation in biology?
Biological nitrogen fixation (BNF) can be defined as the conversion of atmospheric dinitrogen (N2) to ammonia (NH3) under the combined action of biological and chemical activities (Franche et al., 2009). From: Microbial Management of Plant Stresses, 2021.
What is men by symbiotic nitrogen fixation?
Symbiotic nitrogen fixation is part of a mutualistic relationship in which plants provide a niche and fixed carbon to bacteria in exchange for fixed nitrogen.
How was nitrogen first discovered?
Nitrogen was discovered by chemist and physician Daniel Rutherford in 1772. He removed oxygen and carbon dioxide from air and showed that the residual gas would not support combustion or living organisms. At the same time there were other noted scientists working on the problem of nitrogen.
How was nitrogen discovered by Daniel Rutherford?
Answer and Explanation: Daniel Rutherford discovered nitrogen by doing an experiment in which he turned an empty bottle over, trapping the air in the bottle. He burned a candle in the bottle, which used up the oxygen, leaving behind nitrogen.
When was Rhizobium discovered?
1889The first known species of rhizobia, Rhizobium leguminosarum, was identified in 1889, and all further species were initially placed in the Rhizobium genus. Most research has been done on crop and forage legumes such as clover, alfalfa, beans, peas, and soybeans; more research is being done on North American legumes.
Who discovered nitrogen and how was it discovered?
Nitrogen was discovered in 1772 by chemist and physician Daniel Rutherford, when he removed oxygen and carbon dioxide from air, demonstrating that the residual gas would not support living organisms or combustion, according to the Los Alamos National Laboratory.
What are the three types of nitrogen fixation?
1. Biological nitrogen fixation 2. Atmospheric nitrogen fixation 3. Industrial nitrogen fixation
What are the 7 steps of the nitrogen cycle?
1. Nitrogen fixation. 2. Assimilation 3. Ammonification 4. Nitrification 5. Denitrification 6. Dissimilatory nitrate reduction to ammonium 7. Anaer...
How nitrogen fixation takes place in the soil?
Nitrogen fixation is carried out naturally in soil by microorganisms that are termed diazotrophs. These include bacteria such as Azotobacter and Ar...
What is nitrogen fixation, and why is it important?
The process of fixing atmospheric nitrogen into the soil through various means such as lightning or by certain organisms is called nitrogen fixatio...
What is an example of nitrogen fixation?
Nitrogen fixation in the root nodules of leguminous plants or by the symbiotic association of Azolla and Anabaena in the paddy fields are two examp...
What is nitrogen fixation?
Biological nitrogen fixation is generally referred to the biological process by which nitrogen (N2) in the atmosphere is converted into ammonia. The growth of all organisms depends on the availability of mineral nutrients and nitrogen is required to biosynthesize the basic building blocks of life, e.g. nucleotides for DNA and amino acids for proteins. Formally, nitrogen fixation also refers to other abiological conversions of nitrogen, such as its conversion to nitrogen dioxide. N2 is cannot be used by most organisms because there is a triple bond between the two nitrogen atoms, making the molecule almost inert. In order for nitrogen to be used for growth it must be "fixed" (combined) in the form of ammonium (NH4) or nitrate (NO3) ions. Micro-organisms like bacteria, actinobacteria and certain types of anaerobic bacteria can do this. Some of these organisms are called 'Free-Living Nitrogen-fixing Bacteria' as they live independently of other organisms while others live in intimate 'Symbiotic Associations' with plants or with other organisms (e.g. protozoa). These organisms use an enzyme called nitrogenase to convert atmospheric nitrogen into ammonia which can be easily used by various organisms including plants. Plants that contribute to nitrogen fixation include the legume family – Fabaceae – with taxa such as clover, soybeans, alfalfa, lupines and peanuts. They contain symbiotic bacteria called Rhizobia within nodules in their root systems, producing nitrogen compounds that help the plant to grow and compete with other plants. When the plant dies, the fixed nitrogen is released, making it available to other plants and this helps to fertilize the soil
What is the role of nitrifying bacteria in soil?
He showed that nitrifying bacteria are responsible for transforming ammonia to nitrates in the soil.
What is the enzyme that converts nitrogen into ammonia?
These organisms use an enzyme called nitrogenase to convert atmospheric nitrogen into ammonia which can be easily used by various organisms including plants. Plants that contribute to nitrogen fixation include the legume family – Fabaceae – with taxa such as clover, soybeans, alfalfa, lupines and peanuts.
Why is enrichment culture important?
It improved agricultural practices and proved that nitrogen-fixing bacteria are important in agriculture. Enrichment culture is still used in food industry and biotechnology. Further research in this process led to a better understanding of how these microrganisms work.
How did he postulate that any microbe growing in the medium had to be able to derive its?
By making medium that did not contain a source of fixed nitrogen and then incubating in the presence of air (containing N2), he postulated that any microbe growing in the medium had to be able to derive its nitrogen by performing aerobic nitrogen fixation.
What ions are used to make nitrogen?
In order for nitrogen to be used for growth it must be "fixed" (combined) in the form of ammonium (NH4) or nitrate (NO3) ions. Micro-organisms like bacteria, actinobacteria and certain types of anaerobic bacteria can do this.
When a plant dies, what happens to the nitrogen?
When the plant dies, the fixed nitrogen is released, making it available to other plants and this helps to fertilize the soil. History. Biological nitrogen fixation was discovered by the Dutch microbiologist Martinus Beijerinck. In 1885, he became a microbiologist at the Netherlands Yeast and Alcohol Manufactory in Delft where he isolated ...
What are the Types of Nitrogen Fixation?
Nitrogen fixation is carried by physicochemical and biological means. Only 10 % of natural nitrogen fixation takes place by physicochemical means, whereas 90 % is carried out by biological means. Thus, we can classify nitrogen fixation in following two types:
What is the process of converting nitrogen into nitrogenous compounds?
The process of conversion of atmospheric nitrogen into nitrogenous compounds by microorganisms such as bacteria, fungi and algae is known as Biological Nitrogen Fixation (BNF) or diazotrophy. Some prokaryotes such as bacteria and cyanobacteria that can fix atmospheric nitrogen are called nitrogen fixers or diazotrophs.
How is nitrogen converted into ammonia?
Nitrogen needs to be converted into different forms like ammonia, nitrites or nitrates through nitrogen fixation via various means as the molecular form of nitrogen is of no use to plants and animals. Nitrogen plays a vital role in the life of living organisms. Hence nitrogen fixation is a very necessary and crucial process. Nitrogen fixation is carried by physicochemical (only 10 % and biological 90 % means. Biological nitrogen fixation is mainly performed by a set of microorganisms such as bacteria, fungi, cyanobacteria, etc.
How does nitrogen fixation occur?
Nitrogen fixation occurs with the help of microorganisms as a part of the nitrogen cycle either by natural means or via industrial methods.
Why is nitrogen fixation important?
The process of nitrogen fixation is very important as the molecular form of nitrogen is of no use to plants and animals.
What is the name of the enzyme that reduces nitrogen to ammonia?
The atmospheric nitrogen is reduced to ammonia in the presence of a catalyst known as nitrogenase. This enzyme is found naturally in certain microorganisms like symbiotic ( Rhizobium and Frankia) and non-symbiotic or free-living ( Azospirillum, Azotobacter and BGA).
What did Beijerinck discover?
Later, Beijerinck discovered that there were certain bacteria that were responsible for fixing nitrogen. He named them ‘rhizobia’. He also discovered the symbiotic relationship between the root nodules of leguminous plants and the bacteria.
What is ammonia made of?
In industry, ammonia is synthesized from atmospheric nitrogen and hydrogen by the Haber-Bosch method, a process that Fritz Haber developed about 1909 and which soon after was adapted for large-scale production by Carl Bosch. Commercially produced ammonia is used to make a wide variety of nitrogen compounds, including fertilizer and explosives.
What is the most economical nitrogen fixation process?
The Haber-Bosch process directly synthesizes ammonia from nitrogen and hydro gen and is the most economical nitrogen-fixation process known. About 1909 the German chemist Fritz Haber ascertained that nitrogen from the air could be combined with hydrogen under extremely high pressures and moderately high temperatures in the presence ...
What is nitrogen used for in agriculture?
Nitrogenous materials have long been used in agriculture as fertilizers , and in the course of the 19th century the importance of fixed nitrogen to growing plants was increasingly understood. Accordingly, ammonia released in making coke from coal was recovered and utilized as a fertilizer, as were deposits of sodium nitrate (saltpetre) from Chile. Wherever intensive agriculture was practiced, there arose a demand for nitrogen compounds to supplement the natural supply in the soil. At the same time, the increasing quantity of Chile saltpetre used to make gunpowder led to a worldwide search for natural deposits of this nitrogen compound. By the end of the 19th century it was clear that recoveries from the coal-carbonizing industry and the importation of Chilean nitrates could not meet future demands. Moreover, it was realized that, in the event of a major war, a nation cut off from the Chilean supply would soon be unable to manufacture munitions in adequate amounts.
What is the arc process?
The arc process, however, was costly and inherently inefficient in its use of energy, and it was soon abandoned for better processes. One such method used the reaction of nitrogen with calcium carbide at high temperatures to form calcium cyanamide, which hydrolyzes to ammonia and urea.
What are the two types of nitrogen fixers?
Two kinds of nitrogen-fixing microorganisms are recognized: free-living (nonsymbiotic) bacteria, including the cyanobacteria (or blue-green algae) Anabaena and Nostoc and genera such as Azotobacter, Beijerinckia, and Clostridium; and mutualistic (symbiotic) bacteria such as Rhizobium, associated with leguminous plants, and various Azospirillum species, associated with cereal grasses.
What are root nodules?
root nodules. The roots of an Austrian winter pea plant ( Pisum sativum) with nodules harbouring nitrogen-fixing bacteria ( Rhizobium ). Root nodules develop as a result of a symbiotic relationship between rhizobial bacteria and the root hairs of the plant.
What is nitrogen fixation?
nitrogen fixation, any natural or industrial process that causes free nitrogen (N 2 ), which is a relatively inert gas plentiful in air, to combine chemically with other elements to form more-reactive nitrogen compounds such as ammonia, nitrates, or nitrites. Under ordinary conditions, nitrogen does not react ...
How did Rutherford isolate nitrogen?
In order to isolate nitrogen, Rutherford had to remove oxygen from regular air, long before technology made the process easier. So Rutherford trapped a mouse in a container of air and let the mouse breath in all the oxygen. Then he burned a candle and a piece of phosphorus to make sure all the oxygen was gone.
What was the first thing Rutherford discovered?
He called it “noxious air,” but in fact he was the first to successfully isolate nitrogen. Thanks to Rutherford’s creative experiment, nitrogen was discovered. It wouldn’t take long for other scientists to realize that nitrogen could be used in fertilizers, to make plastics, and to keep tires strong and safe.
Why is nitrogen in tires?
Nitrogen is also a key component in the tire inertion process, since unlike oxygen, nitrogen does not cause corrosion inside tires and it cannot combust. Plus, nitrogen doesn’t leak out of tires as quickly as regular air. That’s one reason why airplane tires, NASCAR tires, and many car tires are inerted with nitrogen instead of regular air.
Who is Genevieve Carlton?
Post Author: Genevieve Carlton Ph.D. Ph.D. - Research Historian from Northwestern University . A writer and researcher she has published pieces for Ranker, Stacker and Atlas Obscura. She has published a nonfiction history book with the University of Chicago Press and a number of scholarly articles with top journals.
Is nitrogen in the atmosphere?
And nitrogen is everywhere–– 78% of the earth’s atmosphere is made of nitrogen, and it is the fifth most abundant element in the universe. But even though nitrogen is in every breath of air, it was surprisingly difficult for scientists to isolate nitrogen gas.
Who discovered nitrogen?
A clever Scottish botanist is credited with discovering nitrogen because he perfected a method for isolating the element in 1772. His name was Daniel Rutherford, and he managed to separate atmospheric nitrogen when he was only a 23-year-old student.
How many people died in the fertilizer explosion?
The resulting blast killed more than 500 people (including those workers), destroyed 80 percent of the homes in the town, and ripped the roofs off of homes 15 miles away. It’s difficult to blame them, though, since no one knew at the time about the fertilizer’s explosive properties.
What is the most transformational technological development of the modern age?
Advertisement. Advertisement. For this reason alone, the Haber-Bosch process is considered by many scientists and historians to be the most transformational technological development of the modern age. But the case gets even stronger once you consider all of its unintended consequences.
How does nitrogen affect ecosystems?
Reactive nitrogen infiltrates surface streams and groundwater, polluting drinking wells. Excess levels of nitrogen in some ecosystems bolsters some species at the expense of others, leading to overall reduced biodiversity. Advertisement.
How does nitrogen fertilizer affect the environment?
Nitrogen fertilizers also stimulate natural bacteria to produce more nitrous oxide, which contributes to acid rain and is a greenhouse gas that traps 300 times more heat per molecule than does carbon dioxide. Reactive nitrogen infiltrates surface streams and groundwater, polluting drinking wells.
What percentage of nitrogen is used for livestock?
Last month, the United Nations Environment Program released a study detailing some of the compounding sources of inefficiency. For example, worldwide about 80 percent of nitrogen harvested in crops and grass goes to feed livestock instead of feeding people directly.
How much of the world's nitrogen goes into fertilizer?
More than 80 percent of that reactive nitrogen goes into fertilizers for agriculture. A 2008 paper in Nature Geoscience contains a remarkable graph showing how closely world population growth has tracked the increase in fertilizer production—and how a world without it could sustain only 3.5 billion people.
What is the nitrogen cascade?
This nitrogen cascade manifests in a variety of ways. Nitrogen oxides contribute to the production of ground-level ozone, or smog, which increases the risk of serious respiratory illness and cancer ; a different problem arises in the stratosphere, where these gases destroy beneficial UV-blocking ozone.
What is the most common catalyst used in explosives?
Synthetic ammonia from the Haber process was used for the production of nitric acid, a precursor to the nitrates used in explosives. Today, the most popular catalysts are based on iron promoted with K 2 O, CaO, SiO 2, and Al 2 O 3. Earlier, molybdenum was also used as a promoter.
How much ammonia is produced by the Haber process?
As of 2018, the Haber process produces 230 million tonnes of anhydrous ammonia per year. The ammonia is used mainly as a nitrogen fertilizer as ammonia itself, in the form of ammonium nitrate, and as urea. The Haber process consumes 3–5% of the world's natural-gas production (around 1–2% of the world's energy supply).
What was the Haber process used for?
Although the Haber process is mainly used to produce fertilizer today, during World War I it provided Germany with a source of ammonia for the production of explosives, compensating for the Allied Powers ' trade blockade on Chilean saltpeter .
How is iron catalyst made?
In industrial practice, the iron catalyst is obtained from finely ground iron powder, which is usually obtained by reduction of high-purity magnetite (Fe 3 O 4 ). The pulverized iron metal is burnt (oxidized) to give magnetite or wüstite (FeO, ferrous oxide) of a defined particle size. The magnetite (or wüstite) particles are then partially reduced, removing some of the oxygen in the process. The resulting catalyst particles consist of a core of magnetite, encased in a shell of wüstite, which in turn is surrounded by an outer shell of iron metal. The catalyst maintains most of its bulk volume during the reduction, resulting in a highly porous high-surface-area material, which enhances its effectiveness as a catalyst. Other minor components of the catalyst include calcium and aluminium oxides, which support the iron catalyst and help it maintain its surface area. These oxides of Ca, Al, K, and Si are unreactive to reduction by the hydrogen.
What is the process of making ammonia?
Main process of ammonia production. Fritz Haber, 1918. The Haber process, also called the Haber–Bosch process, is an artificial nitrogen fixation process and is the main industrial procedure for the production of ammonia today. It is named after its inventors, the German ...
Why is nitrogen gas unreactive?
Nitrogen gas (N 2) is very unreactive because the atoms are held together by strong triple bonds. The Haber process relies on catalysts that accelerate the scission of this triple bond.
What pressure is used for ammonia synthesis?
The steam reforming, shift conversion, carbon dioxide removal, and methanation steps each operate at pressures of about 2.5–3.5 MPa (25–35 bar; 360–510 psi), and the ammonia synthesis loop operates at pressures ranging from 6 to 18 MPa (60 to 180 bar; 870 to 2,610 psi), depending upon which proprietary process is used.
