
Scientists Who Discovered Atoms
- Democritus to John Dalton. In around 400BC the Greek philosopher Democritus proposed the theory that all matter was made up of many small particles.
- Thompson’s Plum Pudding. Building on these ideas and his recent discovery of electrons, J.J.Thompson proposed a new model of the atom in 1904.
- Rutherford’s Gold. ...
Who discovered atoms are made of all substance?
Who discovered atoms make up everything? In the early 1800s, the scientist John Dalton noticed that chemical substances seemed to combine and break down into other substances by weight in proportions that suggested that each chemical element is ultimately made up of tiny indivisible particles of consistent weight.
Who discovered that atoms are small hard particles?
father of the atom; first to claim that matter is made of small, hard particles he called "atomos." John Dalton created first Atomic Theory; atoms are tiny solid balls
Who discovered atoms have a positive nucleus?
The atomic nucleus was discovered by Ernest Rutherford, who proposed a new model of the atom based on Geiger-Marsden experiments. The atomic nucleus occupies only about 10-12 of the total volume of the atom, but it contains all the positive charge and most of the total mass of the atom.
Who invented the first atom?
Who Invented the first Atom
- Childhood Life of John Dalton. John Dalton was born on 5 or 6 September 1766 in the Cockermouth English Lake District to a Quaker tradesman family.
- Interest in the Weather. For the next 15 years, Dalton recorded numerous weather observations. ...
- The World of Elements. ...
- A Humble Hero Honored. ...
- Books by John Dalton

Who discovered atoms have parts?
John DaltonThis idea of tiny, indivisible bits of matter persisted until the 1800's. John Dalton (1766-1844), a great chemist, really started the modern atomic hypothesis.
Who first saw the atom?
Erwin W. Mueller, a physicist and the first man to see an atom—through a special miscroscope that he developed—died yesterday at George Washington University Hospital, Washington. He was 65 years old and a resident of State College, Pa. Dr.
Who are the 5 scientists who discovered atoms?
Oct 21, 1803. JOHN DALTON by google images. ... Apr 30, 1897. J.J THOMSON by google images. ... Dec 14, 1900. MAX PLANCK by google images. ... Apr 30, 1905. ALBERT EINSTEIN by google images. ... Jul 10, 1913. NEILS BOHR by google images. ... Jan 1, 1917. ERNEST RUTHERFORD by google images. ... Jan 28, 1932. JAMES CHADWICK by google images. ... Dec 2, 1942.More items...•
Who is the father of atom?
John Dalton at times was known as the father of modern atomic theory. In 1803, he speculated that all atoms of a given element are identical in size and mass. Dalton; John Dalton reasoned that elements were composed of smaller atoms. This lead to the atomic theory of matter.
Who named atom?
DemocritusBut when it comes to the word atom, we have to go to ancient Greece of 400 B.C. And there was a brilliant philosopher named Democritus, and he proposed the Greek word atomos, which means uncuttable. And so as he explained, all matter was eventually reducible to discrete, small particles or atomos.
When did atoms get discovered?
The first modern evidence for atoms appears in the early 1800s when British chemist John Dalton discovered that chemicals always contain whole number ratios of atoms.
Can the atom be seen?
Atoms are so small that it's almost impossible to see them without microscopes. But now, an award-winning photo shows a single atom in an electric field—and you can see it with the naked eye if you really look hard.
What is the history of atom?
Democritus was a Greek philosopher who was the first person to use the term atom (atomos: meaning indivisible). He thought that if you take a piece of matter and divide it and continue to divide it you will eventually come to a point where you could not divide it any more.
How did Democritus discover the atom?
Democritus had a thought experiment. The idea was if you took a material and divided it half, you would have a smaller but identical chunk. If you keep dividing your material, there should eventually be a point where you've reached the smallest representative element of your material. That element is the"atom".
Who created the first successful model of an atom?
Niels Bohr created the first successful model of an atom. The model showed that the electrons orbit the nucleus just like the planets orbit the sun. This is incorrect however, considering that it is impossible to know exactly how they are actually moving.
What did Thompson conclude about atoms?
Thompson then concluded that atoms must contain negatively charged particles; discovering the electron . He also made the Plum Pudding model of an atom where the electrons were evenly spread throughout a sphere of positvely charged material. http://www.nobelprize.org/nobel_prizes/physics/laureates/1906/thomson.jpg.
How did James Chadwick prove the existence of neutrons?
James Chadwick proved the existance of neutrons by bombarding beryllium with alpha particles. While doing this, he noticed a particle with a mass very close to that of a proton being released. Considering the fact that they were not bent by electrical fields and were highly penetrating, these particles were electrically neutral.#N#http://web.me.com/dtrapp/people2.f/Chadwick.jpg
Which atom has a very small mass?
Neutron. Ernest Rutherford showed that the atom was a lot of empty space, with a small positively charged nucleus that contained most of the mass, and a very small mass of negatively charged particles orbiting the nucleus.
Who first discovered the atom?
In fact, the initial theory of the atom was first recorded by the Greek scientist and philosopher Democritus, all the way back in 440 B.C. Democritus defined his take on what would later become atomic theory by starting with a stone. As he put it, if you were to divide a stone, it would be two smaller stones.
What did John Dalton discover about atoms?
Then in 1808, Chemist John Dalton discovered an interesting fact as he was studying the various elements. In some cases, elements, like tin and oxygen, could merge into various compounds. However, the proportions of each element in every compound always stayed at small numbers, no matter what. If this was the case, there must have been some sort of smaller base unit for the elements. This would be the basis for the atomic theory that he would present that year. Some of his theories, like atoms being indivisible and the foundation for all matter, would be similar to Democritus.
What is the name of the atom that was 2,000 times smaller than hydrogen?
Called corpuscles at first, these were 2,000 times smaller than hydrogen, and would earn Thomson the Nobel Prize. Quiz yourself: Pop Quiz: Chemistry. From there, our knowledge of the atom only grew further and further, from Albert Einstein discovering how to weigh an atom, to learning about the structure of the nucleus, ...
What was Democritus's theory of matter?
The prevailing theory in those days was that all matter consisted of fire, air, earth, and water.
Where does the word "atom" come from?
The word atom is derived from the Greek term for indivisible because, at the time, we thought that it was impossible to split an atom and that they were the smallest known thing in the universe. Now, we know that’s not the case at all. Every atom is made of the smaller components protons, neutrons, and electrons, ...
Who discovered that atoms are not the smallest particles?
And so with all this in mind, our understanding of the atom would continue to evolve. In 1897, J.J. Thomson discovered that atoms were, in fact, not the smallest you could go in terms of particles. At the time, he was experimenting with cathode rays and electric discharge, and while investigating a mysterious glow, he discovered electrons. Called corpuscles at first, these were 2,000 times smaller than hydrogen, and would earn Thomson the Nobel Prize.
Is an atom a building block of life?
While they may provide a few unpleasant high school chemistry flashbacks, atoms are one of the true building blocks of life as we know it. However, there’s one natural question many think of when it comes to atoms. If an atom is so small, how were we able to discover their existence?
How did physicists discover the existence of atoms?
In order to understand how heat engines worked — along with all the attendant concepts like temperature, pressure and entropy — physicists realized that they could view gases and fluids as if they were composed of a nearly numberless quantity of tiny, even microscopic, particles. For example, "temperature" really measures the average motion of all those gas particles hitting your thermometer, transferring their energy to it.
Who discovered that the atom was not indivisible?
Thus, almost a hundred years after Dalton conclusively argued for the existence of the indivisible atom, and at the same time as Einstein was providing a way to directly measure those atoms, Thomson and Rutherford discovered that the atom wasn't indivisible at all. Instead, it was made of even tinier bits.
What did Einstein do with Brownian motion?
Brownian motion was just one of those random unexplained facts of life, but Einstein saw in that a clue. By treating the fluid as something composed of atoms, he was able to derive a formula for how much the innumerable collisions from the fluid particles would nudge that grain around. And by putting this connection on solid mathematical ground, he was able to provide a pathway for going from something you can see (how much the grain moves around in a given amount of time) to something you can't (the mass of the particles of the fluid).
What are these little particles telling us about gold atoms?
And this "nucleus" must be positively charged. Since the total charge of the atom had to be neutral, then the electrons must be very very tiny and swimming, orbiting or dancing around that nucleus in a loose cloud.
How often do alpha particles ricochet off gold?
But every once in a while, the particles would careen off in a random direction. And once in a great while (about 1 out of every 20,000 shots, and yes, the scientists counted manually), an alpha particle ricocheted off the gold, slammed back the way it had come.
What did Dalton find?
Instead, he found that a certain amount of one element might combine with an equal amount of another element. Or with twice or three times the other element. Dalton found only simple proportions, everywhere, in all cases. If matter was ultimately indivisible, if it was made of atoms, then only simple proportions and ratios would be allowed when combining elements.
What did ancient cultures know about matter?
Ancient cultures were certainly aware of of the general idea that matter was composed of more fundamental elements (though they disagreed quite a lot about what exactly counted as an element) and knew that these elements combined in interesting and fruitful ways to make complex things, like chairs and beer.
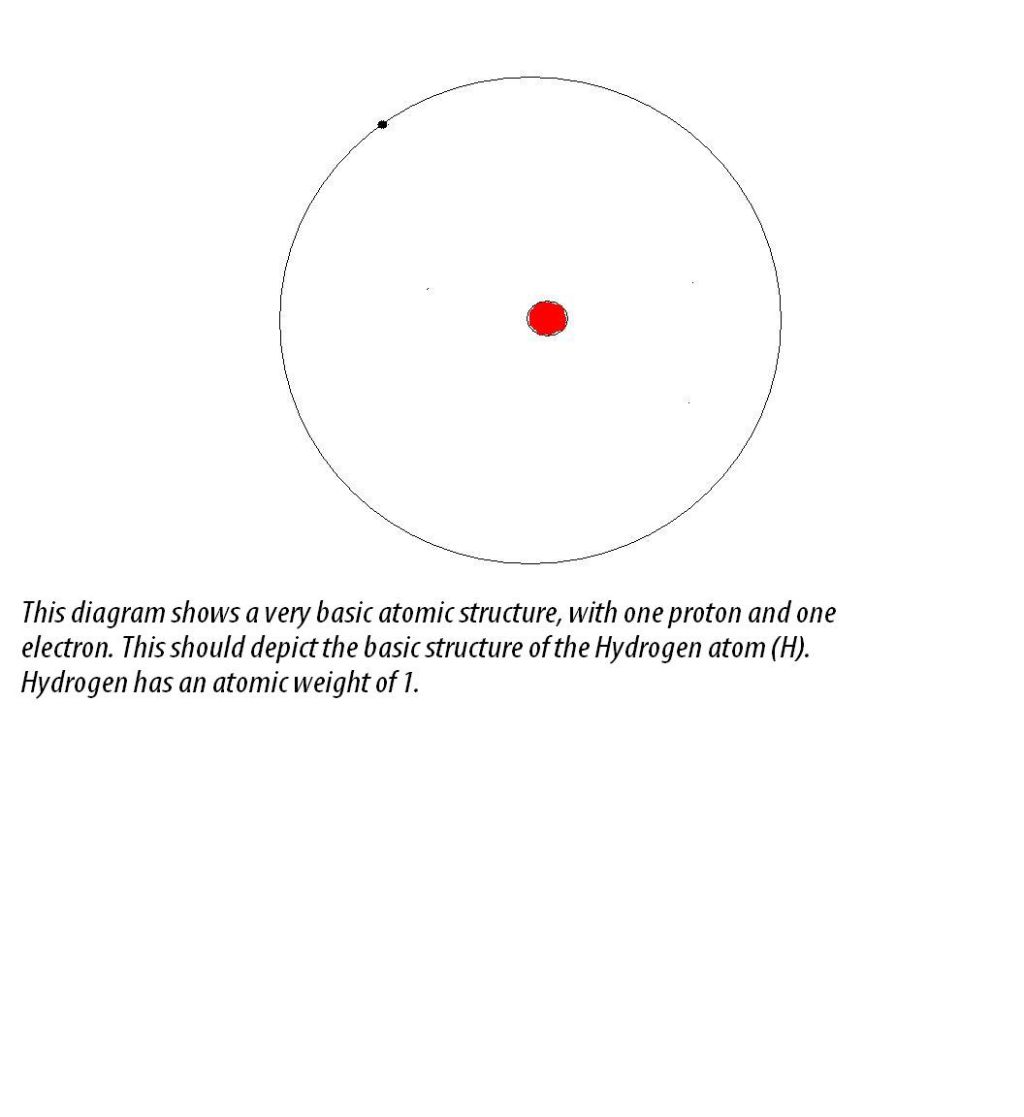
What Is Atomic Structure?
Discovery of Charged Sub-Atomic Particles in An Atom
- In \(1803,\) John Dalton stated the atom is the smallest and indivisible particle of matter. Later, with the discovery of sub-atomic particles, this theory was knocked out. Let’s learn about the discoveries of these sub-atomic particles (electron, proton, and neutron) one by one: 1. Discovery of Electron In \(1897,\) based on cathode rays’ experime...
Evolution of Atomic Models
- 1. Ancient Theory– The word ‘atom’ actually was coined by Ancient Greek that means ‘indivisible’. They outlined the basic idea that everything in the universe is made of atoms, which are invisible and indivisible spheres of matter of endless type and numbers. 2. Dalton’s Atomic Model– In \(1803,\) an English chemist John Dalton developed a more scientific definition of an atom. He …
Atomic Structure of Carbon
- The atomic number of a carbon atom is \(6.\) Carbon has two electronic shells; the first shell holds two electrons, and the second shell holds four electrons out of a possible eight spaces. When atoms tend to bond, they share electrons present in their outermost shell. Since carbon has four empty spaces in its outermost shell, that enables it to bond to four other atoms. Thus, carb…
Summary
- Thus, we can conclude that atoms are the smallest and ultimate building blocks of all matter in the universe. An atom consists of negatively charged electrons around a central nucleus that contains a mass of positively charged protons and electrically neutral neutrons. The entire history of the development of the structure of an atom is very interesting right from the beginning when …