What is the special property of carbon?
Why do electrons not bond carbon atoms?
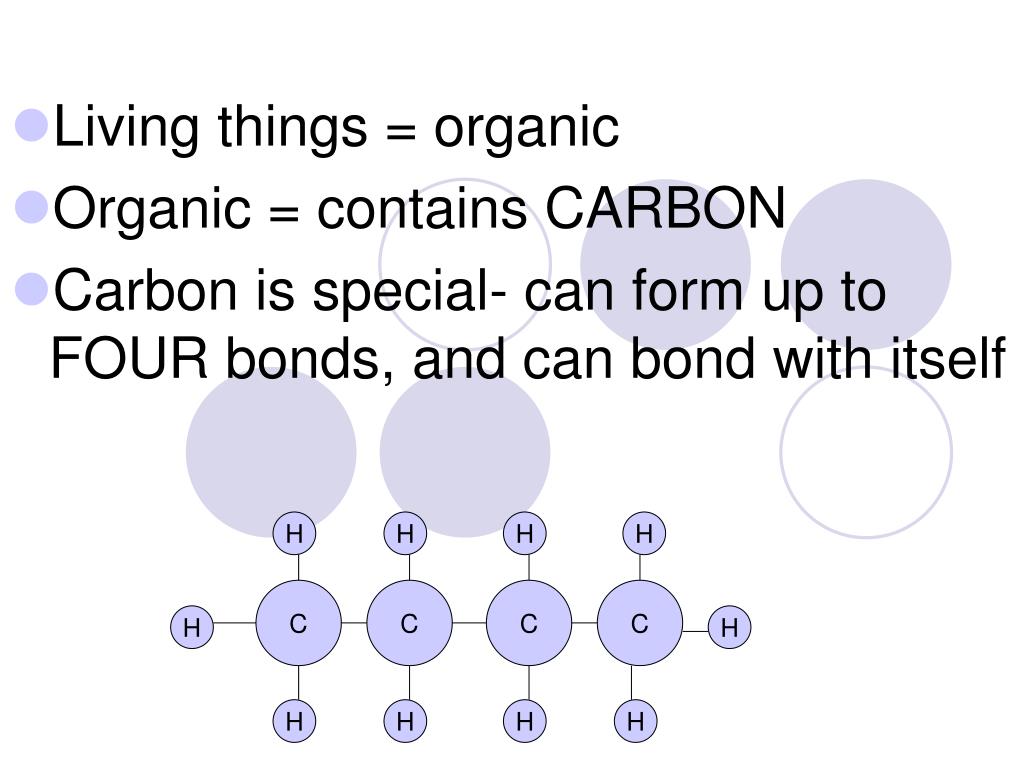
What are the properties of carbon?
Why does a molecule have a long chain of carbons?
Why does carbon form a large number of compounds?
How many electrons does carbon have?
What is a molecule that differs by even one atom or one bond position?
See 2 more

Why can carbon form large and complex structures?
The reason is carbon's ability to form stable bonds with many elements, including itself. This property allows carbon to form a huge variety of very large and complex molecules. In fact, there are nearly 10 million carbon-based compounds in living things!
Why carbon can make long chains?
Carbon atoms can form long chains by sharing their valence electrons with other carbon atoms.
Why can carbon make long chains rings and branched molecules?
Carbon is more than just another element. Of all the elements it is the only one that can form stable linkages to itself as well as to other atoms such as hydrogen and oxygen. This unique ability allows for the formation of carbon frameworks such as long chains, branched chains, rings, spheres and tubes.
What are long chains of carbon molecules called?
Therefore, option A Catenation is correct, the property of Carbon to form long chains or rings as catenation. Q. State the terms : (a) Substances whose atoms or molecules are arranged in a definite pattern.
What are long chains of carbon called?
Answer and Explanation: Polymers are substances made from the joining of many repeating molecular units (not of the same element). Hydrocarbons are compounds containing hydrogen and carbon.
How does the structure of a carbon atom enable it to form large molecules?
Carbon atoms are uniquely suited to build large molecules because they have four valence electrons. This allows carbon atoms to form four single bonds to other atoms, or two double bonds, or one double bond and two single bonds.
Can carbon form long and branched chains?
Carbon has a unique property of ability to form long straight and branched chains called catenation.
What ability allows carbon atoms to form a large number of molecules?
form covalent bonds(i) Carbon has the ability to form covalent bonds with other atoms of carbon giving rise to a large number of molecules through a self-linking property. (ii) Carbon compounds do not show isomerism.
Are longer carbon chains possible?
Currently, palytoxin and maitotoxin are believed to have the longest carbon chains in nature (more than 100 A in length), except for biopolymers. The structural properties of such marine huge molecules are highlighted, especially with regard to the length and shape of their carbon chains.
Can carbon form long or short chains?
Carbon has a unique property of ability to form long straight and branched chains called catenation.
What does a longer carbon chain mean?
The longer carbon chains result in heavier molecules forming. The longer the molecule, the more energy needed to boil it, so it has a higher boiling point.
How do you make a carbon chain longer?
Cyanide ions are very useful in organic synthesis because they can be used to increase the length of the carbon chain. The negative charge on the carbon in a cyanide ion allows it to act as a nucleophile. When a cyanide ion is incorporated into an organic molecule, a nitrile functional group forms.
Two reasons for large number of carbon compounds: - Toppr Ask
Click here👆to get an answer to your question ️ Explain why carbon forms compound mainly by covalent bonds. Explain in brief two main reasons for carbon forming a large number of compounds. Why does carbon form strong bonds with most other elements?
Why does carbon form so many compounds? - Quora
Answer (1 of 8): Carbon is tetravalent, i.e., it can be linked to 4 monovalent groups, which could be same are different. 1. The four bonds are directed in tetrahedral form 2. It can form single, double and triple bonds with itself and with other elements which are capable of forming such bonds ...
Why does carbon form such a large number of compounds? - Toppr Ask
Carbon is the only element that can form so many different compounds because each carbon atom can form four chemical bonds to other atoms and because the carbon atom is just the right, small size to fit in comfortably as parts of very large molecules. This property is called the catenation
Why does carbon form such a large number of compounds?
Hint: To answer this question we should know that, carbon is a non-metal element in the periodic table and non-metals are electronegative and form bonds by electron sharing. When atoms of an element are self-linked, they form a large number of compounds. Complete step by step answer: Carbon forms large number of compound due to following reasons:
Why Carbon is the Basis for so many Compounds
Carbon is not only a part of nearly everything in our lives, it makes life as we know it possible. Without carbon, the compounds and chemical processes necessary for life would not exist.
Why is carbon a stable bond?
Because of this, it can form long chain molecules, each having different properties. Carbon remains in balance with other chemical reactions in the atmosphere and water because of its stability. 3.
Why Is Carbon So Important In Biology?
Carbon is important in biology because without it, life itself would not exist. Carbon is important in everyday life for all living beings in order for them to live, grow, and reproduce. Carbon compounds are also very versatile and they are in many objects we use every day. Remember, the presence of carbon determines whether something is organic or inorganic.
What Is The Carbon Cycle?
Carbon is the fourth most abundant element on earth, and it is a finite resource because it cycles through the earth in so many forms. Without carbon, life as we know it would cease to exist because it is the main element in organic compounds that make up living things. The presence or absence of carbon determines whether an organism is organic or inorganic.
What are allotropes made of?
Allotropes are materials made from the same element , but their atoms fit together differently. Carbon exists on earth in three different allotropes: amorphous, graphite, and diamond. Almost every industry on the planet uses some form of carbon in their every day operations, and we highlight a few of those here. 1.
Why is carbon a finite resource?
Carbon is the fourth most abundant element on earth, and it is a finite resource because it cycles through the earth in so many forms. Without carbon, life as we know it would cease to exist because it is the main element in organic compounds that make up living things.
What is the most important element in life?
Carbon, which so many of us take for granted, is actually one of the most important elements to life as we know it. Carbon 's molecular structure gives it the ability to form stable bonds with other elements, including itself, which makes it the central element of organic compounds. It makes up almost 20% of the weight of an organism, ...
Why is carbon considered a macromolecule?
Because of its ability to form these bonds, carbon can create very large and complex molecules called macromolecules that make up living organisms. This is part of why this versatile element is considered the backbone, or basic structural component, of these molecules.
What is the special property of carbon?
Carbon have very special property is catenation means C can make covalent bonds with several other C atoms which is unique property. 3. The C make bonding with H,S,O,F,Cl,N in 99% of organic compounds. 4. C is an element which makes = bond & triple bonds with many other C & other constituents elements.
Why do electrons not bond carbon atoms?
The fifth reason is that all of the electrons that are not being used to bond carbon atoms together into chains and rings can be used to form bonds with atoms of several other elements. The most common other element is hydrogen, which makes the family of compounds known as hydrocarbons.
What are the properties of carbon?
The properties are as follows.. 1. 4 valence electrons means carbon make 4 covalent bond with others. 2. Carbon have very special property is catenation means C can make covalent bonds with several other C atoms which is unique property. 3. The C make bonding with H,S,O,F,Cl,N in 99% of organic compounds. 4. C is an element which makes = bond & triple bonds with many other C & other constituents elements. 5. C is an element which will show hybridisation & isomeric structure means ISOMERS & ISOMERISM etc.... These are the very special property of C which is not found in the other elements. read less
Why does a molecule have a long chain of carbons?
There are two main reasons: It forms a large number of bonds with other atoms (4 per carbon atom) and it really likes bonding to other carbon atoms. This forms a long chain of carbons (often called the skeleton of the molecule) which can be very long.
Why does carbon form a large number of compounds?
There are lots of reasons why carbon does forms large number of carbon compounds .I’m giving U few of them. The properties which carbon forms many compound is also know as Catenation. Catenation: The tendency of an element to form chains of identical atoms is called catenation. This tendency is observed in the case of non-metals showing covalency ...
How many electrons does carbon have?
Having the atomic number 6, every carbon atom has a total of six electrons. Two are in a completed inner orbit, while the other four are valance electrons—outer electrons that are available for forming bonds with other atoms.
What is a molecule that differs by even one atom or one bond position?
And a molecule that differs by even one atom or one bond position is a molecule of a different compound.
