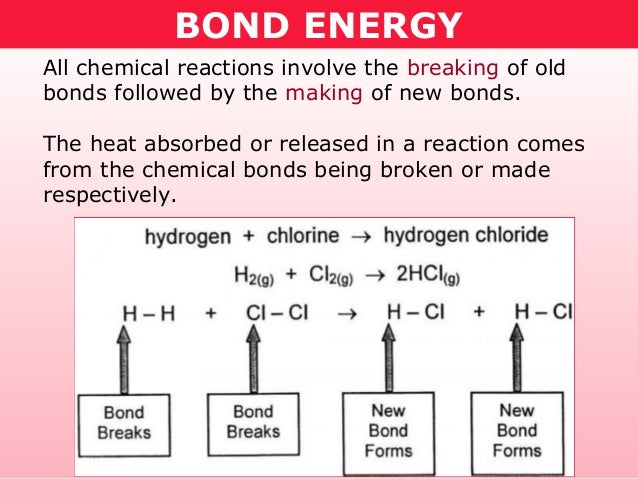
-the thioester bond in acetyl-CoA is a high energy bond oxidative metabolism summary -stepwise fashion, released energy recovered in manageable form at each exergonic step -these packets of energy are conserved by the synthesis of a few types of high energy intermediates who's exergonic breakdown drives the endergonic process
Full Answer
What is a thioester bond?
The thioester bond is what biochemists call a high-energy bond, equivalent to the phosphate bonds in adenosine triphosphate ( ATP ), which is the main supplier of energy in all living organisms. ... It is revealing that thioesters are obligatory intermediates in several key processes in which ATP is either used or regenerated.
What is the role of thioesters in ATP production?
In other words, thioesters could have actually played the role of ATP in a "thioester world" initially devoid of ATP. Eventually, [these] thioesters could have served to usher in ATP through its ability to support the formation of bonds between phosphate groups.
What is the Energy conserved in a thioester reaction?
The reaction works in reverse as well - the energy of a thioester bond (ΔG°′ = −7.5 kcal/mol for hydrolysis) can be conserved in the terminal phosphoanhydride bond of ATP. Other types of thioesters that occur in biochemical processes are indicated.
Why is a thioester a better acylating agent than an ester?
Furthermore, the C-S bond is weaker than the C-O bond, and the thiolate (or thiol, if protonated) is a better leaving group than alkoxide (or alcohol). These factors all make a thioester in general a better acylating agent than an ester.
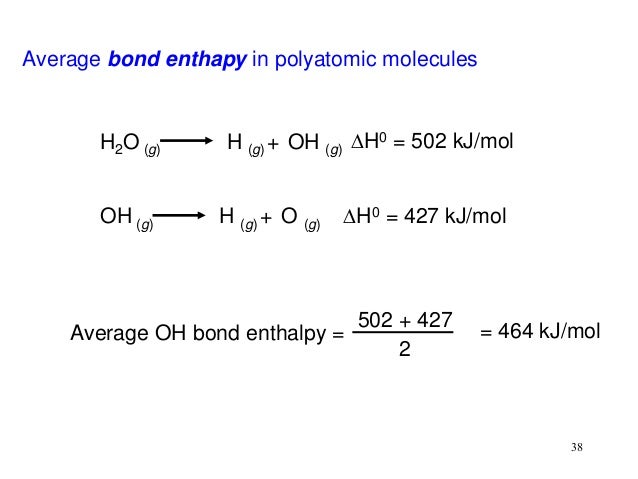
Is thioester a high energy bond?
The thioester bond is what biochemists call a high-energy bond, equivalent to the phosphate bonds in adenosine triphosphate (ATP), which is the main supplier of energy in all living organisms. ...
Why thioesters are high energy compound?
Thioesters were probably involved in the formation of energy-rich phosphates via acylphosphates and pyrophosphates as precursor compounds. The thioester bond is a high-energy bond and probably thioesters had the same function as ATP as an energy supplier.
Why are thioester bonds important?
Thioester bonds are not uncommon in biochemical systems, being formed as intermediates in the reaction mechanisms of a number of enzymes (LIU 1976; DOUGLAS 1987). Thioester bonds are, however, important structural and functional features of certain proteins, including C3.
What is the role of the thioester in this reaction?
Thioesters are involved in the synthesis of all esters, including those found in complex lipids. They also participate in the synthesis of a number of other cellular components, including peptides, fatty acids, sterols, terpenes, porphyrins, and others.
What makes a compound energy-rich?
Energy-rich compounds: Such compounds in biological system whose ΔG0' of hydrolysis yields free energy equal to or greater than -25 kJ/mol are called energy-rich compounds or high-energy compounds, while, those whose ΔG0' of hydrolysis yields free less than -25 kJ/mol are called low-energy compounds.
What makes a compound high energy?
For biochemistry HighEnergy is defined in terms of ATP: if a compound's free energy for hydrolysis isequal to or greater than ATP's then it is “High Energy,” if its free energy of hydrolysis is less than ATP's then it is not a “high energy” compound. Note that ATP has two high energy anhydride bonds (AMP ~P~P).
Is thioester a good leaving group?
A thioester is more reactive than an ester, for example, because a thiolate (RS-) is a weaker base and better leaving group than an alcoxide (RO-). Recall from chapter 7 that the pKa of a thiol is about 10, while the pKa of an alcohol is 15 or higher: a stronger conjugate acid means a weaker conjugate base.
What is the role of thioester in glycolysis?
Thioester is involved in the formation of the first glycolysis intermediate with an acyl-phosphate group, which is part of the slow step of glycolysis e. Thioester is involved in the formation of the first glycolysis intermediate with an acyl-phosphate group, which posses a ΔG for hydrolysis that is highly endergonic.
Why is Acetyl-CoA high energy?
The acetyl group (indicated in blue in the structural diagram on the right) of acetyl-CoA is linked to the sulfhydryl substituent of the β-mercaptoethylamine group. This thioester linkage is a "high energy" bond, which is particularly reactive.
Are thioester bonds unstable?
If we're talking about thermodynamics, and stability with regard to hydrolysis, then yes, thioesters are generally less stable than esters.
Is the hydrolysis of a thioester more or less energetically favorable than the hydrolysis of a simple ester?
Is the hydrolysis of a thioester more or less energetically favorable than the hydrolysis of a simple ester? Explain. The ΔG°′ for the hydrolysis of acetyl CoA has a large negative value: The hydrolysis of a thioester is thermodynamically more favorable than that of an oxygen ester because the electrons of the C=O.
Is thioester bond covalent?
Upon proteolytic activation, the thioester becomes “exposed” on the surface of the molecule and reactive with nucleophiles. Ultimately, a covalent bond is formed between the acyl group of the thioester and the amino or hydroxyl groups of the target.
Why is Acetyl-CoA high energy?
The acetyl group (indicated in blue in the structural diagram on the right) of acetyl-CoA is linked to the sulfhydryl substituent of the β-mercaptoethylamine group. This thioester linkage is a "high energy" bond, which is particularly reactive.
What is the role of thioester in glycolysis?
Thioester is involved in the formation of the first glycolysis intermediate with an acyl-phosphate group, which is part of the slow step of glycolysis e. Thioester is involved in the formation of the first glycolysis intermediate with an acyl-phosphate group, which posses a ΔG for hydrolysis that is highly endergonic.
Do thioesters have resonance?
The thioester bond is a high energy bond. This is apparently because resonance structures which can occur in esters with alcohols, and which stabilizes them, cannot occur in thioesters. The greater size of the sulfur atom (compared to the carboxyl oxygen) diminishes the stability of the resonance forms.
How does Acetyl-CoA form ATP?
The citric acid cycle, where acetyl CoA is modified in the mitochondria to produce energy precursors in preparation for the next step. Oxidative phosphorylation, the process where electron transport from the energy precursors from the citric acid cycle (step 3) leads to the phosphorylation of ADP, producing ATP.
What are thioester-containing proteins?
The thioester-containing proteins (TEPs) are accessory proteins of the complement system, homologous to proteins such as C3, C4, a2M and CD109s. TEPs promote the opsonization of microorganisms and their elimination by phagocytosis in some invertebrates by activating the Toll signaling pathway (Bou et al., 2011; Dostálová et al., 2017). Seven TEP genes have been functionally characterized through molecular cloning in scallops, such as CfTEP and CfA2M in C. farreri ( Zhang et al., 2007a; Xue et al., 2017) and PyC3, PyA2M, PyTEP1, PyTEP2 and PyCD109 in Patinopecten yessoensis ( Liao et al., 2018). The family of TEPs in invertebrates is diverse and the difference between variants makes this family a potential model to explore the functional divergence in bivalve immunity. Recently, the structural analysis of several scallop TEPs showed the existence of a highly variable central region produced by the splicing of six exons, which has an important impact in the specificity of the immune response since the number of produced isoforms differs according to the immune challenge (Zhang et al., 2009a ). In C. farreri, it was further demonstrated that CfTEP, like a C3 protein, undergoes fragmentation due to the action of endogenous serum proteases ( Xue et al., 2017 ).
What is the role of thiol ester in biochemistry?
The central role of thiol ester compounds in nutritional biochemistry is illustrated by considering some features of the mechanisms of the reactions catalyzed by citrate synthase and acetyl-CoA carboxylase. A brief background on the properties of the thiol ester bond is first presented. This material has been simplified and is intended only to clarify why the reactions employ thiol esters of carboxylic acids, rather than oxygen esters or the free, unesterified carboxylic acid.
What is the thiol ester linkage?
The thiol ester linkage is an important concern to those studying the mechanisms of action of acetyl-CoA carboxylase, citrate synthase, propionyl-CoA carboxylase, and HMG-CoA synthase.
What is the role of enolization in stabilizing the 2-carbanion?
Stabilization of the 2-carbanion by enolization is thought to be a critical step in the reactions catalyzed by citrate synthase and acetyl-CoA carboxylase. With the former enzyme, the carbanion attacks an incoming molecule of OAA. As shown in Figure 4.78, condensation of acetyl-CoA with OAA forms citryl-CoA. Hydrolysis of citryl-CoA then produces citric acid and acetyl-CoA. In the case of acetyl-CoA carboxylase, the carbanion attacks an incoming molecule of CO 2/ resulting in formation of malonyl-CoA ( Figure 4.79 ).
What is FIGURE 4.73?
FIGURE 4.73. Condensation of positive and negative carbons to form a covalent bond. The carbanion does not normally occur in solution. It is formed at the active site of enzymes where it exists only momentarily.
Which protein removes thioester linked palmitate?
Thioester linked palmitate is removed from proteins by acyl protein thioesterases, which include APT1 and APT2 as well as the α/β hydrolase domain containing proteins ABHD17A, B, and C (Won et al., 2018).
Which structural elements are responsible for the unusual active center conformation and the extremely low esterolytic activity of resting?
In conclusion, the mutational studies identified some of the structural elements responsible for the unusual active center conformation and the extremely low esterolytic activity of resting-state factor D. They include unique residues lining the specificity pocket (Lys 192, Thr 214, Ser 215, Arg 218, and Val 219 ), residues forming the surface loop 184−188 (Glu 184, Ser 185, Asn 186, Arg 187, Arg 188 ), and Ser 94. Results of esterolytic assays indicate that these residues act synergistically.
What is the function of thioesters in chemistry?
Thioesters provide useful chemoselectivity in the synthesis of biomolecules. A reaction unique to thioesters is the Fukuyama coupling, in which the thioester is coupled with an organozinc halide by a palladium catalyst to give a ketone. Thioesters are components of the native chemical ligation method for peptide synthesis.
What is the thioester in biochemistry?
Biochemistry. Structure of acetyl coenzyme A , a thioester that is a key intermediate in the biosynthesis of many biomolecules. Thioesters are common intermediates in many biosynthetic reactions, including the formation and degradation of fatty acids and mevalonate, precursor to steroids.
What is the structure of a thioester?
General structure of a thioester. In chemistry thioesters are compounds with the functional group R–S–CO–R'. They are analogous to carboxylate esters with the sulfur in the thioester playing the role of the linking oxygen in the carboxylate ester. They are the product of esterification between a carboxylic acid and a thiol.
How are thionoesters made?
They can also be made by the reaction of Lawesson's reagent with esters or by treating pinner salts with hydrogen sulphide. An alternatively, various thionoesters may be prepared through the transesterification of an existing methyl thionoester with an alcohol under base-catalyzed conditions.
What are thioesters in ATP?
It is revealing that thioesters are obligatory intermediates in several key processes in which ATP is either used or regenerated. Thioesters are involved in the synthesis of all esters, including those found in complex lipids. They also participate in the synthesis of a number of other cellular components, including peptides, fatty acids, sterols, terpenes, porphyrins, and others. In addition, thioesters are formed as key intermediates in several particularly ancient processes that result in the assembly of ATP. In both these instances, the thioester is closer than ATP to the process that uses or yields energy. In other words, thioesters could have actually played the role of ATP in a "thioester world" initially devoid of ATP. Eventually, [these] thioesters could have served to usher in ATP through its ability to support the formation of bonds between phosphate groups.
How to make thioesters?
Synthesis. The most typical route to thioester involves the reaction of an acid chloride with an alkali metal salt of a thiol: Another common route entails the displacement of halides by the alkali metal salt of a thiocarboxylic acid. For example, thioacetate esters are commonly prepared by alkylation of potassium thioacetate:
What is the dehydration agent for thioesters?
A typical dehydration agent is DCC. Efforts to improve the sustainability of thioester synthesis have also been reported utilising safer coupling reagent T3P and greener solvent cyclopentanone. Acid anhydrides and some lactones also give thioesters upon treatment with thiols in the presence of a base.
What are thioesters in cellular processes?
In addition, thioesters are formed as key intermediates in several particularly ancient processes that result in the assembly of ATP.
What are thioesters in ATP?
It is revealing that thioesters are obligatory intermediates in several key processes in which ATP is either used or regenerated. Thioesters are involved in the synthesis of all esters, including those found in complex lipids.
What is the name of the group that forms when a sulfhydryl is bonded with sulfur?
A thioester forms when a sulfhydryl (whose general form is written as an organic group, R, bonded with sulfur and hydrogen, hence R-SH) joins with a carboxylic acid (R'-COOH). A molecule of water (H2O) is released in the process, and what remains is a thioester: R-S-CO-R'. ... The thioester bond is what biochemists call a high-energy bond, ...
What is the name of the compound that is derived from the bonding of sulfur with an acyl group?
Thioester. Thioesters are compounds resulting from the bonding of sulfur with an acyl group with the general formula R-S-CO-R'. They are the product of esterification between a carboxylic acid and a thiol (as opposed to an alcohol in regular esters ). Additional recommended knowledge.
Is thionoester an isomeric compound?
Thionoesters are isomeric with thioesters. In a thionoester, sulfur replaces the carbonyl oxygen in an ester. Methylthionobenzoate is C 6 H 5 C (S)OCH 3. Such compounds are typically prepared by the reaction of the thioacyl chloride with an alcohol, but they can also be made by the reaction of Lawesson's reagent with esters.

Overview
Biochemistry
Thioesters are common intermediates in many biosynthetic reactions, including the formation and degradation of fatty acids and mevalonate, precursor to steroids. Examples include malonyl-CoA, acetoacetyl-CoA, propionyl-CoA, cinnamoyl-CoA, and acyl carrier protein (ACP) thioesters. Acetogenesis proceeds via the formation of acetyl-CoA. The biosynthesis of lignin, which comprises a large fraction of the Earth's land biomass, proceeds via a thioester derivative of caff…
Synthesis
The most typical route to thioester involves the reaction of an acid chloride with an alkali metal salt of a thiol:
RSNa + R′COCl → R′COSR + NaCl
Another common route entails the displacement of halides by the alkali metal salt of a thiocarboxylic acid. For example, thioacetate esters are commonly prepared by alkylation of pota…
Reactions
Thioesters hydrolyze to thiols and the carboxylic acid:
RC(O)SR' + H2O → RCO2H + RSH
The carbonyl center in thioesters is more reactive toward amine nucleophiles to give amides:
In a related reaction, but using a soft-metal to capture the thiolate, thioesters are converted into esters. Thioesters provide useful chemoselectivity in the synthesis of biomolecules.
Thionoesters
Thionoesters are isomeric with thioesters. In a thionoester, sulfur replaces the carbonyl oxygen in an ester. Methyl thionobenzoate is C6H5C(S)OCH3. Such compounds are typically prepared by the reaction of the thioacyl chloride with an alcohol.
They can also be made by the reaction of Lawesson's reagent with esters or by treating pinner salts with hydrogen sulphide. An alternatively, various thionoesters may be prepared through the trans…
See also
• Thiocarboxylic acid
• Liebeskind-Srogl coupling
• Aldrithiol-2