Glyceraldehyde has one chiral center and therefore exists as two different enantiomers with opposite optical rotation: In the D/L nomenclature, either D from Latin Dexter meaning "right", or L from Latin Laevo meaning "left" In the R/S nomenclature, either R from Latin Rectus meaning "right", or S from Latin Sinister meaning "left"
What is D and L form of glyceraldehyde?
Glyceraldehyde is chiral, and its two isomers are labeled d and l (typically typeset in small caps in published work). Click to see full answer. Likewise, what is D and L form?
Is glyceraldehyde an aldose?
It is the simplest of all common aldoses. It is a sweet, colorless, crystalline solid that is an intermediate compound in carbohydrate metabolism. The word comes from combining glycerol and aldehyde, as glyceraldehyde is glycerol with one alcohol group oxidized to an aldehyde.
How many isomers does glyceraldehyde have?
Glyceraldehyde is chiral, and its two isomers are labeled d and l (typically typeset in small caps in published work). Click to see full answer.
How is L-glyceraldehyde synthesized?
Its enantiomer, the L-glyceraldehyde is synthesized in the laboratory. Interestingly, in many experiments carried out by Emil Fischer and other scientists, it was determined that shortening the carbon chain of most naturally occurring carbohydrates, it is possible to obtain the D-glyceraldehyde.
Are D-glyceraldehyde and L glyceraldehyde enantiomers?
For glyceraldehyde, the S enantiomer is also called L, and the R enantiomer is called D.
What is the difference between L glyceraldehyde and D-glyceraldehyde?
D-glyceraldehyde is dextro-rotatory, that is, it rotates the plane polarised light to right side whereas L-glyceraldehyde is laevo-rotatory, that is, it rotates the plane polarised light to left side.
What is the relationship between D and L sugars?
D- and L- notation provides a quick shorthand for designating enantiomers. D-Glucose is the enantiomer of L-Glucose, for example. As L-Alanine is the enantiomer of D-Alanine. if the OH on the bottom chiral centre points to the left, it is referred to as L- .
Is glyceraldehyde an L or D?
In the D/L system, glyceraldehyde is used as the configurational standard for carbohydrates. Monosaccharides with an absolute configuration identical to (R)-glyceraldehyde at the last stereocentre, for example C5 in glucose, are assigned the stereo-descriptor D-. Those similar to (S)-glyceraldehyde are assigned an L-.
What is the difference between D form and L form?
The main difference between L and D isomers is in the position of –OH group in the penultimate carbon atom. In D isomer, the OH- group of the penultimate carbon is positioned on the right side whereas, in L isomer, the OH- group of the penultimate carbon is positioned on the left side.
Is D and L the same as and?
d- and l- is the exact same with (+) and (-) notation. D-L system (also called Fischer–Rosanoff convention) is mainly used for naming α-amino acids and sugars. It compares the relative configurations of molecules to the enantiomers of glyceraldehyde. This convention is still in common use today.
What does it mean to say that D and L glyceraldehyde are enantiomers?
The assignment of D and L is used to distinguish between two molecules that relate to each other with respect to reflection; with one molecule being a mirror image of the other. These types of molecules are referred to as chiral for this reason, and the two pairs are called enantiomers. L and D Isomers.
What is difference between D and L sugars?
Summary – D vs L Glucose The difference between D and L glucose is that in D-glucose, three hydroxyl groups and one hydrogen group are in the right side whereas, in L-glucose, the three hydroxyl groups and one hydrogen group are in the left side.
What's the difference between D and L sugars?
Main Difference – D vs L Glucose Glucose is a sugar molecule that is found as either D-Glucose or L-Glucose in nature. The main difference between D and L Glucose is that D-Glucose rotates plane polarized light clockwise whereas L-Glucose rotates plane polarized light anticlockwise.
What is D and L?
The notations D and L are used to describe the configurations of carbohydrates and amino acids. Glyceraldehyde has been chosen as arbitrary standard for the D and L notation in sugar chemistry. Because, this has an asymmetric carbon and can exist as a pair of enantiomers.
What is D and L structure?
The d/l system (named after Latin dexter and laevus, right and left) names molecules by relating them to the molecule glyceraldehyde. Glyceraldehyde is chiral, and its two isomers are labeled d and l (typically typeset in small caps in published work).
What is D and L isomers?
When we talk about D and L isomers we are basically referring to two different forms of enantiomers. Here the main concept to understand is that the two molecules (stereoisomers) are usually related to each other by a reflection. What it means is that the molecules are mirror images of each other.
What are the reactions that are proposed for the formation of D-glyceraldehyde and L-
The reactions proposed for the formation of D-glyceraldehyde and L-alanine involve the reactions of carbon monoxide either with itself (condensation) or with an imine as follows. However, it is well known that these reactions do not proceed easily. A mechanism is needed whereby this could have occurred.
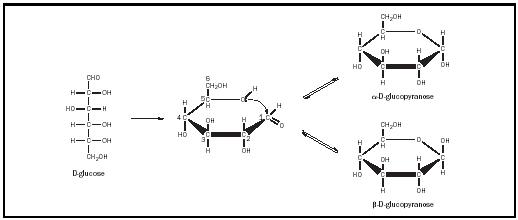
How many chiral carbon atoms does D-glucose have?
Since D -glucose is an aldohexose, it must possess four chiral carbon atoms and can exist in 2 4 = 16 stereoisomers (see Table I ).
What is the stereoselective addition of amines to -alkoxyalkenoates
The stereoselective addition of amines to γ-alkoxyalkenoates provides access to homochiral carbapenem intermediates. 191 Addition of benzylamine to the ester derived from D-glyceraldehyde acetonide yields a β-amino ester with R stereochemistry at C3. Running the reaction at higher temperatures leads to mixtures of diastereomeric products. Interestingly, both the E -and Z - alkenes give the same stereochemical result. This method has also been applied to the synthesis of C-methyl analogues of L-acosamine. 192,193
How to represent monosaccharides?
The system commonly used to represent three-dimensional linear molecules such as monosaccharides two dimensionally is the Fischer projection formula. This affords an unambiguous way of depicting monosaccharides, as follows. (1) The carbon chain is drawn vertically, with the carbonyl group at (or nearest to) the top and the last carbon atom in the chain (i.e., the one farthest from the carbonyl group) at the bottom. (2) Each carbon atom is rotated around its vertical axis until all of the vertical (C C) bonds in the chain lie below an imaginary curved plane such as that of a rolled piece of paper, and all of the horizontal bonds (parallel to the x axis) lie above the plane of the paper. The curved plane is then flattened, and the projection of the molecule is represented as viewed (see Fig. 2).
How are pyridines formed?
Pyridines were formed when vinyl azides were reacted with monocyclic cyclopropanols in the presence of Mn (acac) 3, whereas 2-azabicyclo [3.3.1]non-2-en-1-ol derivatives were obtained from bicyclic cyclopropanols using a catalytic amount of Mn (acac) 3 ( Scheme 1.39) [116].
When was the relative configuration of D-glucose established?
The relative configuration of D -glucose was established by Emil Fischer in 1891 and constituted at the time a monumental achievement, for which he earned a Nobel prize. Nowadays, the determination of the absolute configuration of a monosaccharide offers no difficulty, because the configurations of a large number of related compounds are available. The unknown is simply converted to a compound of known configuration by means of reactions that do not affect the configuration at the chiral center (s).
Where is the carbonyl group in monosaccharides?
(1) The carbon chain is drawn vertically, with the carbonyl group at (or nearest to) the top and the last carbon atom in the chain (i.e., the one farthest from the carbonyl group) at the bottom.
Carbon Monoxide Clusters in the Formation of D-Sugars, L-Amino Acids, Ribonucleotides and Deoxyribonucleic Acids in Prebiotic Molecular Evolution on Earth
A property of most biological molecules is molecular asymmetry or chirality (Greek for hand) [ 1 ], such that the molecules are not identical to their mirror images, termed enantiomorphs.
Carbohydrates
The stereochemistry of each stereogenic center of a monosaccharide can be assigned by the R,S notation. However, late in the nineteenth century, the German chemist Emil Fischer devised a stereochemical nomenclature that preceded the R,S notation. It is still in common use for carbohydrates and amino acids ( Chapter 14 ).
Retention Mechanisms in Different HPLC Types
Serban C. Moldoveanu, Victor David, in Essentials in Modern HPLC Separations, 2013
Synthesis and modifications of carbohydrates, using biotransformations
Alison M Daines, ... Sabine L Flitsch, in Current Opinion in Chemical Biology, 2004
What is the chemical formula for glyceraldehyde?
Chemical compound. Glyceraldehyde ( glyceral) is a triose monosaccharide with chemical formula C 3 H 6 O 3. It is the simplest of all common aldoses. It is a sweet, colorless, crystalline solid that is an intermediate compound in carbohydrate metabolism.
How is glyceraldehyde prepared?
Glyceraldehyde can be prepared, along with dihydroxyacetone, by the mild oxidation of glycerol, for example with hydrogen peroxide and a ferrous salt as catalyst. Dihydroxyacetone, the simplest ketose, is an isomer of glyceraldehyde.
How many chiral centers does glyceraldehyde have?
Glyceraldehyde has one chiral center and therefore exists as two different enantiomers with opposite optical rotation: In the D/L nomenclature, either D from Latin Dexter meaning "right", or L from Latin Laevo meaning "left".
What is the D/L system?
In the D/L system, glyceraldehyde is used as the configurational standard for carbohydrates. Monosaccharides with an absolute configuration identical to ( R )-glyceraldehyde at the last stereocentre, for example C5 in glucose, are assigned the stereo-descriptor D-. Those similar to ( S )-glyceraldehyde are assigned an L-.
What is the D and L notation?
The D and L notation is applied in reference to glyceraldehyde which naturally occurs in the D form. Its enantiomer, the L-glyceraldehyde is synthesized in the laboratory. Interestingly, in many experiments carried out by Emil Fischer and other scientists, it was determined that shortening the carbon chain of most naturally occurring carbohydrates, ...
Which method is used to describe the absolute configuration of chiral centers in carbohydrates?
Although the R and S system, we are familiar with, can be used to designate the absolute configuration of chiral centers in carbohydrates, an older method, proposed in 1906 by a New York University chemist, M. A. Rosanoff, is often the preferred way of describing the stereochemistry of sugars.
Is glucose a D isomer?
For example, glucose one of the most common and important carbohydrates also used extensively for the initial studies, was found to exist naturally as a D isomer. The enantiomer, L-glucose can still be prepared synthetically:
Is D sugar dextrorotatory?
Even though the “D” notation was initially used as an abbreviation to dextrorotatory (turning the plane of polarized light clockwise) since D-glucose is in fact dextrorotatory, it was found later that not all D sugars are dextrorotatory.
Is D a diastereomer or an enantiomer?
Yes, we stated that D and L isomers are enantiomers since all the chiral centers have opposite configuration. This is true, however, when dealing with cyclic forms of sugars, you need to keep in mind epimers which are diastereomers that differ in the configuration of only one chiral center. And if these diastereomers are cyclic hemiacetals like ...
