Converting a Fischer Projection To A Haworth (And Vice Versa)
- Starting with a Fischer projection, it’s not too hard to convert it to a Haworth once you know a few tricks.
- For C-2, C-3, and C-4, if the OH is on the right hand side of the Fischer, it will be down in the Haworth. ...
- If the sugar is D, draw the C-5 CH 2 OH pointing up.
...
Interchanging any three groups on a carbon does a bond rotation.
- H → OH.
- OH→ CH2OH.
- CH2OH→ H.
What is the difference between Fischer projection and Haworth projection?
The “right” groups on carbons 2, 3, and 4 in the Fischer projection go onto the “bottom” positions in Haworth The “left” groups on carbons 2, 3, and 4 in the Fischer projection go onto the “up” positions in Haworth
How do you convert Fischer to Haworth?
The first step in converting a Fischer to a Haworth is to draw in these wedges and to number the carbons. Next, let’s turn this molecule on its side, 90 degrees clockwise. [it must be clockwise – see note]
What are the applications of Fischer projections?
An important application of Fischer projections is the ease of conveying the stereochemistry of carbohydrates and their conversion from Fischer, Haworth, and chair structures. A good strategy here is to convert the Newman projection to a bond-line structure and from there get to the Fischer projection as we did above.
How do you draw a Haworth stem?
Draw a Haworth’s “stem” like in the picture above Number your atoms 1 through 5 starting from the anomeric carbon and going clockwise If your sugar was D, then the carbon #6 is going to be looking “up” (down for the L sugar) The “right” groups on carbons 2, 3, and 4 in the Fischer projection go onto the “bottom” positions in Haworth
What is the difference between Fischer and Haworth projection?
The key difference between Fischer projection and Haworth projection is that the Fischer projection shows the open chain structure of organic molecules, whereas the Haworth projection shows the closed-cyclic structure of organic molecules.
What is Haworth projection formula?
In chemistry, a Haworth projection is a common way of writing a structural formula to represent the cyclic structure of monosaccharides with a simple three-dimensional perspective.
How do you convert Fischer projection to Newman projection?
3:358:32Converting Newman to Fischer Projections (vid 4 of 5) - YouTubeYouTubeStart of suggested clipEnd of suggested clipThe forward carbon has an H to the right hydrogen to the left and a ch3 going down the rear carbonMoreThe forward carbon has an H to the right hydrogen to the left and a ch3 going down the rear carbon has a hydrogen to the right bromine to the left and a ch3.
How do you convert Fischer projections to bond lines?
This means, for example, when you are converting a Fischer projection to a bond-line, you can simply draw the zig-zag with the correct number of carbons and add the groups by randomly assigning them wedge and dash notation.
What is Fischer projection formula?
A fischer projection formula is a convention used to depict a stereo formula in two-dimensional spaces without destroying the stereochemical information, the absolute configurations, at chiral centres.
What is R and S in Fischer projections?
For a sugar drawn in a Fischer projection with the most oxidized group at the top (i.e. a carboxylic acid or aldehyde), a chiral center with OH on the right will be R, and a chiral center with OH on the left will be S. This makes assigning R and S to sugars in the Fischer projection a very quick process.
Can you flip a Fischer projection?
Because the "up" and "down" aspects of the bonds don't change, a Fischer projection may be rotated by 180 degrees without changing its meaning. A Fischer projection may not be rotated by 90 degrees. Such a rotation typically changes the configuration to the enantiomer.
How do you read Fischer projections?
10:1512:40Fischer Projections How to Draw and Interpret for Single and ... - YouTubeYouTubeStart of suggested clipEnd of suggested clipAnd you rotate at 90 degrees you're swapping all your dashes and wedges getting the exact mirrorMoreAnd you rotate at 90 degrees you're swapping all your dashes and wedges getting the exact mirror image but if you take that Fischer projection and flip it upside down.
What is D or L in Fischer projection?
If the hydroxyl group (or amino group for amino acids) is pointing to the right in the Fischer Projection, the sugar (or amino acid) is designated as D. If the hydroxyl group (or amino group for amino acids) is pointing to the left in the Fischer projection, the sugar (or amino acid) is designated as L.
How do you convert Fischer projection to zigzag?
about the conversion of a Fischer to zig-zag projection is that the placement of two adjacent groups (on the same side of the Fischer projection) to the oppo- site sides of the zig-zag back- bone can provide the correct zig-zag structure. This is true only if we are concerned with the relative stereochemistry.
How do you convert to bond line structures?
Converting a Condensed Structure to a Bond-line Number all the atoms (except for H's). ... Simply connect all the atoms from left to right. ... Rearrange the atoms such that they have the correct number of bonds: (It will be very beneficial to learn the functional groups at this point)More items...
How do you know if a Fischer projection is chiral?
If there is an absence of plane of symmetry in the Fischer projection, then the Fischer projection is chiral. If there are two chiral centers present in the Fischer projection and the groups attached to both the chiral centers are the same.
What is the projection formula?
The vector projection of a vector onto a given direction has a magnitude equal to the scalar projection. The formula for the projection vector is given by p r o j u v = ( u ⋅ v | u | ) u | u | .
What does projection formula mean?
Definition of projection formula : a perspective formula projected so as to represent it in two dimensions — compare structural formula.
What is projection formula in organic chemistry?
A formal two-dimensional representation of a three-dimensional molecular structure obtained by projection of bonds (symbolized as lines) onto a plane with or without the designation of the positions of relevant atoms by their chemical symbols.
What is the use of projection formula?
Projection formulae is the length of any side of a triangle is equal to the sum of the projections of other two sides on it. Note: We observe in the above diagram BD and CD are projections of AB and AC respectively on BC. Now, substitute the value of BD and CD in equation (ii) we get, a = b cos C - (-c cos B)
What projection is used for D-fructose?
So here’s the final challenge. Given the Fischer projection, try drawing the Haworth projection ( -furanose form) for D-fructose, below:
What is the D-ribose converted into?
Here’s D-ribose converted into its -furanose Haworth projection. [Notice that the C1-OH is on the opposite face of the molecule from the C4– substituent (CH2OH), which makes the configuration alpha (α) ]
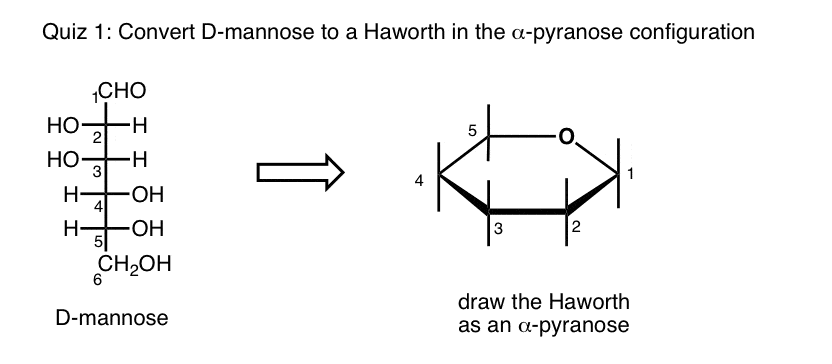
What is mannose hexose?
Mannose is a hexose where the configuration at C2 is flipped relative to glucose.
What side of the Haworth does a group end up on?
This gives us the following shortcut. If a group is on the left side of the Fischer, it will end up on the top face of the Haworth, and if it is on the right side of the Fischer, it will end up on the bottom face of the Haworth.
What bond breaks the C 1 -O pi bond?
This sets us up to draw the C 5 -OH attacking the carbonyl carbon, forming a new O–C 1 single bond and breaking the C 1 -O pi bond.
What is the term for C-1 carbon?
The standard terminology for these two configurations at the C-1 carbon (sometimes called the “anomeric carbon”) uses the terms alpha (α) and beta (β).
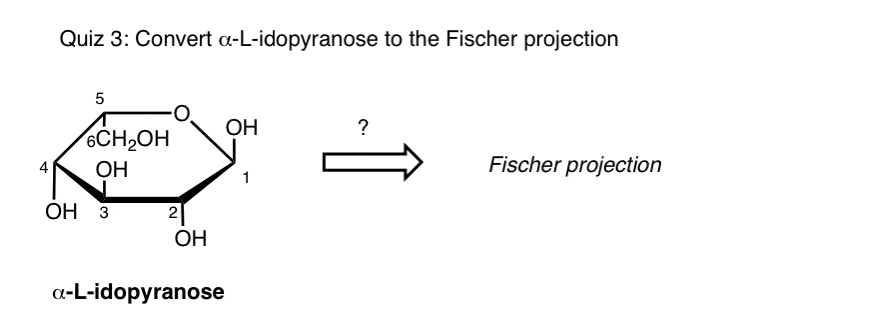
Can you convert a Fischer to a Haworth?
Voila – there’s your conversion of a Fischer to a Haworth. If you’re keen, you can even turn the Haworth into a chair, if you start with a template and map all the substituents as we described in the last post.
What do the horizontal and vertical lines mean?
I know it is hard to believe, but it is written this way for a reason. Here is the typical way you will see the Fischer projection written in your textbook.
Start by tipping your molecule to the right
Alright…no matter your carbohydrate structure, start by tipping it over to the right. When you do this, this will put your aldehyde (CHO) group on the right side.
Is your OH group pointing up or down?
So – here is the cool part. Once you have tipped your structure, look at your OH groups.
What projection is used for D-fructose?
So here’s the final challenge. Given the Fischer projection, try drawing the Haworth projection ( β-furanose form) for D-fructose, below:
What is the D-ribose converted into?
Here’s D-ribose converted into its α-furanose Haworth projection. [Notice that the C1-OH is on the opposite face of the molecule from the C4– substituent (CH2OH), which makes the configuration alpha (α) ]
What is mannose hexose?
Mannose is a hexose where the configuration at C2 is flipped relative to glucose.
What side of the Haworth does a group end up on?
This gives us the following shortcut. If a group is on the left side of the Fischer, it will end up on the top face of the Haworth, and if it is on the right side of the Fischer, it will end up on the bottom face of the Haworth.
What bond breaks the C 1 -O pi bond?
This sets us up to draw the C 5 -OH attacking the carbonyl carbon, forming a new O–C 1 single bond and breaking the C 1 -O pi bond.
Which groups point up in the Haworth?
Notice that all the groups that were pointing “up” in our linear molecule (e.g. the C3-OH) also point “up” in the Haworth, and all the groups that point “down” in the linear molecule (e.g. C2-OH and C4-OH) point “down” in the Haworth.
Is Fischer bond flat?
Although all the bonds in the Fischer might be drawn “flat”, it’s not meant to be understood that way! [ See this earlier post on D and L sugars, for example].
What side of the molecule is the methyl group on?
Let’s look at the molecule from the right side. This will put the CN and Br groups as wedge lines, the OH and Cl as dash lines. The zig-zag will have the methyl on the bottom-left side and the ethyl group on the right side respectively:
How many o rotations do you need for a zig zag?
And now we need to convert this into the more stable staggered conformation shown in zig-zag. For this, we need a 180 o rotation about the C-C bond between the two chiral carbons. This puts the methyl group (on the rightmost) up and the OH with the Br pointing down changing their wedge and dash notation:
How to convert a Newman projection to a bond line?
So, first, decide the direction you are going to use. It can be any – right or left unless specified in the question.
What is the application of Fischer projections?
An important application of Fischer projections is the ease of conveying the stereochemistry of carbohydrates and their conversion from Fischer, Haworth, and chair structures.
How many wedge lines are there in a molecule?
There are two wedge and two dash lines which may look strange to you since we always have one of each and then the two solid lines, but it is okay-it all depends on the direction we are looking at the molecule.
Which side of the bond line is the top group?
When doing so, the top group ( aldehyde) is going to be the left side of the bond-line structure. Keep in mind that the horizontal lines are pointing towards you in the Fischer projection. Notice that they are also pointing to the viewer on the side which means the H and the OH are going to be wedge lines when we look at the structure from the left (this is not the final zig-zag structure yet – we are only looking at the molecule from a different direction):
Is a Newman projection the same as a bond line?
Regardless of the molecule shown in bond-line, Newman, or Fischer projection, it is still the same molecule! And therefore, it must have the same absolute configuration of all the chiral centers. This means, for example, when you are converting a Fischer projection to a bond-line, you can simply draw the zig-zag with the correct number ...
What are the most common carbohydrates in living cells?
Glucose, galactose, and mannose are among the most common carbohydrates in living cells. Also, six-membered cyclic forms (pyranoses) are also prevalent in nature, so they are very important. Finally, this is a very typical type of an exam question! So you wanna make sure you really know how to convert Fischer to Haworth and chair and back.
How many members are in pyranose?
Pyranose is a 6-membered ring that is traditionally drawn with the anomeric carbon to the right: Haworth projection of a pyranose. In this type of a representation, we assume that the bottom of the structure is oriented towards the observer. This way, we don’t have to show it with the wedges every single time.
Why is anomeric carbon important in chemistry?
The anomeric carbon has a special place in the chemistry of carbohydrates because it doesn’t have a set stereochemistry. This means that it can be either facing “up” or “down” depending on how the cyclization happened. While, there are factors that can make one orientation more favorable than the other one, I’m not going to go over those here to keep this post as much to the point as possible. If you’re curious about the stability of the anomeric -OH in the “up” or a “down” position, look up “anomeric effect” to dive into the topic deeper.
What are some examples of aldohexoses?
Aldohexoses: carbohydrates that have an aldehyde functional group and contain six carbons. Examples of aldohexoses are glucose and galactose which are essential for the living organisms on Earth. Pyranoses: six-membered cyclic structures that result from the hemiacetal formation between the aldehyde on the 1st carbon in the molecule and ...
Which carbons go to the bottom of the Fischer projection?
The “right” groups on carbons 2, 3, and 4 in the Fischer projection go onto the “bottom” positions in Haworth
What is a Fischer projection?
As you probably know, Fischer projections are a very convenient way of drawing a long-chain molecule with a lot of chiral carbons, such as a carbohydrates. In a Fischer projection, we put the most oxidized atom (an aldehyde function) on the top and the rest of the chain following from there.
Why do I keep the same color scheme?
I kept the same color scheme to make it a little easier to see what’s going on and track the groups as we go from one structure to another.