change in thermal energy = mass × specific heat capacity × change in temperature
- mass of block in example = 1.00 kg
- change in thermal energy = mass × specific heat capacity × change in temperature
What is specific heat?
How to find the heat capacity of a substance?
What is specific heat capacity?
Why is water a high specific heat?
Why does iron take more energy to heat up water than metals?
Does specific heat capacity change?
Is heat capacity given?
See 2 more
What does C stand for in Q MC ∆ T?
The amount of heat gained or lost by a sample (q) can be calculated using the equation q = mcΔT, where m is the mass of the sample, c is the specific heat, and ΔT is the temperature change.
What is the formula to calculate specific heat?
The formula for specific heat capacity, C , of a substance with mass m , is C = Q /(m ⨉ ΔT) . Where Q is the energy added and ΔT is the change in temperature.
What is the easiest way to calculate specific heat?
2:544:38Thermodynamics: Specific Heat Capacity Calculations - YouTubeYouTubeStart of suggested clipEnd of suggested clipHundred and seventy-five joules of heat to that specific heat through that piece of silver and we'reMoreHundred and seventy-five joules of heat to that specific heat through that piece of silver and we're gonna change its that what that will change its temperature by forty three point six degrees. And
What is Q MC ∆ H?
0:083:355.1 Calculating ΔH using q = mcΔT (SL) - YouTubeYouTubeStart of suggested clipEnd of suggested clipSo in this video we'll be using the equation Q equals MC delta T where Q is heat in joules M is massMoreSo in this video we'll be using the equation Q equals MC delta T where Q is heat in joules M is mass in grams and C is the specific heat capacity.
What is the unit for specific heat?
The specific heat capacity is defined as the quantity of heat (J) absorbed per unit mass (kg) of the material when its temperature increases 1 K (or 1 °C), and its units are J/(kg K) or J/(kg °C).
How do you solve specific heat problems?
0:0110:53Solving specific heat problems - YouTubeYouTubeStart of suggested clipEnd of suggested clipOkay specific key problems. There are three the equation looks like this Q equals M times C timesMoreOkay specific key problems. There are three the equation looks like this Q equals M times C times delta T or if you'd rather Q equals M times C times temperature final minus temperature initial that's
What is Q in specific heat formula?
where Q is the quantity of heat transferred to or from the object, m is the mass of the object, C is the specific heat capacity of the material the object is composed of, and ΔT is the resulting temperature change of the object.
How do you find the specific heat of an experiment?
Steps to determine the specific heat capacity.Place a beaker on a balance and press zero.Now add the oil to the beaker and record the mass of the oil.Read the starting temperature of the oil.Connect a joulemeter to the immersion heater.Time for thirty minutes.More items...
How do you find the specific heat capacity of a metal?
Use Q = sm∆T to determine the heat capacity of the metal. (Make sure to use the heat given off by the metal, the mass of the metal, and the temperature change of the metal in this calculation.) 1.
What are the 3 formulas of heat?
Different formulas for HeatH = (VI)t.H = (I 2 R)t.H = (V 2 /R)t.
Is Q the same as Delta H?
Q is the energy transfer due to thermal reactions such as heating water, cooking, etc. anywhere where there is a heat transfer. You can say that Q (Heat) is energy in transit. Enthalpy (Delta H), on the other hand, is the state of the system, the total heat content.
What unit is Q in Q MC ∆ T?
In the equation Q=mcΔt: Q= the heat energy (Joules), m= the mass of the object/substance being heated (in this case it's water; also important in this situation to remember that 1ml of water is equal to 1g of water), c= the specific heat of the object/substance being heated (again…
How do you calculate specific heat in chemistry?
9:5851:14Specific Heat Capacity Problems & Calculations - Chemistry TutorialYouTubeStart of suggested clipEnd of suggested clipAnd specific heat capacity heat capacity is calculated by taking the energy change or the energyMoreAnd specific heat capacity heat capacity is calculated by taking the energy change or the energy absorbed or released by a substance. And dividing it by the change in temperature that occurs. So heat
What is Q in specific heat formula?
The amount of heat is represented by "Q". The amount of heat is represented by "J", or Joules. "T" is the temperature of the substance. Specific heat is represented by "Cp".
How do you calculate the specific heat of a metal?
Use Q = sm∆T to determine the heat capacity of the metal. (Make sure to use the heat given off by the metal, the mass of the metal, and the temperature change of the metal in this calculation.)
What is the formula for specific latent heat?
Calculations involving specific latent heat The specific latent heat of water is: l f = 3 ⋅ 34 × 10 5 J k g − 1 for fusion (solid→liquid) or freezing (liquid→solid) l v = 22 ⋅ 64 × 10 5 J k g − 1 for vaporisation (liquid→gas) or condensation (gas→liquid)
How to find specific heat?
The formula is: Cp = Q/mΔT .
What is specific heat?
Specific heat is the amount of energy required to raise one gram of a pure substance by one degree Centigrade. The specific heat of a substance is dependent on both its molecular structure and its phase. The discovery of specific heat sparked the studies of thermodynamics, the study of energy conversion involving heat and the work of a system.
What is the formula for heat supplied?
The formula is: heat supplied= heat capacity/rise in temperature. Thus, heat capacity is = mass of the object or sample.
What is the amount of heat represented by?
The amount of heat is represented by "Q". The amount of heat is represented by "J", or Joules.
Why does metal heat up faster than water?
Metal heats up faster than water because of its low specific heat.
Can temperature be removed from equation?
That's not possible. Q=mass × specific heat capacity x temperature is the formula, temperature cannot be removed from the equation.
Can you solve the heat problem if it is not given?
You will have to use clues to find out if the heat is given. If not, it cannot be solved.
Using Specific Heat Capacity to Find Temperature Change
Step 1: Identify the mass and the specific heat capacity of the substance.
Using Specific Heat Capacity to Find Temperature Change Vocabulary and Formula
Specific heat capacity: the energy required to raise the temperature of one gram of a substance by one degree Celsius.
Using Specific Heat Capacity to Find Temperature Change Example
Find the temperature change for a 100 g piece of copper that gives off 1,925 J of energy in a calorimeter. The specific heat capacity of copper is {eq}0.385 \frac {J} {g\: ^ {\circ}C} {/eq}.
Using Specific Heat Capacity to Find Temperature Change Example
Find the temperature change for a 75.0 g piece of glass that absorbs 5,400 J of energy. The specific heat capacity of glass is {eq}0.753 \frac {J} {g\: ^ {\circ}C} {/eq}.
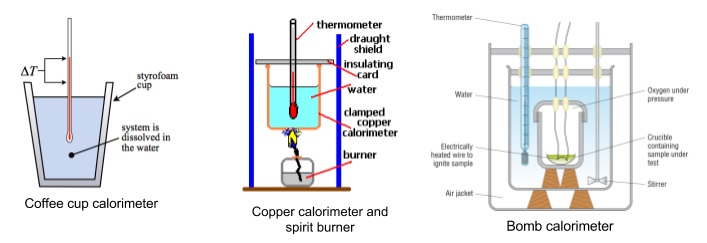
What is the study of heat transfer during physical and chemical changes?
Calorimetry is the study of heat transfer during physical and chemical changes. During calorimetry experiments, we can’t measure the heat that’s transferred directly. But we can measure the change in temperature. We can use equations like the one shown to relate the observed change in temperature to the heat transferred.
Is heat a subject?
In the equation as written, heat is the subject. The subject is the isolated term in a mathematical formula. It is the variable that is being solved for. To answer this question, we need to make 𝑐 the subject. We can do this by rearranging the formula. Before rearranging the formula, we need to understand two rules.
How are specific heat constants related to gas constants?
The specific heat constants for constant pressure and constant volume processes are related to the gas constant for a given gas. This rather remarkable result has been derived from thermodynamic relations, which are based on observations of physical systems and processes. Using the kinetic theory of gases, this same result can be derived from considerations of the conservation of energy at a molecular level.
What is the Greek symbol for specific heat ratio?
We can define an additional variable called the specific heat ratio , which is given the Greek symbol "gamma", which is equal to cp divided by cv:
What does delta h mean?
Let's denote the change by the Greek letter delta which looks like a triangle. So "delta h" means the change of "h" from state 1 to state 2 during a process. Then, for a constant pressure process the enthalpy equation becomes: The enthalpy, internal energy, and volume are all changed, but the pressure remains the same.
Which equation is most convenient to use when dealing with a gas?
If we are dealing with a gas, it is most convenient to use forms of the thermodynamics equations based on the enthalpy of the gas. From the definition of enthalpy:
What is thermodynamics in aerodynamics?
Thermodynamics is a branch of physics which deals with the energy and work of a system. Thermodynamics deals only with the large scale response of a system which we can observe and measure in experiments. In aerodynamics, we are most interested in thermodynamics for the role it plays in engine design and high speed flight .
Does enthalpy change with pressure?
The enthalpy, internal energy, and volume are all changed, but the pressure remains the same. From our derivation of the enthalpy equation , the change of specific enthalpy is equal to the heat transfer for a constant pressure process:
What is the formula for specific heat?
The formula for specific heat capacity, C, of a substance with mass m, is C = Q / (m ⨉ ΔT). Where Q is the energy added and ΔT is the change in temperature. The specific heat capacity during different processes, such as constant volume, Cv and constant pressure, Cp, are related to each other by the specific heat ratio, ɣ= Cp/Cv, or the gas constant R = Cp - Cv.
How to calculate specific heat capacity?
Find the initial and final temperature as well as the mass of the sample and energy supplied.
What is specific heat capacity at constant volume?
The specific heat capacity is the heat or energy required to change one unit mass of a substance of a constant volume by 1 °C. The formula is Cv = Q / (ΔT ⨉ m).
What is the specific heat capacity value of water?
The specific heat of water is 4179 J/kg K, the amount of heat required to raise the temperature of 1 g of water by 1 Kelvin.
How to cool down a sample?
Insert the amount of energy supplied as a positive value. If you want to cool down the sample, insert the subtracted energy as a negative value. For example, say that we want to reduce the sample's thermal energy by 63,000 J. Then Q = -63,000 J.
How much heat does aluminum need?
The specific heat of aluminum is 897 J/kg K. This value is almost 2.3 times of the specific heat of copper. You can use this value to estimate the energy required to heat a 500 g of aluminum by 5 °C, i.e., Q = m x Cp x ΔT = 0.5 * 897* 5 = 2242.5 J.
What is the unit of measurement for specific heat?
Specific heat is measured in BTU / lb °F in imperial units and in J/kg K in SI units.
What is specific heat?
The specific heat (also called specific heat capacity) is an important property of materials because it tells us how much energy will be needed to heat or cool a certain substance. With that information we can know long the cooling or heating process will take, and how much it will cost.
How to find the heat capacity of a substance?
So, if you want to calculate the heat capacity of a substance, you simply multiply the specific heat capacity times the mass of the substance . The heat capacity is written with a capital letter as such: C p.
What is specific heat capacity?
But that shouldn’t be a surprise, since the definition of the specific heat capacity is the amount of heat energy Q that is needed to change the temperature of 1kg by 1 degree, and those are exactly the values that we are given.
Why is water a high specific heat?
The fact that water has a high specific heat means that it is able to retain much more heat without the temperature raising too much, which enables oceans to absorb energy and helps to stabilize Earth’s habitable conditions. This also enables warm-blooded animals, like us humans, to be able to maintain our body temperatures, since we are 60% water.
Why does iron take more energy to heat up water than metals?
This is why it takes much more energy to heat up water than metals. This can be easily seen when we boil water. After turning the stove on, it can be seen that the pot itself gets hot very quickly, much faster than the water in it.
Does specific heat capacity change?
As we can see, we get the same answer as in Example 1, since the specific heat capacity of a substance (in this case water) does not change.
Is heat capacity given?
Very often the value for the heat capacity is not given, but you have to look it up on a table, like the one on the left. Here it is very important to beware of the units.