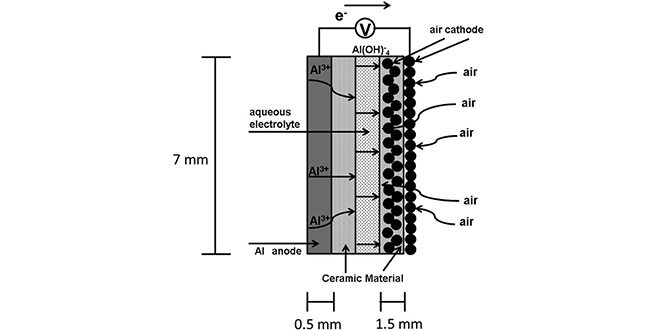
How Does an Aluminum air Battery Work?
- The air from the atmosphere gets sucked into the air cathode system, which contains a catalyst.
- oxygen gets separated from the air and reacts with water to generate hydroxide (OH+) ions
- At the anode, which is made of pure virgin aluminum plate, the ionic oxygen (O+) reacts with aluminum trihydroxy-ide [ AI (OH)2].
Why aluminium air batteries?
Aluminum air battery’s high energy density mean that they have a high potential of being used in electrical vehicles. Making an aluminum air battery is quite simple – and can be done using simple household goods.
What is the energy density of aluminum air batteries?
Aluminum–air batteries are remarkable due to their high energy density (8.1 kWh kg −1 ), light weight (2.71 g cm −3 ), environmentally friendly, good recyclability, and low cost [137,138]. Aluminum–air batteries consist of an aluminum anode, an air cathode and an electrolyte which is salty, alkaline, and nonaqueous solutions.
What is the difference between lithium-ion and aluminium batteries?
This has restricted their use to mainly military applications. However, an electric vehicle with aluminium batteries has the potential for up to eight times the range of a lithium-ion battery with a significantly lower total weight. Aluminium–air batteries are primary cells, i.e., non-rechargeable.
How to make a simple aluminum air battery?
Procedure of Making Simple Aluminum Air Battery. Just take a piece of aluminum foil and spread it on a table. In a pot make a saturated solution of water and salt. Take a piece of bloating paper. Get the piece of bloating paper soaked by saturated salt solution. Then get the soaked piece of bloating paper spread over the aluminum foil.
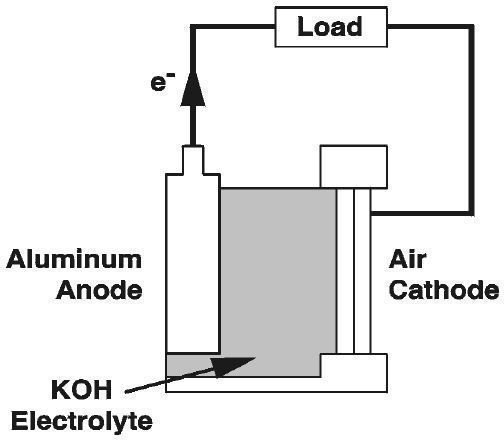
Are aluminum air batteries rechargeable?
Aluminium–air batteries are primary cells, i.e., non-rechargeable. Once the aluminium anode is consumed by its reaction with atmospheric oxygen at a cathode immersed in a water-based electrolyte to form hydrated aluminium oxide, the battery will no longer produce electricity.
How does metal-air battery work?
A metal-air battery uses some type of metal (like aluminum) for the anode, air as the cathode, along with a liquid electrolyte. In the case of aluminum, oxygen from the air then combines with the metal to create aluminum hydroxide, which activates the electrolysis process and creates a current.
What are the disadvantages of aluminum air battery?
Despite its low cost, simple operation, and reduced environmental impact, aluminum batteries based on aqueous or protic systems exhibit fatal drawbacks, such as the passivating oxide film formation decreasing the battery voltage and efficiency, hydrogen side reactions, and material corrosion.
How long does Aluminium air battery last?
The result is an aluminum-air prototype with a much longer shelf life than that of conventional aluminum-air batteries. The researchers showed that when the battery was repeatedly used and then put on standby for one to two days, the MIT design lasted 24 days, while the conventional design lasted for only three.
How do you recharge Aluminium air battery?
The newly invented aluminum-air battery can work just by refilling salty water or normal water once in a while. Aluminum is an abundant, cheap and safe material which can be applied for metal-air batteries.
Can metal-air batteries be recharged?
Metal-air batteries can be visualized as a fuel cell which uses metal as fuel. A simple metal-air battery contains a metal anode, an air cathode which is open to air and a suitable electrolyte. In order to be used as an energy storage device, the battery must be electrically rechargeable.
How can I make Aluminium air battery at home?
0:5810:23How To Make an Aluminum Air Battery - YouTubeYouTubeStart of suggested clipEnd of suggested clipI chose to go with powdered graphite i'm going to use some paint to mix with the graphite powder soMoreI chose to go with powdered graphite i'm going to use some paint to mix with the graphite powder so it doesn't get everywhere. And i can easily apply it next i need some aluminum for the anode.
Can aluminum air battery be recycled?
Battery Recycling Legislation A depleted aluminum-air battery is fully recyclable, allowing for the emergence of commercial enterprises that remanufacture such batteries for continual re-use in transportation propulsion and stationary energy storage applications.
Can Aluminium air batteries outperform Li ion for EVS?
6:0810:33Can Aluminum Air Batteries Outperform Lithium Ion ... - YouTubeYouTubeStart of suggested clipEnd of suggested clipPower aluminum air has big advantages over a lithium-ion battery the favorite choice for evs.MorePower aluminum air has big advantages over a lithium-ion battery the favorite choice for evs.
Can aluminium replace lithium?
Aluminum Battery Technology Could Replace Lithium When a cell recharges, aluminum ions return to the negative electrode and exchange three electrons per ion, lithium does the same exchange but at the rate of only one per second.
Who invented Aluminium air battery?
Trevor JacksonOne of them is the aluminium-air (Al-air) power source. Trevor Jackson, an engineer and former officer in the UK's Royal Navy, began experimenting with Al-air batteries in 2001 at his modest workshop in Callington, Cornwall.
Where are aluminum air batteries used?
Sometimes known as “Metal-Air” batteries, these have been successfully used in “off-grid” applications for many years, just as batteries powering army radios. The most attractive metal in this type of battery is aluminum because it is the most common metal on Earth and has one of the highest energy densities.
How do you make a metal air battery?
15:0416:17Homemade Metal Air Battery - Part 1 - YouTubeYouTubeStart of suggested clipEnd of suggested clipAnd potassium hydroxide one mole of potassium hydroxide instead of this one you can also use someMoreAnd potassium hydroxide one mole of potassium hydroxide instead of this one you can also use some sodium hydroxide and in this video i show you the aluminium air battery iron air battery.
What is a metal cell battery?
Lithium metal batteries are lithium batteries with an anode from lithium metal. The term 'lithium metal batteries' came into use only after lithium-ion batteries had been invented. Most lithium metal batteries are non-rechargeable. However, rechargeable lithium metal batteries are also under development.
What is the metal used in batteries?
Nickel, cobalt, and lithium as battery raw materials Nickel, cobalt and lithium are key metals used in today's active cathode materials and the chemistries deployed in high performance batteries.
What is the best electrolyte for a battery?
For batteries, you should select an electrolyte that includes an element used in one or both of the half-cells. For example, if one of the half-cell reactions is with copper, a good choice of an electrolyte is CuCO3 or CuCl2. Both of these will support the half-cell by ensuring that there are Cu2+ ions in solution.
How many mA does an aluminum battery generate?
This large reaction area makes it possible for the simple aluminum–air battery to generate 1 volt (1 V) and 100 milliamps (100 mA). This is enough power to run a small electrical device and provides a safe and easy way to make a powerful battery at home or in school.
How to make an air cell out of aluminum?
Add a heaping spoonful of activated charcoal on top of the paper towel, then gently crush the charcoal into fine bits using the back of the spoon. Pour some of the salt-water solution onto the charcoal until it is dampened throughout . Make sure the charcoal doesn't touch the foil directly; you should have three distinct layers, like a sandwich. This is your aluminum–air cell.
What happens when the foil from one cell is in contact with the foil from the cell above it?
If the foil from one cell is in contact with the foil from the cell above it, the electrons will bypass the paper towel and activated charcoal and move directly into the second piece of foil, which has a lower resistance than the charcoal layer.
How to make a battery?
To make a voltaic pile, repeat Assembly steps 1–4 to construct additional aluminum–air cells. Stack two or three aluminum–air cells on top of each other to see if you can make a more powerful battery. Clip one lead to the bottom piece of foil and place the other lead in the top charcoal pile. Press down firmly on the pile to reduce the internal resistance of the battery, but make sure that the foil pieces don’t touch each other. If the foil from one cell is in contact with the foil from the cell above it, the electrons will bypass the paper towel and activated charcoal and move directly into the second piece of foil, which has a lower resistance than the charcoal layer. This effectively shorts out the lower cell, which no longer contributes to the overall power output.
How to get battery to work on a clip?
If the battery doesn’t seem to be working after a few seconds, you may need to reduce its internal resistance. Try increasing the contact area between the clip and the charcoal by folding the entire battery over the clip—like a taco—and pressing down hard. Make sure that the clip stays buried in the charcoal. If you are using a motor, you can also try kickstarting it by briefly spinning the flag.
How does a battery generate electricity?
To generate electrical energy, this battery relies on oxidation of aluminum at the anode, which releases electrons, and a reduction of oxygen at the cathode, which uses electrons. The movement of electrons through an external circuit generates an electric current that can be used to power simple devices.
What can you use to make a battery?
Use aluminum foil, salt water, and activated charcoal to construct a simple battery strong enough to power a small motor or light. Grade Bands:
How to use aluminum foil on cell?
Connect the copper foil on the top and the aluminum foil on the bottom with clip leads to an LED and/or electric meter. Wet the exposed paper towel on each cell with the salt electrolyte solution until it will not absorb more .
Where are aluminum cells housed?
Individual aluminum cells are housed in old pill bottles and connected in series.
What is used as an insulator between layers in a multiple cell pile?
Schematic of multiple cell pile using copper foil on styrofoam which is used as an insulator between layers.
What is a model pouch cell made of?
A model pouch cell has layers consisting of aluminum, felt with charcoal, copper tape and an insulator made from styrofoam food trays.
Why is the power output of an aluminum battery so large?
The power output is large because of the high performance of the air electrode and the high conductivity of the electrolyte.
What is the electrolyte solution for aluminum air batteries?
Neutral and alkaline aqueous solutions are applicable as the electrolyte solution of an aluminum–air battery, and the performance strongly depends on the choice of electrolyte.
What is the energy of lithium?
At a nominal potential of about 3 V, the theoretical specific energy for a lithium/air battery is over 5000 Wh kg −1 for the reaction forming lithium hydroxide (LiOH) (Li+1/4 O 2 +1/2 H 2 O=LiOH) and 11 000 Wh kg −1 for the reaction forming lithium peroxide (Li 2 O 2) (Li+O 2 =Li 2 O 2) or for the reaction of lithium with dissolved oxygen in seawater, rivaling the energy density for hydrocarbon fuel cells and far exceeding Li-ion battery chemistry that has a theoretical specific energy of about 400 Wh kg −1. The high specific energy for metal/air chemistries has been long recognized, as evidenced by the development and commercial success of the Zn/air battery. However, lithium/air chemistry introduces some rather unique challenges. Lithium is a reactive alkali metal and is incompatible with conventional aqueous electrolytes typically used for oxygen electrochemistry. Corrosion of the negative electrode due to reaction of the metal anode with water and oxygen in aqueous electrolytes is a well-recognized problem for zinc/air (and aluminum/air batteries), but it is clearly a much more serious issue for lithium metal. The corrosion rate for lithium metal in aqueous electrolytes is on the order of several A cm −2 for neutral aqueous electrolytes and drops to tens of mA cm −2 in highly basic media. Accordingly, with the exception of some early work by E. Littauer on lithium/water batteries, the historical development of lithium/air batteries has been dominated by the use of aprotic nonaqueous electrolytes. Recently, the invention of solid electrolyte protected lithium electrodes (PLEs) has lifted this restriction for lithium/air chemistry to include the use of a broad range of aggressive protic and aprotic solvents, including aqueous electrolytes. Prior to the use of PLEs, lithium/air battery development was confined to the use of aprotic nonaqueous electrolytes that form a passivating solid electrolyte interphase (SEI) on the surface of lithium metal ( Figure 1 ). The high energy density of metal/air systems is derived in large part from the availability of oxygen in ambient air, and except under extremely dry conditions ambient air has significant levels of moisture. In some cases, developers have used dry oxygen instead of air in the positive electrode to limit lithium corrosion, but this is impractical and adversely affects the energy density of the battery. Although the use of nonaqueous electrolyte may temporarily stabilize the lithium electrode in a lithium/air cell, the presence of moisture in the cathode air stream will ultimately lead to ingress of water into the nonaqueous electrolyte and life-limiting lithium corrosion. Some researchers have attempted to delay this process by coating the lithium metal electrode with a polymer electrolyte, but most, if not all, polymer electrolytes provide a poor barrier to moisture. The use of nonaqueous electrolytes poses another dilemma for lithium/air chemistry in that the discharge product is highly insoluble lithium peroxide, Li 2 O 2 and Li 2 O, which may precipitate in the pore space of the gas diffusion electrode (GDE), thereby impeding the discharge reaction and severely limiting the capacity and energy density of the nonaqueous lithium/air battery. To date, the majority of published discharge curves for nonaqueous lithium/air batteries are cathode limited most likely due to precipitation and poor utilization of the cathode. The use of exotic nonaqueous or protic solvents might increase the solubility of lithium peroxide, but for conventional lithium electrodes, this would lead to rapid corrosion of the anode. Accordingly, the development of lithium/air batteries might have remained a laboratory curiosity had it not been for the development of water-stable PLEs, allowing the use of nonconventional media such as protic and aqueous electrolytes. In order to develop the PLE, water-stable high-conductivity Li + solid electrolytes are required.
What are the problems with Al-air batteries?
Certain problems exist that are intrinsic to the battery, including anode corrosion, pore blockage, sluggish ORR for non-noble cathodes, carbonation in aqueous electrolytes, the lack of compelling reports for viable secondary Al–air batteries and the absence of a defined method for replacing the anode in a primary battery. Further research on Al–air battery needs to focus on optimizing the combination of cathode, anode, electrolyte and other battery components that will allow ease of scalability, good performance, and economic production.
Why is aluminum battery thermally unstable?
This reaction is exothermic and the battery system often requires thermal management to suppress possible electrolyte boiling. The low actual voltage (large overvoltage) also contributes to the thermal instability of the aluminum–air battery.
What is an iron air battery?
Iron–air battery is a type of metal–air battery that combines metallic negative electrode having a low redox potential with air positive electrode. The basic components of iron–air battery are iron negative electrode, air positive electrode, and alkaline electrolyte. The theoretical voltage for this battery system is based on Fe (II)/Fe (0) followed by Fe (III)/Fe (II) redox reaction at the negative electrode and the oxygen reduction at the positive electrode is around 1.28 V. Therefore, the theoretical specific energy is around 1000 Wh kg −1. Although the iron–air battery system has these charge–discharge features and abundance of electrode material, it is a minor candidate as a primary battery compared to zinc–air battery or aluminum–air battery since both the expected capacity and the expected working voltage of iron negative electrode are lower than those of zinc and aluminum. Moreover, as described in this article, the electrode reaction of iron is difficult in some ways compared to zinc. Therefore, presently, almost all commercial metal–air batteries include zinc negative electrode, and commercial iron–air primary battery is very difficult to be found.
What elements are used to prevent corrosion of aluminum?
The important candidates are gallium, indium, magnesium, thallium, and tin. Bismuth, manganese, lead , and zinc are also reported to be effective. A trace amount of iron is considered to be detrimental with respect to the corrosion. The corrosion behavior also depends on the alloying method and heat treatment condition. An excessively stable surface film degrades the discharge rate performance of the battery.
Is aluminum air battery better than lithium?
They're also cheaper and safer than lithium-ion batteries. With that said, there's a major caveat.
Will there be an EV battery breakthrough?
We could honestly probably report on an EV battery breakthrough every week, though most of these aren't actually breakthroughs, but technology and chemistries that have been in the testing stages for years. However, we'd like to think that eventually, many new battery technologies will come to the forefront. If new tech and advancements weren't bound to happen, we probably wouldn't have lithium-ion batteries or electric cars in the first place.
Can aluminum batteries be recycled?
These aluminum-oxygen batteries can't be recharged. Instead, drivers will head to a gas station and quickly swap them. The used batteries will then be recycled. They're also reportedly less toxic than other batteries, as well as easier to recycle. Visit the Autoblog link below for more details.

Overview
Commercialization
Aluminium as a "fuel" for vehicles has been studied by Yang and Knickle. In 2002, they concluded:
The Al/air battery system can generate enough energy and power for driving ranges and acceleration similar to gasoline powered cars...the cost of aluminium as an anode can be as low as US$ 1.1/kg as long as the reaction product is recycled. The total fuel efficiency during the cycle process in Al/air electric vehicles (EVs) can be 15% (present stage) or 20% (projected), compara…
Electrochemistry
The anode oxidation half-reaction is Al + 3OH → Al(OH) 3 + 3e -2.31 V.
The cathode reduction half-reaction is O 2 + 2H 2O + 4e → 4OH +0.40 V.
The total reaction is 4Al + 3O 2 + 6H 2O → 4Al(OH) 3 +2.71 V.
About 1.2 volts potential difference is created by these reactions and is achievable in practice when potassium hydroxide is used as the electrolyte. Saltwater electrolyte achieves approximat…
See also
• List of battery types
• Zinc–air battery
• Potassium-ion battery
• Metal–air electrochemical cell
• Aluminum-ion battery
External links
• Aluminum battery from Stanford offers safe alternative to conventional batteries
• Aluminium battery can charge phone in one minute, scientists say
• Simple homemade aluminum-air battery