How is benzoic acid prepared from ethyl benzoate Benzoic acid, C₇H₆O₂, is a colorless crystalline solid and a simple aromatic carboxylic acid. The name is derived from gum benzoin, which was for a long time its only known source. Benzoic acid occurs naturally in many plants and serves as an intermediate in the biosynthesis of many second…Benzoic acid
How to prepare ethyl benzoate on a microscale?
Microscale preparation of ethyl benzoate In association with Nuffield Foundation No comments Try this class practical to prepare the ester ethyl benzoate on a microscale by warming ethanol and benzoic acid In this experiment, students mix benzoic acid and ethanol in a plastic pipette, before warming the mixture in a water bath.
How do you reflux ethyl benzoate and sodium hydroxide?
A mixture of ethyl benzoate (2 ml) and sodium hydroxide solution (15 ml, 10%) is refluxed in a round bottomed flask fitted with a water condenser on a water bath (temp. 90-100 oC) for about 30 minutes till the ester layer disappears. Then the solution is cooled and acidified with HCl.
What is the yield of separated benzoic acid precipitate?
The separated benzoic acid precipitate is filtered and recrystallised from hot water. Yield: 1.2 g, m.p.: 122 oC. Molecular formula of ethyl benzoate = C 9H 10O 2. Molecular formula of benzoic acid = C 7H 6O 2.
What is the use of benzoic acid in skin care?
Aim: To prepare benzoic acid from alkyl benzoate by Hydrolysis reaction. Use: It is used as a topical agent with salicylic acid to treat skin problems like irritation and inflammation which may cause by burns, insect bites, fungal infections or eczema.
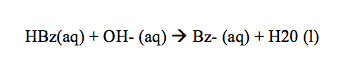
How is benzoic acid obtained from ethyl benzoate?
(i) Benzoic acid from ethyl benzoate : When an ethyl benzoate is heated with dil. H2SO4, undergoes hydrolysis to form benzoic acid and ethyl alcohol.
How do you make benzoic acid from benzoate?
In all cases, the benzoic acid is further refined to produce sodium benzoate. One way this is done is by dissolving the acid in a sodium hydroxide solution. The resulting chemical reaction produces sodium benzoate and water. The crystals are isolated by evaporating off the water.
How is benzoic acid prepared?
How can benzoic acid be prepared? Industrially, benzoic acid can be prepared by employing oxygen gas for the partial oxidation of toluene. It can be noted that this process usually employs manganese or cobalt naphthenate as catalysts. This compound can also be prepared via the hydrolysis of benzamide and benzonitrile.
How is ethyl benzoate prepared?
50 g benzoic acid are dissolved in 100 g absolute alcohol, 10 g concentrated sulphuric acid are added, and the mixture is boiled under a reflux condenser for 4 hours.
How is benzoic acid prepared in the lab?
A mixture of 5 ml (or 5.1 g) of benzonitrile and 75 ml of 10% aqueous sodium hydroxide is boiled under a reflux condenser until oily drops of benzonitrile disappears. The condenser is then detached and boiled in the open flask for a few minutes in order to remove free ammonia.
How do you make benzoic acid from benzyl alcohol?
- We can use an acidic, neutral or alkaline solution of KMnO4 to oxidise any primary alcohol to carboxylic acid. So, we can use alkaline potassium permanganate solution to convert benzyl alcohol to benzoic acid.
How is benzoic acid prepared from Grignard reagent write the preparation of benzoic acid from dry ice?
c) Using dry ice to prepare benzoic acid. When dry ice or solid carbon dioxide CO2 is added to a solution of phenyl magnesium iodide in dry ether, an adduct is formed. The adduct formed is a magnesium salt of carboxylic acid. When this adduct is hydrolyzed in the presence of an acid, it gives benzoic acid.
Where does benzoic acid come from?
Its most common natural source is gum benzoin, a resin found in the bark of trees of the genus Styrax. Most benzoic acid produced today is synthetic. Its first industrial synthesis was the hydrolysis of benzotrichloride to calcium benzoate, followed by acidification.
How is benzoic acid prepared from Grignard reagent?
1 Answer. When phenyl magnesium bromide is treated with dry ice (solid carbon dioxide) in the presence dry ether, complex is obtained which on acidification gives benzoic acid.
What is the common name of ethyl benzoate?
As with many volatile esters, ethyl benzoate has a pleasant odor described as sweet, wintergreen, fruity, medicinal, cherry, pish and grape. It is a component of some fragrances and artificial fruit flavors....Ethyl benzoate.NamesPreferred IUPAC name Ethyl benzoateIdentifiersCAS Number93-89-03D model (JSmol)Interactive image31 more rows
How is benzoic acid obtained from phenyl ethene?
1 Answer. When phenyl ethene is heated with strong oxidising agents like acidic KMnO4 or acidic K2Cr2O7, benzoic acid is obtained.
What products are formed when ethyl benzoate is treated with water and h2so4?
Therefore, the final product of the reaction of ethyl benzoate with water and sulfuric acid in presence of heat is benzoic acid.
What is benzoic acid used for?
Use: It is used as a topical agent with salicylic acid to treat skin problems like irritation and inflammation which may cause by burns, insect bites, fungal infections or eczema.
How are esters hydrolysed?
Principle: Esters are hydrolysed either by an acid or a base. Alkaline hydrolysis of ester is irreversible which is also called saponification. Acid hydrolysis of ester is a reversible reaction. Acid hydrolysis of esters can occur by more than one type of mechanism, the common mechanism is: The alkaline hydrolysis, which occurs through a nucleophilic acyl substitution. Here Ethyl benzoate on hydrolysis with sodium hydroxide gives benzoic acid and ethyl alcohol where OH– ion of sodium hydroxide act as a nucleophile.1
Teaching notes
After the practical, you may wish to build a class activity around writing an equation for the reaction that has occurred during the experiment.
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry. This collection of over 200 practical activities demonstrates a wide range of chemical concepts and processes.
