
How elements are formed
- Elements and the ‘Big Bang’ theory. During the formation of the universe some 14 billion years ago in the so-called ‘Big...
- Elements and our Sun. For most of their lives, stars fuse elemental hydrogen into helium in their cores. Two atoms of...
- Dying stars. When a star’s core runs out of hydrogen, the star begins to die out. The dying star expands...
How were naturally elements formed?
Elements and the ‘Big Bang’ theory. During the formation of the universe some 14 billion years ago in the so-called ‘Big Bang’, only the lightest elements were formed – hydrogen and helium along with trace amounts of lithium and beryllium. As the cloud of cosmic dust and gases from the Big Bang cooled, stars formed, and these then grouped together to form galaxies.
How elements found in the universe were formed?
• There are three cosmic stages through which specific groups of elements were formed. (1) The big bang nucleosynthesis formed the light elements (H, He, and Li). (2) Stellar formation and evolution formed the elements heavier than Be to Fe. (3) Stellar explosion , or supernova, formed the elements heavier than Fe.
What are the seven elements of nature?
The 7 Elements In Practice In addition to what we might think of as the classical elements: earth, water, fire, air, and gas (space), in many ancient eastern concepts of self there are an additional two: consciousness (or thought itself) and the element of spirit .
What are the 5 elements of the Earth?
What are the Qualities of the 5 Elements of Nature?
- Warm and Cold. Every component is either warm or cold, and this relates to a male or female sexual orientation. ...
- Moist and Dry. The second pair of characteristics is dampness or dryness. ...
- Elements of Opposite Nature. Since every element imparts one of its characteristics to one other element, which leaves one element random.
How are heavy elements formed?
Heavy elements can be formed from light ones by nuclear fusion reactions; these are nuclear reactions in which atomic nuclei merge together. The simplest reactions involve hydrogen, whose nucleus consists only of a single proton, but other fusion reactions, involving mergers of heavier nuclei, are also possible. When the universe formed in an initial state of very high temperature and density, the big bang, the first elements to exist were the simplest, primarily hydrogen and helium (two protons). But we, and Earth, and all other objects including elements other than hydrogen and helium, are made of heavier elements, so a major question for scientists is how these heavier elements were created.
How are elements identified?
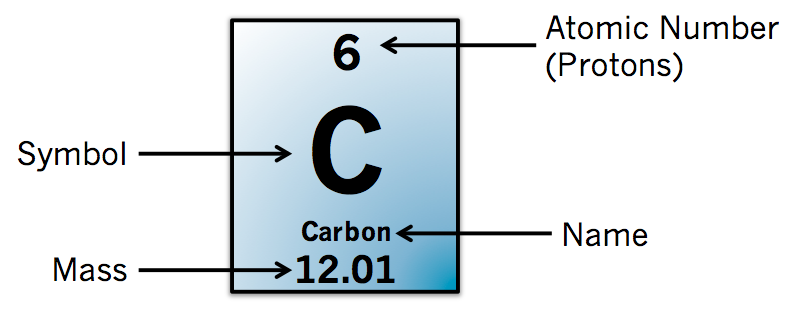
Elements are identified by the number of protons in the nuclei of their atoms. For example, an atom having six protons in its nucleus is carbon, and one having 26 protons is iron. There are over 80 naturally occurring elements, with uranium (92 protons) being the heaviest (heavier nuclei have been produced in reactors). Nuclei also contain certain neutrons, usually in numbers greater than the number of protons. The number of neutrons in the atoms of a given element varies.
How are elements heavier than iron made?
How then are elements heavier than iron made? There are two processes, both triggered by the addition of neutrons to atomic nuclei: the s (slow) process and the r (rapid) process. In both processes, a nucleus captures a neutron, which emits an electron and decays into a proton, a reaction called a beta decay. One proton at a time, these processes build up elements heavier than iron. Some elements can be made by either process, but the s process can only make elements up to bismuth (83 protons) on the periodic table. Elements heavier than bismuth require the r process.
What were the elements that were found in the Big Bang?
During the formation of the universe in the big bang, only the lightest elements were formed: hydrogen, helium, lithium, and beryllium. Hydrogen and helium dominated; lithium and beryllium were only made in trace quantities. The other 88 elements found in nature were created in nuclear reactions in the hearts of stars and in the huge stellar explosions known as supernovas. Stars like the sun and planets like Earth containing elements other than hydrogen and helium could only form after the first generation of massive stars exploded as supernovas, and scattered the atoms of heavy elements throughout the galaxy to be recycled.
What happens when a star's core runs out of hydrogen?
When the star ’ s core runs out of hydrogen, the star begins to die out. The processes that occur during this period form the heavier elements.
What happens to the stars during a supernova?
During a supernova, the star releases as much energy as the sun does in 10 billion years and also releases the large number of neutrons needed for the r process, creating new elements during the outburst. The elements that were made during the red giant stage, and those that are made during the supernova
How do stars produce iron?
Massive stars produce iron and the lighter elements by the fusion reactions described above, as well as by the subsequent radioactive decay of unstable isotopes. Elements heavier than iron are more difficult to make, however. Unlike nuclear fusion of elements lighter than iron, in which energy is released, nuclear fusion of elements heavier than iron requires energy. Thus, the reactions in a star ’ s core stop once the process reaches the formation of iron.
Why are elements in ionic form?
The elements are in ionic form because the universe is still very hot—too hot to form atoms. Approximately 380,000 years after the Big Bang was the epoch of recombination. After years of expanding and cooling the universe was finally ready for the nuclei to capture the electrons.
Why are the elements in the universe ionic?
The elements are in ionic form because the universe is still very hot— too hot to form atoms.
How does a star spend 90% of its lifetime fusing hydrogen into helium?
A star spends 90% of its lifetime fusing hydrogen into helium. After the hydrogen has been depleted, it begins fusing helium into higher elements. With each new stage of elemental fusion, the core becomes denser, and the outer layers of the stars begin to expand, gradually turning into a red giant.
How does a star burn fuel?
A star spends 90% of its lifetime fusing hydrogen into helium.
How did stars keep their cores from collapsing?
To keep their cores from collapsing under gravity, the stars needed to tap into a constant source of energy. This energy was eagerly provided by the release of binding energy.
What is the process of annihilating a proton and antiproton?
These collisions produce particle (protons) and antiparticles (anti-protons) in a process called pair formation. The universe continuously churned out more and more of these pairs. On the other hand, these proton and anti-proton pairs were annihilating each other and transforming back into photons and radiation.
What is the meaning of nucleosynthesis?
Nucleosynthesis and the life of stars. Formation of elements heavier than iron. Delivery to Earth. Conclusion. Everything that has ever existed or ever will exist has been made possible by some permutation or combination of the elements found on a periodic table. That colorful array of elements holds an entire universe of information.
How are elements formed?
Elements are formed by the gravitational forces in stars causing the fusion of lighter elements into the heavier ones.
Can we fuse elements in a laboratory?
We can fuse limited elements in laboratory experiments (and fusion nuclear bombs!), but it is energetically very unfavorable, so we must use the elements found in nature.
Where are elements formed?
Elements are formed deep within the cores of certain types of star. Find out more in this interactive.
Where are the elements we come into contact with?
Apart from hydrogen and helium, formed just after the Big Bang, most of the elements we come into contact with have been made in the stars. Planet Earth and all of its life forms are made of elements formed billions of years ago deep within the cores of stars now long dead. Acknowledgement: MediaCollege.com.
What are the elements in a planetary nebula?
Elements such as helium, carbon, oxygen, nitrogen, neon and smaller amounts of heavier elements are present. Planetary nebulae play an important part in the chemical evolution of the galaxy, allowing these elements to be returned to the interstellar medium. The remains of the carbon core of a red giant evolve into a white dwarf star. The Eskimo Nebula in Gemini is a good example.
How did the solar system form?
Scientists believe that the Solar System was formed when a cloud of gas and dust in space, made up of elements formed deep within stars, started to collapse, forming a solar nebula. This began to spin as it collapsed, eventually giving birth over billions of years to the Solar System as we know it today.
What happens to the outer region of a supernova?
The supernova is the final stage in the life of massive stars. The outer region of the star collapses and it Instantly rebounds off the inner core in a cataclysmic explosion.
How many different processes are involved in forming elements?
There are actually 19 different processes involved with forming the elements and those 19 are just the ones that we are aware of…there may be more.
How do stars form new elements?
Nuclear fusion makes element form. Stars create new elements by squeezing elements together in a process of nuclear fusion. First, stars fuse hydrogen atoms into helium. Helium atoms then fuse to create beryllium, and so on, until fusion in the star's core has created every element up to iron.
What happens to hydrogen in the first stage of fusion?
In the first stage, hydrogen collide and form lithium then it collide with hydrogen and forms helium then helium and lithium collide to form heavier hydrogen. After thousands of fusions of gases ( don't know how scientist keep this formula in mind ) heavier element like carbon and oxygen form.
What happens when nuclear fusion is accomplished?
If this is accomplished, the furnace of nuclear fusion is ignited. In nuclear fusion, smaller atomic nuclei are fused together to form a larger nucleus. For example, hydrogens fuse to form a helium nucleus. This is the first step and is why the elements in the universe today are 99% hydrogen and helium.
Why do stars lose energy?
The stars began to lose energy when they must use more energy to form larger elements due to the lack of hydrogen.
How many processes are there in matter?
Some of the 19 processes I refer to are everything from Hydrogen and Helium fusion to the triple-alpha process, neutron capture, the s-process, proton capture, and photodisintegration. Those are just a few ways matter is created. While it
Why is hydrogen a potential star?
The hydrogen was not uniformly distributed in the early universe and gravity did what gravity does and pulled together hydrogen into potential stars. I say “potential” because the mass must be great enough to have the compressive pressure great enough to produce the kinds of temperatures great enough to result in temperatures in the tens ...
What elements were formed in the early universe?
The early universe (left) was too hot for electrons to remain bound to atoms. The first elements — hydrogen and helium — couldn’t form until the universe had cooled enough to allow their nuclei to capture electrons (right), about 380,000 years after the Big Bang.
When did protons and neutrons form?
Protons and neutrons began forming shortly after, from about 10-6 to 1 second after the Big Bang. Within about 3 minutes after the Big Bang, conditions cooled enough for these protons and neutrons to form hydrogen nuclei. This is called the era of nucleosynthesis.
How long did it take for hydrogen to form?
Hydrogen didn’t appear until the universe had spread out — and subsequently cooled — enough for the first protons and neutrons, and later simple atoms, to form. Between about 10-12 and 10-6 second after the Big Bang, neutrinos, quarks, and electrons formed. Protons and neutrons began forming shortly after, from about 10-6 to 1 second after ...

History
Formation of Elements
- During most of their lives, stars fuse hydrogen into helium in their cores, but the fusion process rarely stops at this point; most of the helium in the universe was made during the initial big bang. When the star’s core runs out of hydrogen, the star begins to die out. The processes that occur during this period form the heavier elements. The dyin...
Manufacturing Heavy Elements
- How then are elements heavier than iron made? There are two processes, both triggered by the addition of neutrons to atomic nuclei: the s (slow) process and the r (rapid) process. In both processes, a nucleus captures a neutron, which emits an electron and decays into a proton, a reaction called a beta decay. One proton at a time, these processes build up elements heavier t…
Key Terms
- Beta decay—The splitting of a neutron into a proton and an electron. Fusion—The conversion of nuclei of two or more lighter elements into one nucleus of a heavier element. r process—Rapid process, the process by which some elements heavier than iron are made in a supernova. Red giant—An extremely large star that is red because of its relatively cool surface. s process—Slo…
Resources
- BOOKS
McWilliam, Andrew and Michael Rauch. Origin and Evolution of the Elements. Cambridge, UK: Cambridge UniversityPress, 2004. - PERIODICALS
Hester, J. Jeff, et al. “The Cradle of the Solar System.” Science. 304 (2004): 1116-1117. Kirshner, Robert. “The Earth’s Elements.” Scientific American(October 1994): 59. Nittler, Larry R. “Nuclear Fossils in Stardust.” Science. 303 (2004): 636-637.
The Appearance of Matter
The Appearance of Protons and Anti-Protons
- When the universe was slightly less than 0.0001 seconds old, it started experiencing a new form of disturbance. The cosmic energy, which had been hanging around as high-energy radiation, began colliding with each other. These collisions produce particle (protons) and antiparticles (anti-protons) in a process called pair formation. The universe continuously churned out more and mo…
The Appearance of Neutrons
- The rapidly expanding universecaused some protons to smash against electrons, giving birth to neutrons (for every 7 protons, there is 1 neutron). At this point, the universe was a few seconds older and much colder (only one billion Kelvin). The protons and neutrons came together to form the nucleus/ion of the first element Hydrogen (H), which further fused with another hydrogen nuc…
Nucleosynthesis and The Life of Stars
- Over the course of time, the universe further cooled down, dense gas clouds came together due to gravity, and created the first star-forming regions. As the clouds got smashed together, they started forming hot and heavy cores that didn’t want to get any bigger. The hot core started burning itself to prevent even more clouds from clumping together. Thus began a contest betwe…
Formation of Elements Heavier Than Iron
- Remember the last few seconds and the shockwave we just mentioned? As the star is dying and exploding into a supernova, it releases a huge amount of energy (the temperature rises to billions of Kelvins) and a very dense cloud of neutrons.
Delivery to Earth
- The universe is a gargantuan recycling bin; it recycles and reuses every piece of matter that was once created by a process called chemical enrichment. Millions of galaxies, stars, and planets were formed and will continue to form using the same primordial matter left over after the Big Bang. The young universe was ¾ Hydrogen and ¼ Helium, while the rest of the matter was negl…
Conclusion
- Believe it or not, but all the atoms in our bodies are older than the solar system itself! They were created in a series of events following the one event that started it all 13.8 billion years ago. The gold in our jewelry and the zinc in our batteries were created in the final moments of a star’s life. The oxygen and carbon in our soda, the iron in our blood, and the calcium in our teeth were forg…
Suggested Reading
- Chemical Evolution Across Space and Time: From the Big Bang to Prebiotic Chemistry: 981 (ACS Symposium Series) Check on Amazon
- Midnight in Chernobyl: The Untold Story of the World’s Greatest Nuclear Disaster Check on Amazon
- Caveat Emptor Check on Amazon