What is the molecular weight of sodium polyacrylate?
0.54 g/mL at 25 °C (lit.) Sodium Polyacrylate (ASAP) is an acrylic sodium salt based hydrophilic polymer with carbonyl and sodium as major groups present in the structure. Both groups facilitate the polymeric chain with absorption potential.
What is sodium polyacrylate water absorbent resin?
Sodium polyacrylate water-absorbent resin is a new type of functional polymer material widely developed and researched at home and abroad in recent years. It is a kind of strong hydrophilic polymer compound with a loose network structure and low cross-linking degree.
Is sodium polyacrylate acidic or alkaline?
Generally speaking, sodium polyacrylate is weakly acidic because it has very little acrylic residue. (Sodium polyacrylate is formed by the polymerization reaction of acrylic acid and sodium hydroxide. ) As the strongest water-absorbing material at present, sodium polyacrylate can absorb up to several thousand times its own weight of pure water.
What is sodium polyacrylate (SAP)?
Sodium polyacrylate is a kind of super absorbent polymer (also called SAP). It is a functional polymer used in a variety of common products such as paper diapers, pets pads, water-retaining material (to help the soil retain water), instant snow, and so on. It is known for its superior absorbency:

What are the properties of sodium polyacrylate?
Sodium polyacrylate (SO-dee-um pol-ee-AK-ruh-late) is an odorless, grainy white powder. Its most impressive property is its ability to absorb large amounts of fluid, up to 800 times its volume of distilled water and lesser amounts of other liquid mixtures.
Is sodium polyacrylate water soluble?
Sodium polyacrylate is soluble in water. Crosslinked sodium polyacrylate, such as found in diapers will swell in water. This is driven by the dissociation of the sodium carboxylate salt in the water and the hydrogen bonding interactions between the water and the polymer.
Is sodium polyacrylate polar or nonpolar?
The Sodium Polyacrylate polymer, shown below in blue, contains a polar charge that allows it to combine with other liquids by forming hydrogen bonds.
Is acrylic acid hydrophobic or hydrophilic?
Most hydrophilic polymers are grouped by the chemistry of their structure. For example, acrylics include acrylic acid, acrylamide, and maleic anhydride polymers and copolymers.
What happens when sodium polyacrylate is added to water?
Sodium polyacrylate contains a high number of sodium ions within each particle. Water is highly attracted to sodium ions. So when the water is poured into the beaker containing the sodium polyacrylate, it moves into the individual powder particles and expands the polymer particles to become a solid like gel.
Why can sodium polyacrylate absorb water?
Sodium polyacrylate is an example of a super-absorbing polymer. It is a cross-linked (network) polymer that contains sodium atoms. It absorbs water by a process called osmosis.
Can sodium polyacrylate absorb oil?
FAQ. Does the SAP absorbs petroleum products? No. Sodium polyacrylate is designed for absorbing water-based liquids.
What happens when you add salt to sodium polyacrylate?
Salt causes water to leak out of the sodium polyacrylate because it disrupts the attraction between water and the polymer chain. When salt is added, it breaks up into negative and positive ions, which are also attracted to the water molecules.
What can sodium polyacrylate be used for?
Sodium polyacrylate is an absorbent polymer that is used as an emulsion stabilizer, a hair fixative, a film former, skin conditioner, and a viscosity agent [1]. You will see them as the gel like crystals in baby diapers, which are makes them absorbent.
How do you know if its hydrophobic or hydrophilic?
Something defined as hydrophilic is actually attracted to water, while something that is hydrophobic resists water. This means when hydrophobic items come in contact with liquids, water is encouraged to bead up and roll off the surface- almost pushing it away like a magnet pushes away metal objects.
How do you know if a compound is hydrophobic or hydrophilic?
If all the bonds in a molecule are nonpolar, then the molecule itself is nonpolar. Some examples of nonpolar covalent bonds are C-C and C-H bonds. 2. Even if a molecule has polar covalent bonds, if these bonds are arranged symmetrically, the molecule overall will be hydrophobic.
How do you know if a bond is hydrophobic or hydrophilic?
1:364:01Hydrophilic vs. Hydrophobic - YouTubeYouTubeStart of suggested clipEnd of suggested clipSo if you look if you look at this molecule. You can see that double bonded oxygen and that OAHMoreSo if you look if you look at this molecule. You can see that double bonded oxygen and that OAH group that hydroxide group on the right hand side and then you can see the amine group the nitrogen.
What is mixed with acrylic acid?
But some kind of sodium polyacrylate is mixed up with small amounts of acrylic acid, a leftover from the manufacturing process.
What is the best absorbent material?
Sodium polyacrylate – the superior absorbency material. Sodium polyacrylate is a functional polymer used in a variety of common products such as paper diapers, pets pads, water-retaining material (to help the soil retain water), instant snow, and so on. It is known for its superior absorbency:
What are water beads made of?
Water beads are made of SAP. It can absorb a large amount of water and turn into a bigger ball.
Is sodium polyacrylate safe?
Occupational Safety and Health Administration that list potential hazards of chemicals in great detail), sodium polyacrylate is totally safe.
Can SOCO polymer be used as a controlled release agent?
-It with fibrous shapes potentially can be used to make high.#N#-Can be used as an internal sealant in cement-based materials.#N#-A special use of SOCO Polymer in concrete could be as a controlled release agent.
Does sodium polyacrylate absorb water?
It is known for its superior absorbency: Sodium polyacrylate can absorb hundreds times its own weight in water. It starts out as a powder and as it comes into contact with moisture, it swells into its gel form. Unlike other absorbent materials, it’s not easy to squeeze the moisture out of this gel.
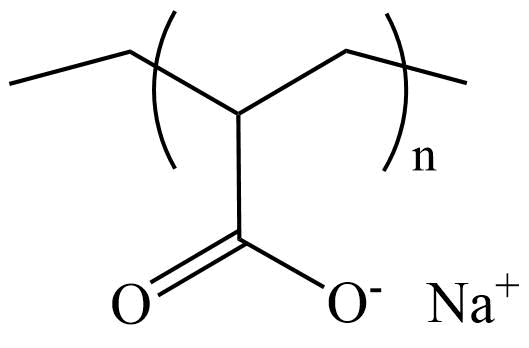
What is sodium polyacrylate?
Sodium Polyacrylate. Definition. Sodium Polyacrylate is a new type of functional polymer material and an important chemical product. Its solid product is white (or light yellow) lumps or powder, and the liquid product is a colorless (or light yellow) viscous liquid.
How is methyl acrylate formed?
First, methyl acrylate is formed by the reaction of acrylic acid and methanol, and the suspension or latex after polymerization of methyl acrylate is heated in an aqueous solution of sodium hydroxide to prepare sodium polyacrylate. 4. Hydrolysis method.
What is the process of aggregation of acrylic acid and caustic soda?
1. Aggregation method. Firstly, acrylic acid and caustic soda are reacted to produce sodium acrylate monomer, and then the monomer is polymerized into sodium polyacrylate under the trigger of persulfate and reducing agent. 2.
What is the role of water based paint?
Water-based paint, play a dispersing role ; Textile printing and dyeing auxiliary, play the role of slurry dispersion; Auxiliary of ceramic processing, which plays a dispersing role; As a washing aid in detergents, it has a dispersion effect; anti-fouling redeposition effect and thickening effect;
How is acrylic acid made?
First, acrylic acid is polymerized into polyacrylic acid under the action of a redox agent, and then polyacrylic acid is neutralized with caustic soda to produce sodium polyacrylate. 3. Saponification method.
Is acrylamide hydrolyzed?
First, acrylamide is polymerized to produce polyacrylamide, and then polyacrylamide is hydrolyzed to produce sodium polyacrylate under alkaline conditions. Source: Organized from the Internet. Reference materials: 1. Sodium polyacrylate. chemicalbook. 2. Synthesis of sodium polyacrylate and its research progress.
General description
Sodium Polyacrylate (ASAP) is an acrylic sodium salt based hydrophilic polymer with carbonyl and sodium as major groups present in the structure. Both groups facilitate the polymeric chain with absorption potential. It can be used as a superabsorbent material as, in the presence of a liquid, the ASAP chain swells and forms a gel substance.
Application
Absorbent for aqueous-based spills. Retention and slow release aid for water.
Features and Benefits
Superabsorbent polymer. Absorbs many times its weight of aqueous fluids.
Abstract
Sodium polyacrylate (PAAS) intercalated layered double hydroxide (LDH) composites were fabricated and applied for removal of methylene blue (MB), Pb 2+ and Hg 2+ ions in aqueous solution. The properties of PAAS/LDH, such as surface area, surface charge and hydrophilicity, were investigated.
1. Introduction
With the development of industrialization, a serious environmental problem might occur by the discharge of wastewater containing toxic organic and inorganic pollutants.
2. Materials and methods
Aluminum nitrate, magnesium nitrate, sodium hydroxide, lead nitrate, lead nitrate, ammonium persulfate and methylene blue (MB) were all AR grade. Low-molecular weight sodium polyacrylate (molecular weight: 1250, 2500 and 5100) was purchased from Aladdin Co. (China).
3. Results and discussions
The X-ray diffraction pattern of LDH and PAAS/LDH with different molecular weight of PAAS were presented in Fig. 1 (a). The pristine LDH had typical layered double hydroxide structures with sharp and symmetric (003), (006) and (009) planes.
4. Conclusion
LDH was intercalated with PAAS to be used as an effective adsorbent for simultaneous adsorption of MB, Pb 2+ and Hg 2+ in aqueous solution. The successful intercalation of PAAS was confirmed by XRD, FTIR and elemental analysis.
CRediT authorship contribution statement
Meng Chen: Writing - original draft, Methodology, Conceptualization. Ran Bi: Data curation, Software. Ran Zhang: Funding acquisition, Writing - review & editing. Feng Yang: Funding acquisition, Editing. Feifei Chen: Supervision, Conceptualization, Writing - original draft, Writing - review & editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
What are the basic soil properties of RB and SPB?
Basic soil properties of RB and SPB, including liquid limit, plastic limit, dry density, free swell index (FSI), permeability coefficient (k) and cation exchange capacity ( CEC) are present in Table 2. Dry density is determined by a wax seal method, while the liquid plastic limit is determined by a combined liquid plastic apparatus. Both RB and SPB samples were subjected to the free swelling test using the volume change according to ASTM D5890. The hydraulic conductivity test was performed using flexible-wall permeameters according to ASTM D5084, “Standard test method for the measurement of the hydraulic conductivity of saturated porous materials using a flexible wall permeameter. A flexible-wall membrane cell and associated hydraulic control system similar to that of Kang and Shackelford was used in their study [24]. The CEC values could be calculated and measured according to the literature [7] and following formula [9]: (14) CEC ( m e q / 100 g) = 50 c w × 10 - 5 where c is the measured concentration of ammonia (mol/L), 50 mL is the volume of water, w is the mass of the air-dried clay sample (mg), and 10 −5 is a conversion factor.
What is the thermodynamic stability of RB and SPB?
The thermodynamic stability of RB and SPB has been investigated by thermogravimetric analysis (TGA), and the DTG curve is the first derivative with respect to temperature, which was shown in Fig. 4. The first weight loss for the RB was approximately 1% in the temperature 161 °C, and for the SPB, it did not happen in this temperature range. This weight loss in both minerals is attributed to the physisorbed water found between their interlayers and the hydration water of the exchangeable cations (non-structural water) [34], [8]. Hence, it can be determined that the amount of adsorbed water decreased or was at a low level, when the bentonite was modified with the polymer (sodium polyacrylate), which indicates that the hydrophilic nature of the bentonite sheets changed and the bentonite sheets may be adjacent to the hydrophobic side of the polyacrylate sodium. The second weight increase for the RB was about 2.5% in the temperature of 365 °C, and that for the SPB continued decreasing. This phenomenon is caused by the oxidation reaction of metal compounds in RB at this temperature, which increases part of the mass. However, owing to the packaging of polymers and the acidification and washing during the synthesis process, the metal compounds in SPB are relatively few and not easy to oxidize with oxygen. Furthermore, the weight loss of RB in the temperature range between 380 and 700 °C corresponds to the hydroxyl groups at the end of the sheets of the bentonite, which are associated with the internal structure of the sheets. However, the weight loss of SPB in the temperature range between 285 and 645 °C, except corresponding to the dehydroxylation of the sheets of the bentonite, corresponds to the beginning of the decomposition of the organic chains of the polyacrylate sodium [20]. And this process does not occur in the RB; hence, the last weight loss for RB was approximately 19.1% and this was less than that of SPB for 23.4%. Organic matter is completely converted to carbon dioxide at the high temperature of 700 °C.
What is the effect of temperature on sorption rate of RB and SPB?
The sorption percentage of RB and SPB for Pb (II) was higher at higher temperatures, which suggested that the process of adsorption interaction was an endothermic process. When the temperature increases from 15 to 40 °C, the sorption rate of the RB increases by more than 12%; however, the sorption rate of the SPB increases by less than 5% in both low and high concentrations, which indicates that the SPB is better than RB in terms of temperature stability.
