See more
How is thallium determined?
Thallium is determined in natural waters by electrothermal atomic absorption spectrometry after precon centration. Thallium is oxidized and retained as the tetrachlorothallate (III) ion on an anion exchange column, followed by elution with ammonium sulfite solution. A concentration factor of 400 is achieved.
What is HSDB in chemistry?
Hazardous Substances Data Bank (HSDB) The thallium is usually precipitated and recovered as a sparingly soluble salt (cementation thallium and thallium amalgam) from liquors produced in a variety of concentration methods, with and without pretreatment, and also from liquors that are themselves the raw materials.
Where is thallium obtained?
In the past, thallium was obtained as a by-product from smelting other metals; however, it has not been produced in the United States since 1984. Currently, all the thallium is obtained from imports and from thallium reserves..In its pure form, thallium is odorless and tasteless.
What is the best matrix modifier for thallium?
Palladium, about 5-10 ug as Pd (NO3)2, is confirmed as an effective matrix modifier for thallium. A pyrolysis temperature of 1000 °C can be used, which is a much higher temperature than could be used with the previously proposed matrix modifier, H2SO4. The recovery of thallium was quite good in the presence of large amounts of NaCl but was inadequate in the presence of KCl. Abandoning the pyrolysis step and the matrix modifier proved to be convenient for determining thallium in both an NaCl and a KCl matrix. Thallium was fully recovered in these matrices in the presence of about 30 ug of either salt, or about 0.15%, assuming a 20 mul sample.
How to treat a contaminated eye?
If patient is not breathing, start artificial respiration, preferably with a demand-valve resuscitator, bag-valve-mask device, or pocket mask, as trained. Perform CPR as necessary. Immediately flush contaminated eyes with gently flowing water. Do not induce vomiting. If vomiting occurs, lean patient forward or place on left side (head-down position, if possible) to maintain an open airway and prevent aspiration. Keep patient quiet and maintain normal body temperature. Obtain medical attention. / Thallium and Related compounds/
How to dispose of thallium?
1. Ventilate area of spill. ... 2. For small quantities, sweep onto paper or other suitable material, place in sealed container for disposal. Large quantities may be reclaimed; However, if this is not practical, collect spilled material in the most convenient & safe manner & deposit in sealed containers for disposal. 3. Liquids containing sol thallium compounds may be absorbed in vermiculite, dry sand, earth, or a similar material & deposited in sealed containers for disposal. / Thallium and thallium cmpd/
What is the atomic number of thallium?
Thallium is a naturally occurring trace metalloid element with atomic symbol Tl, atomic number 81 , and atomic weight 204.383. Thallium is used in manufacturing, as well as in some chemical reactions and medical procedures. Use as a pesticide has been banned in the United States due to toxicity from accidental exposure. It is characterized as an odorless, bluish-white, metallic soft solid that turns to a gray powder with an aromatic odor when exposed to air. Exposure occurs by inhalation of fumes, ingestion, or contact.
What is the colorless, more stable thallous ion?
In water the colourless, more stable thallous ion, Tl +, resembles the heavier alkali metal ions and silver; the compounds of thallium in its +3 state are easily reduced to compounds of the metal in its +1 state.
What is the name of the material that emits light when X-rays are absorbed?
1 percent thallium have been found to emit light when X-rays are absorbed in the material. These devices are known as scintillators, and when used in conjunction with a photomultiplier tube they can easily detect the burst of light from a single X-ray photon. Furthermore, the amount….
How many electrons are in Boron?
They are characterized as a group by having three electrons in the outermost parts of their atomic structure. Boron, the lightest of these elements, is a metalloid. Aluminum, gallium, indium, and thallium are silvery white metals. Nihonium has only been….
What is the heaviest element in the boron group?
Natural thallium, the heaviest of the boron group elements, consists almost entirely of a mixture of two stable isotopes: thallium-203 (29.5 percent) and thallium-205 (70.5 percent).
What are the two forms of thallium?
Two crystalline forms of the element are known: close-packed hexagonal below about 230 °C (450 °F) and body-centred cubic above. Natural thallium, the heaviest of the boron group elements, consists almost entirely of a mixture of two stable isotopes: thallium-203 (29.5 percent) and thallium-205 (70.5 percent). Traces of several short-lived isotopes occur as decay products in the three natural radioactive disintegration series: thallium-206 and thallium-210 (uranium series), thallium-208 (thorium series), and thallium-207 (actinium series).
Is thallium a metal?
Thallium (Tl), chemical element, metal of main Group 13 (IIIa, or boron group) of the periodic table, poisonous and of limited commercial value. Like lead, thallium is a soft, low-melting element of low tensile strength. Freshly cut thallium has a metallic lustre that dulls to bluish gray upon exposure to air.
How many elements are in the periodic table?
The periodic table is made up of 118 elements. How well do you know their symbols? In this quiz you’ll be shown all 118 chemical symbols, and you’ll need to choose the name of the chemical element that each one represents.
What is thallium used for?
Most thallium is used by the electronics industry in photoelectric cells. Thallium oxide is used to produce special glass with a high index of refraction, and also low melting glass that becomes fluid at about 125K. An alloy of mercury containing 8% thallium has a melting point 20°C lower than mercury alone.
What does the image of thallium mean?
Image explanation. The image reflects the origin of the element’s name (from Greek ‘thallos’, meaning ‘a green shoot or twig’), its toxicity and its use in the manufacture of reflective glass. Appearance. A soft, silvery-white metal that tarnishes easily. Uses. The use of thallium is limited as it is a toxic element.
How many radioisotopes are there in the universe?
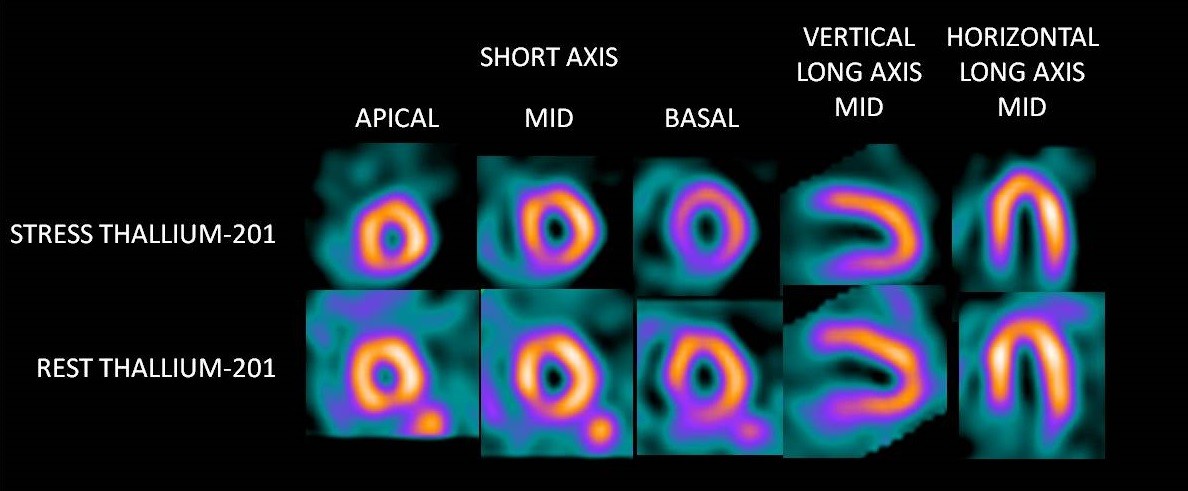
Alongside the two stable isotopes, there are a further 23 radioisotopes, though most of them with fleeting half lives. One of them, thallium 201, is useful in nuclear medicine. Its injected into the bloodstream and will find its way into all tissues with the help of the sodium/potassium membrane pump.
What is the oxidation state of an atom?
The oxidation state of an atom is a measure of the degree of oxidation of an atom. It is defined as being the charge that an atom would have if all bonds were ionic. Uncombined elements have an oxidation state of 0. The sum of the oxidation states within a compound or ion must equal the overall charge.
What is density in science?
Density is the mass of a substance that would fill 1 cm 3 at room temperature. Relative atomic mass. The mass of an atom relative to that of carbon-12. This is approximately the sum of the number of protons and neutrons in the nucleus.
Where is thallium found?
Thallium is found in several ores. One of these is pyrites, which is used to produce sulfuric acid. Some thallium is obtained from pyrites, but it is mainly obtained as a by-product of copper, zinc and lead refining.
How are elements organized into blocks?
Elements are organised into blocks by the orbital type in which the outer electrons are found. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The number of protons in an atom.
How many electrons are in thallium?
There are around eighty-one electrons present in the atom of thallium with an electronic configuration of [Xe] 4f 14 5d 10 6s 2 6p 1. This element can be generated artificially. It can be obtained by smelting of lead and zinc. It is generally obtained as a by-product in the production of sulphuric acid.
What is a thallium?
Thallium is not freely found in nature. It is a soft grey post- transition metal.
What state does a sulfate oxidize?
It oxidizes at +3 and +1 oxidation states forming ionic salts.
Is thalium carcinogenic?
Thallium is suspected to be carcinogenic. Hence direct contact should be avoided with the skin.
How long does thallium-202 last?
Thallium-202 (half-life 12.23 days) can be made in a cyclotron while thallium-204 (half-life 3.78 years) is made by the neutron activation of stable thallium in a nuclear reactor. In the fully ionized state, the isotope 205 Tl becomes beta-radioactive, decaying to 205 Pb, but 203 Tl remains stable.
How many isotopes does thalassium have?
Thallium ( 81 Tl) has 41 isotopes with atomic masses that range from 176 to 216. 203 Tl and 205 Tl are the only stable isotopes and 204 Tl is the most stable radioisotope with a half-life of 3.78 years. 207 Tl, with a half-life of 4.77 minutes, has the longest half-life of naturally occurring radioisotopes.
