
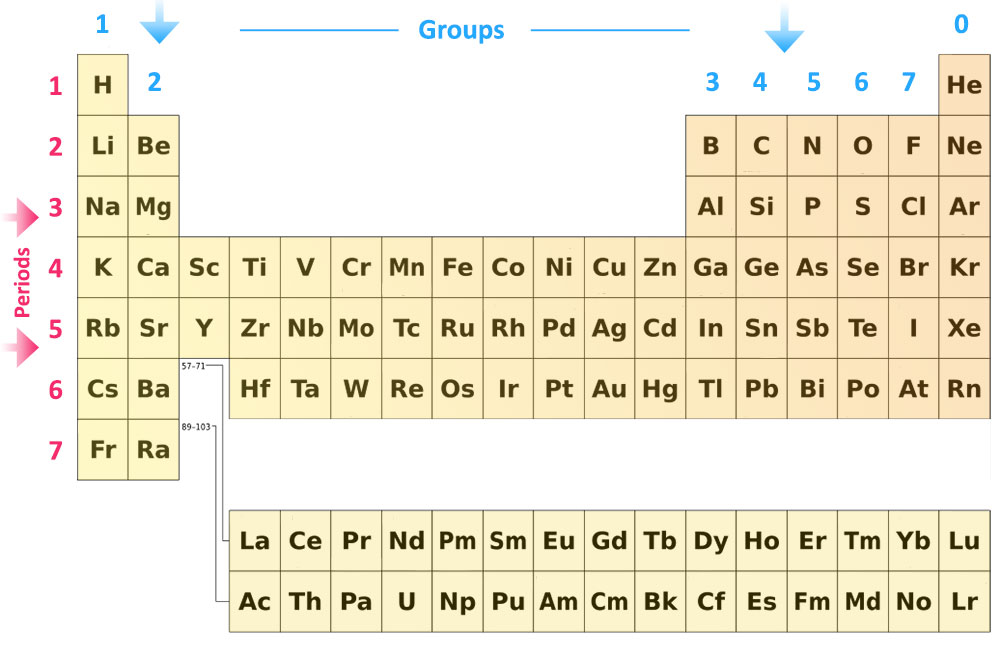
Theory: Periodic Table: It is the table of chemical elements arranged in order of atomic number such that elements with similar atomic structure appear in the vertical columns. Modern periodic table (Figure 1) is based on the modern periodic law.
Full Answer
What is the periodic table?
The periodic table is a graphic description of the periodic law, which states that the properties and atomic structures of the chemical elements are a periodic function of their atomic number.
Why are elements placed in the periodic table?
existence of elements that had yet to be discovered and corrected accepted atomic weights that were wrong Elements as Building Blocks • Periodic Table is organized like a big grid • Elements are placed in specific places because of how they look and act.
How has the periodic table changed over the years?
In the 1910s and 1920s, pioneering research into quantum mechanics led to new developments in atomic theory and small changes to the periodic table. In the 19th century, Mendeleev had already asserted that there was a fixed periodicity of eight, and expected a mathematical correlation between atomic number and chemical properties.
Who invented the periodic table of elements?
The first periodic table to become generally accepted was that of the Russian chemist Dmitri Mendeleev in 1869: he formulated the periodic law as a dependence of chemical properties on atomic mass.
What is the periodic table Theory?
Periodic Table: It is the table of chemical elements arranged in order of atomic number such that elements with similar atomic structure appear in the vertical columns. Modern periodic law: The Modern periodic law states “The chemical and physical properties of elements are a periodic function of their atomic numbers”.
Is the periodic table a law?
The Periodic Law states that the physical and chemical properties of the elements recur in a systematic and predictable way when the elements are arranged in order of increasing atomic number.
What type of science is the periodic table?
chemistryThe periodic table, also known as the periodic table of the (chemical) elements, is a tabular display of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry.
Is the periodic table a discovery or an invention?
Key Takeaways: Who Invented the Periodic Table? While Dmitri Mendeleev usually gets credit for the invention of the modern periodic table in 1869, Alexandre-Emile Béguyer de Chancourtois organized the elements by atomic weight five years earlier.
Who created the periodic law?
Dmitri MendeleevPeriodic table / InventorDmitri Ivanovich Mendeleev was a Russian chemist and inventor. He is best known for formulating the Periodic Law and creating a farsighted version of the periodic table of elements. Wikipedia
Who proposed periodic law?
A portrait of the 27-year-old Dmitri Mendeleev (Credit: Wikimedia Commons) and his 1869 handwritten version of the periodic law of the chemical elements (Credit: Science Photo Library).
How does knowing the periodic table impact the work of scientists?
Identifying patterns and predicting reactions. In fact, the periodic table is so accurate that it allows scientists to predict the chemical and physical properties of elements that hadn't yet been discovered.
How do you explain periodic table to a child?
The periodic table arranges the elements in rows and columns. In the rows, the elements are placed in order of their atomic number. The columns form groups of elements that have similar chemical properties. For example, certain gases are in one column and metals are in another.
How does the periodic table relate to science?
The periodic table of elements puts all the known elements into groups with similar properties. This makes it an important tool for chemists, nanotechnologists and other scientists. If you get to understand the periodic table, and learn to use it, you'll be able to predict how chemicals will behave.
Who arranged the periodic table?
chemist Dmitri MendeleevOne hundred fifty years after Russian chemist Dmitri Mendeleev published his system for neatly arranging the elements, the periodic table it gave birth to hangs in every chemistry classroom in the world and is one of the field's most recognizable symbols.
Why did scientists not accept Mendeleev's periodic table?
Answer and Explanation: Mendeleev's periodic table was rejected because he was unable to tell the location of a hydrogen atom in the periodic table.
Who left gaps for undiscovered elements?
MendeleevPredictions using gaps Mendeleev left gaps in his table to place elements not known at the time. By looking at the chemical properties and physical properties of the elements next to a gap, he could also predict the properties of these undiscovered elements.
What is the periodic table?
The periodic table is an arrangement of the chemical elements, structured by their atomic number, electron configuration and recurring chemical properties. In the basic form, elements are presented in order of increasing atomic number, in the reading sequence.
Who added elements to the periodic table?
Mendeleev himself added these elements to the table as group 0 in 1902, without disturbing the basic concept of the periodic table. In 1905, Swiss chemist Alfred Werner resolved the dead zone of Mendeleev 's table. He determined that the rare-earth elements ( lanthanides ), 13 of which were known, lay within that gap.
What elements were added to Mendeleev's table?
It still had the dead zone, and a row zero was added above hydrogen and helium to include coronium and the ether, which were widely believed to be elements at the time. Although the Michelson–Morley experiment in 1887 cast doubt on the possibility of a luminiferous ether as a space-filling medium, physicists set constraints for its properties. Mendeleev believed it to be a very light gas, with an atomic weight several orders of magnitude smaller than that of hydrogen. He also postulated that it would rarely interact with other elements, similar to the noble gases of his group zero, and instead permeate substances at a velocity of 2,250 kilometers (1,400 mi) per second.
What are the atomic numbers of noble gases?
In 1914, Swedish physicist Johannes Rydberg noticed that the atomic numbers of the noble gases was equal to doubled sums of squares of simple numbers: 2 = 2·1 2, 10 = 2 (1 2 + 2 2 ), 18 = 2 (1 2 + 2 2 + 2 2 ), 36 = 2 (1 2 + 2 2 + 2 2 + 3 2 ), 54 = 2 (1 2 + 2 2 + 2 2 + 3 2 + 3 2 ), 86 = 2 (1 2 + 2 2 + 2 2 + 3 2 + 3 2 + 4 2 ). This finding was accepted as an explanation of the fixed lengths of periods and led to repositioning of the noble gases from the left edge of the table, in group 0, to the right, in group VIII. Unwillingness of the noble gases to engage in chemical reaction was explained in the alluded stability of closed noble gas electron configurations; from this notion emerged the octet rule. Among the notable works that established the importance of the periodicity of eight were the valence bond theory, published in 1916 by American chemist Gilbert N. Lewis and the octet theory of chemical bonding, published in 1919 by American chemist Irving Langmuir.
What are the four elements that were found in 1900?
Four radioactive elements were known in 1900: radium, actinium, thorium, and uranium. These radioactive elements (termed "radioelements") were accordingly placed at the bottom of the periodic table, as they were known to have greater atomic weights than stable elements, although their exact order was not known.
What are the four elements that are considered elements?
The four roots, which were later renamed as elements by Plato, were earth, water, air and fire. Similar ideas about these four elements also existed in other ancient traditions, such as Indian philosophy . A few extra elements were known in the age of alchemy ( zinc, arsenic, antimony, and bismuth ).
What are the elements that are found in ancient times?
Early history. Further information: Classical element. A number of physical elements ( carbon, sulfur, iron, copper, silver, tin, gold, mercury, and lead) have been known from antiquity, as they are found in their native form and are relatively simple to mine with primitive tools.
Atomic Theory
As was written, all matter except dark matter is made of molecules, which are themselves made of atoms. The atoms are the smallest constituents of ordinary matter, which can be divided without the release of electrically charged particles. The atoms consist of two parts. An atomic nucleus and an electron cloud.
See also
We hope, this article, Atomic Theory, helps you. If so, give us a like in the sidebar. Main purpose of this website is to help the public to learn some interesting and important information about radiation and dosimeters.
Why is the periodic table important?
Mendeleev’s introduction of the periodic table of elements is one of the most important milestones in the history of chemistry, as it brought order into the known chemical and physical behaviour of the elements. The periodic table can be seen as parallel to the Standard Model in particle physics, in which the elementary particles known today can be ordered according to their intrinsic properties. The underlying fundamental theory to describe the interactions between particles comes from quantum theory or, more specifically, from quantum field theory and its inherent symmetries. In the periodic table, the elements are placed into a certain period and group based on electronic configurations that originate from the Pauli and Aufbau principles for the electrons surrounding a positively charged nucleus. This order enables us to approximately predict the chemical and physical properties of elements. Apparent anomalies can arise from relativistic effects, partial-screening phenomena (of type lanthanide contraction) and the compact size of the first shell of every l -value. Further, ambiguities in electron configurations and the breakdown of assigning a dominant configuration, owing to configuration mixing and dense spectra for the heaviest elements in the periodic table. For the short-lived transactinides, the nuclear stability becomes an important factor in chemical studies. Nuclear stability, decay rates, spectra and reaction cross sections are also important for predicting the astrophysical origin of the elements, including the production of the heavy elements beyond iron in supernova explosions or neutron-star mergers. In this Perspective, we critically analyse the periodic table of elements and the current status of theoretical predictions and origins for the heaviest elements, which combine both quantum chemistry and physics.
Who created the periodic table of the elements?
In 1869, Dmitri Ivanovich Mendeleev ordered the known elements into what he termed the ‘periodic table of the elements’ (PTE) on the basis of their increasing atomic weight and chemical similarity 1. Mendeleev’s PTE was proposed 5 years after Lothar Meyer had organized the 28 known elements into a table, of which six columns were labelled with valence number and five rows with atomic weight (see Box 1 and, for a historical account, see refs 2, 3, 4, 5, 6, 7, 8 ).
What are the two most abundant elements in the universe?
We start our discussion by mentioning the two most abundant elements in our universe, H and He, synthesized directly in the primordial nucleosynthesis roughly 10 seconds to 20 minutes after the Big Bang 45. These are placed into groups 1 and 18, respectively, although their chemical and physical behaviour is quite distinct compared with their heavier homologues in the PTE. Hydrogen has unique properties and functions: from hydrogen, we get water and biomolecules important for life on our planet. Watson–Crick pairing is a prime example in which hydrogen bonding is required for the stability of DNA 46. Furthermore, let us compare the structure of benzene and its associated rich aromatic chemistry to C 6 Li 6, which consists of ( { {rm {C}}}_ {2}^ {2-}) fragments strongly aggregated through Li bridges 47. We can also consider the recent discovery of superconductors in hydrogen-rich phases 48, 49, 50 and the search for metallic, high-pressure phases of pure hydrogen important for understanding the physics of large-sized planets such as Jupiter 51.
What are the similarities and differences between elements in the PTE?
It has become (almost) a doctrine in chemistry that elements placed in the same group of the PTE show similar chemical and physical behaviour . Similarities and differences are based on the comparison of measurable quantities, either through experimental observables or through their calculations using the corresponding quantum-mechanical (self-adjoint) operators, if necessary to the highest accuracy available within a certain theory. A detailed discussion on the numerous similarities observed is beyond the scope of this Review. For recent accounts, see refs 43, 44 and for main-group elements, see ref. 18. Instead, we focus on some irregularities and anomalies within the PTE that arise from shell-structure and relativistic effects.
What is the standard model of fundamental particles?
The Standard Model of fundamental particles serves physicists in the same way as the periodic table of elements serves chemists, thus, we can consider the Standard Model as ‘ the physicist’s periodic table’. It contains the elementary particles in nature as we currently know.
What is PTE in chemistry?
The PTE is as fundamental to chemists as the table of elementary particles is to physicists (Fig. 2 ). We all know that atoms interact to form chemical bonds. Note that the term ‘chemical bond’ is a fuzzy concept, because it does not strictly correspond to a quantum-mechanical observable.
Which rule states that electrons are assigned first to the lower n value?
The Madelung–Janet (or diagonal) rule states that, when considering consecutive neutral atoms, electrons are assigned to one-particle levels in order of increasing ( n + l) value and, for states of equal ( n + l ), the electrons are assigned first to the lower n value.

Summary
History
In 1817, German physicist Johann Wolfgang Döbereiner began to formulate one of the earliest attempts to classify the elements. In 1829, he found that he could form some of the elements into groups of three, with the members of each group having related properties. He termed these groups triads. Chlorine, bromine, and iodine formed a triad; as did calcium, strontium, and barium; lithi…
Overview
The smallest constituents of all normal matter are known as atoms. Atoms are extremely small, being about one ten-billionth of a meter across; thus their internal structure is governed by quantum mechanics. Atoms consist of a small positively charged nucleus, made of positively charged protons and uncharged neutrons, surrounded by a cloud of negatively charged electrons; the charges …
Periodic trends
As chemical reactions involve the valence electrons, elements with similar outer electron configurations may be expected to react similarly and form compounds with similar proportions of elements in them. Such elements are placed in the same group, and thus there tend to be clear similarities and trends in chemical behaviour as one proceeds down a group. As analogous configurations return …
Classification of elements
Many terms have been used in the literature to describe sets of elements that behave similarly. The group names alkali metal, alkaline earth metal, pnictogen, chalcogen, halogen, and noble gas are acknowledged by IUPAC; the other groups can be referred to by their number, or by their first element (e.g., group 6 is the chromium group). Some divide the p-block elements from groups 13 to …
Current questions
Although the modern periodic table is standard today, some variation can be found in period 1 and group 3. Discussion is ongoing about the placements of the relevant elements. The controversy has to do with conflicting understandings of whether chemical or electronic properties should primarily decide periodic table placement, and conflicting views of how the evidence should be used. A similar potential problem has been raised by theoretical investigations of the superheav…
Future extension beyond the seventh period
The most recently named elements – nihonium (113), moscovium (115), tennessine (117), and oganesson (118) – completed the seventh row of the periodic table. Future elements would have to begin an eighth row. These elements may be referred to either by their atomic numbers (e.g. "element 119"), or by the IUPAC systematic element names which directly relate to the atomic …
Alternative periodic tables
The periodic law may be represented in multiple ways, of which the standard periodic table is only one. Within 100 years of the appearance of Mendeleev's table in 1869, Edward G. Mazurs had collected an estimated 700 different published versions of the periodic table. Many forms retain the rectangular structure, including Janet's left-step periodic table (pictured below), and the m…
Overview
The periodic table is an arrangement of the chemical elements, structured by their atomic number, electron configuration and recurring chemical properties. In the basic form, elements are presented in order of increasing atomic number, in the reading sequence. Then, rows and columns are created by starting new rows and inserting blank cells, so that rows (periods) and columns (groups) s…
Atomic theory and isotopes
In 1907 it was discovered that thorium and radiothorium, products of radioactive decay, were physically different but chemically identical; this led Frederick Soddy to propose in 1910 that they were the same element but with different atomic weights. Soddy later proposed to call these elements with complete chemical identity “isotopes“.
Early history
A number of chemical elements, such as carbon, sulfur, iron, copper, silver, tin, gold, mercury, and lead, have been known since before antiquity, as they are found in their native form and are relatively simple to mine with primitive tools. Around 330 BCE, the Greek philosopher Aristotle proposed that everything is made up of a mixture of one or more roots, an idea originally suggested by the Sicilian philosopher Empedocles. The four roots, which were later renamed as elements by the A…
First categorizations
The history of the periodic table is also a history of the discovery of the chemical elements. The first person in recorded history to discover a new element was Hennig Brand, a bankrupt German merchant. Brand tried to discover the philosopher's stone—a mythical object that was supposed to turn inexpensive base metals into gold. In 1669, or later, his experiments with distilled human urine resulted …
Comprehensive formalizations
Properties of the elements, and thus properties of light and heavy bodies formed by them, are in a periodic dependence on their atomic weight.— Russian chemist Dmitri Mendeleev, formulating the periodic law for the first time in his 1871 article "Periodic regularity of the chemical elements"
French geologist Alexandre-Émile Béguyer de Chancourtois noticed that the ele…
Priority dispute and recognition
That person is rightly regarded as the creator of a particular scientific idea who perceives not merely its philosophical, but its real aspect, and who understands so to illustrate the matter so that everyone can become convinced of its truth. Then alone the idea, like matter, becomes indestructible.— Mendeleev in his 1881 article in British journal Chemical News in a correspondence debate with Meyer over priority of the periodic table invention
Inert gases and ether
The great value of Newland's, Mendeleef's, and Lothar Meyer's generalisation, known as the periodic arrangement of the elements, is universally acknowledged. But a study of this arrangement, it must be allowed, is a somewhat tantalising pleasure; for, although the properties of elements do undoubtedly vary qualitatively, and, indeed, show approximate quantitative rela…
Later expansions and the end of the periodic table
We already feel that we have neared the moment when this [periodic] law begins to change, and change fast.— Russian physicist Yuri Oganessian, co-discoverer of several superheavy elements, in 2019
As early as 1913, Bohr's research on electronic structure led physicists such as Johannes Rydberg to extrapolate the properties of undiscovered elements heavier than uranium. Many agreed that …