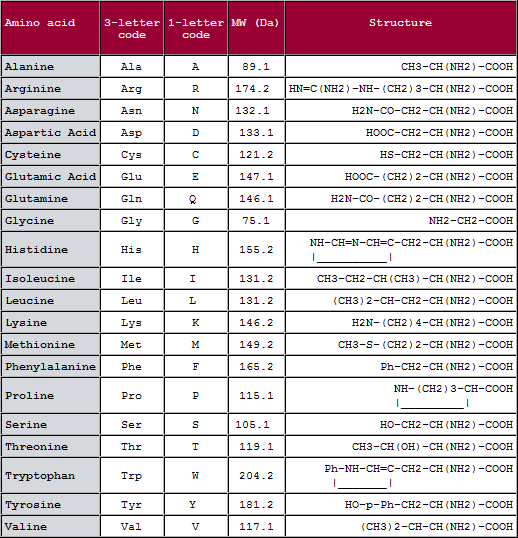
| Amino Acid | Abbreviation | Abbreviation | MW | pKa (25 °C) |
| sidechain | ||||
| Alanine | Ala | A | 89.10 | |
| Arginine | Arg | R | 174.20 | 12.48 |
| Asparagine | Asn | N | 132.12 |
| Amino Acid | Abbreviation | pKa (25 °C) |
|---|---|---|
| Alanine | Ala | 9.87 |
| Arginine | Arg | 8.99 |
| Asparagine | Asn | 8.72 |
| Aspartic Acid | Asp | 9.90 |
How do you determine pKa of an acid?
- Ka = [H+] [A-]/ [HA]
- pKa = - log Ka
- at half the equivalence point, pH = pKa = -log Ka
What are PKA and PKB in acids and bases?
pKa and pKb Table. pKa and pKb are common terms in chemistry that are known as dissociation constants. pKa is acid dissociation constant, and pKb is base dissociation constant. These terms are used to make it easy to work with very large or very small values. The “p” in these terms stands for “negative logarithm”.
Is the formula pKa?
Structure and pKa. The information here is to help you decide which structure of an acid or base will dominate at a particular pH. Let's do a general case. The equation for an acid is just HA = H+ + A- where = means equilibrium. pKa is defined as -log10 K a where K a = [H + ] [A -] / [HA].
What is the general formula of an amino acid?
The general linear formula of an amino acid is R-CH (NH2)-COOH. The 20 common amino acids are grouped in classes according to their side chains: Click here for IMGT classes of the 20 common amino acids 'Physicochemical' properties. All this is further explained here.

How do you find the pKa value of amino acids?
The pKa-values of the amino acid are determined from the full titration graph. To determine pKa1 and pKa2, locate the volume on the graphs half way between the two equivalence point volumes determined from the expanded derivative curves. The pH at this point is in the titration is equal to pKa2.
Which amino acid has 3 pKa values associated with it?
amino acid HistidineThe amino acid Histidine has 3 pKa values.
What amino acid has the lowest pKa?
histidineBasic amino acids Of the basic amino acid side chains, histidine has the lowest pKa (around 6) and is therefore neutral at around physiological pH.
Why do amino acids have 2 pKa values?
A molecule with two pKa values indicates the presence of two different inonizable functional groups.
Which amino acid side chain has the highest pKa?
arginineThe amino acid which has the side chain with the highest pKa value is arginine (R). The pKa value of the side chain of arginine is 12.48.
What is meant by pKa value?
pKa is a number that describes the acidity of a particular molecule. It measures the strength of an acid by how tightly a proton is held by a Bronsted acid. The lower the value of pKa, the stronger the acid and the greater its ability to donate its protons.
Do all amino acid side chains have a pKa?
Physiological pH is near neutral. It would appear that only histidine is of physiological relevance. However, pKa values can be shifted significantly by neighboring charged groups in complex molecular structures....1.4. 1 Acid-base Chemistry of Amino Acids.Amino acidFunctional GroupSide chain pKaArginineGuanidino12.5Lysine-NH210.57 more rows•Aug 18, 2019
Do you need to know pKa values of amino acids for MCAT?
5) Should I Know the pKa of Every Amino Acid for the MCAT? No, you should only know the pKa (which is the negative log of the acid dissociation constant or Ka value) of glutamate, glutamine, arginine, lysine, histidine, and aspartic acid. These amino acids are more sensitive to changes in pH.
How do you determine if an amino acid is acidic or basic?
0:198:39Basic and Acidic Amino Acids - YouTubeYouTubeStart of suggested clipEnd of suggested clipAnd glutamate are the two acidic amino acids. And what that means is their side chains bear fullMoreAnd glutamate are the two acidic amino acids. And what that means is their side chains bear full negative charge at the normal physiological pH of around seven.
What does the pKa of a protein tell you?
pKa values of amino acid side chains play an important role in defining the pH-dependent characteristics of a protein. The pH-dependence of the activity displayed by enzymes and the pH-dependence of protein stability, for example, are properties that are determined by the pKa values of amino acid side chains.
What does a high pKa mean?
pKa: If the pKa of an acid is high, it is a weak acid, and if the pKa of an acid is low, it is a strong acid. pH: If the pH of a system is high, the system is alkaline, but if the pH is low, that system is acidic.
What is the pk1 pk2 values of amino acids?
The pKa1 and pKa2 of an amino acid are 2.3 and 9.7 respectively.
Which amino acid has three pKa values quizlet?
Arginine has three ionizable groups, which have pKa values of 1.82, 8.99, and 12.48 (guanidino), respectively.
How many pKa values does histidine have?
Amino AcidAbbreviationpKa (25 °C)Glutamic AcidGlu2.10GlutamineGln2.17GlycineGly2.35HistidineHis1.8017 more rows
How many pKa values does aspartic acid have?
three pKa valuesThe three pKa values of aspartic acid are 1.89, 3.65 and 9.60.
How many PKAS does glutamic acid have?
2. (15 points) The pKa values for glutamic acid are 2.2, 4.3, and 9.7.
How many pKas are in an amino acid?
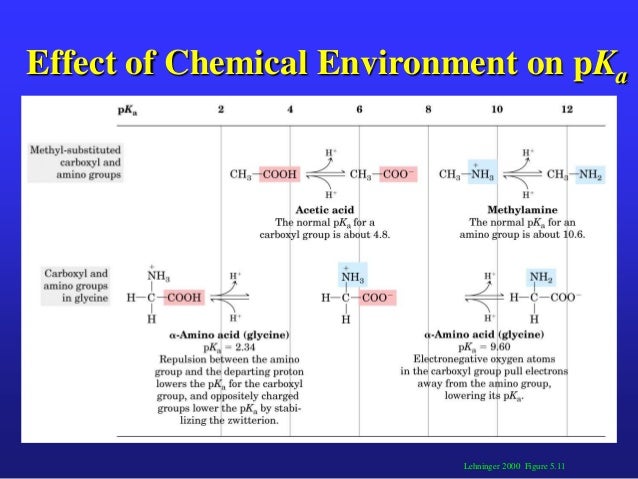
An amino acid has at least two pKa’s, one for the amino group and one for the carboxyl group. The pKa for each group is the negative logarithm (base 10) of the equilibrium constant for its deprotonation. Put more simply, this works out to the pH at which the group is protonated (e.g. - N H 3 +, − C O O H) on half the molecules and deprotonated ( − N H 2, C O O −) on half. If the amino acid has an ionizable group in its side chain, it has a third pKa.
What does pka mean in math?
pKa, or simply pK, represents the tendency towards ionization for a given acidic/basic substance or group. However attention must be paid here, as we’re dealing with -log (Ka), have in mind that lower pK values indicate acidic species that easily ionize, while higher pK values indicate basic species, that do the exact opposite, to remain protonated. Making it simple, the lower the pK, the stronger the acid.
What is the net charge of aspartic acid at pH 3?
The pI is ~3. So at a pH of ~3 the net charge of aspartic acid is 0 . To understand why, one needs to think about what is happening to the alpha-amino and carboxylic groups and carboxyl side chain group. At pH 3, the alpha-amino group will be fully protonated given that the pKa is 9.9. So that contributes a charge of +1. So that means that at pH 3, the sum of the net charge on the alpha carboxylic group and carboxyl R group must add up to -1.
What is the total amount of the alpha carboxyl group at pH 3?
At pH 3, the fraction of the alpha carboxyl group existing as A- is 0.91 and the fraction of the R carboxyl group existing as A- is 0.09. The total amount of A- is 1.0 and contributes a total charge of -1. Adding in the +1 charge of the alpha amino group gives a net charge of 0.
What is the pKa of glycine?
The pKa of glycine (H2N-CH2-COOH) is 2.34. So, if we add two F we can estimate the pKa of difluoroglycine to be
What is the pKa of trichloroacetic acid?
You can get below one with acids like trichloroacetic acid (Cl3COOH) and trifluoroacetic acid (F3COOH) with pKas of 0.7 and 0.23, respectively.
How to separate pKa?
One classic method is to do 2-D separation: chromatography in one dimension, electrophoresis or another chromatography in the other. Knowing the pKa in advance isn’t particularly a help, unless you think electrophoresing near its isoionic point will help separate it from other candidates.