What types of compounds does aluminium form?
Aluminium reacts with most nonmetals upon heating, forming compounds such as aluminium nitride (AlN), aluminium sulfide (Al2S3), and the aluminium halides (AlX3). It also forms a wide range of intermetallic compounds involving metals from every group on the periodic table.
How is aluminum a compound?
Instead, aluminum exists in mineral compounds, like bauxite. Aluminum is an element, not a compound, but it forms ionic compounds with other elements like sulfur, phosphorus, or oxygen. The metallic properties of aluminum include heat and electrical conductivity, low density, ductility, and corrosion resistance.
What are the components of aluminum?
Aluminium is most commonly alloyed with copper, zinc, magnesium, silicon, manganese and lithium. Small additions of chromium, titanium, zirconium, lead, bismuth and nickel are also made and iron is invariably present in small quantities.
What products does aluminum make?
Aluminium is a silvery-white, lightweight metal. It is soft and malleable. Aluminium is used in a huge variety of products including cans, foils, kitchen utensils, window frames, beer kegs and aeroplane parts. This is because of its particular properties.
Does aluminium form ionic compounds?
1 Answer. Aluminium does not form an ionic compound with chlorine. The bonding is covalent and in the solid state forms a layer lattice of Al and Cl atoms covalently bonded. The solid sublimes on heating (turns straight to a gas) and forms Al2Cl6 molecules which may then split up into AlCl3 molecules.
Is a aluminum a compound?
aluminum (Al), also spelled aluminium, chemical element, a lightweight silvery white metal of main Group 13 (IIIa, or boron group) of the periodic table.
What are three chemical properties of aluminum?
Aluminum is a soft and lightweight metal. It has a dull silvery appearance, because of a thin layer of oxidation that forms quickly when it is exposed to air. Aluminum is nontoxic (as the metal) nonmagnetic and non-sparking. Aluminum has only one naturally occurring isotope, aluminium-27, which is not radioactive.
What is aluminum in chemistry?
Aluminium is a silvery-white metal, the 13 element in the periodic table. One surprising fact about aluminium is that it's the most widespread metal on Earth, making up more than 8% of the Earth's core mass. It's also the third most common chemical element on our planet after oxygen and silicon.
What are the 3 ores of Aluminium?
Ores of aluminium are: Bauxite, Cryolite and Corundum.
What are 3 interesting facts about aluminum?
7 Fast Facts About Aluminum#1) It Weighs One-Third Less Than Steel. ... #2) It Doesn't Rust. ... #3) It's the World's Most Abundant Metal. ... #4) It's Recyclable. ... #5) It Was Used Thousands of Years Ago. ... #6) It's Resistant to Heat. ... #7) It's Ductile.
What are the two uses of aluminium?
Aluminium is widely used in the packaging industry for the production of coils, cans, foils, and other wrapping materials. It is also a component of many commonly used items such as utensils and watches. In construction industries, aluminium is employed in the manufacture of doors, windows, wires, and roofing.
Is aluminum foil a compound?
The aluminum foil is simply a very large collection of ordered aluminum atoms that are connected through metallic bonding. Therefore, we classify aluminum foil as an element.
Is aluminum an organic compound?
Inorganic compounds. The vast majority of compounds, including all aluminium-containing minerals and all commercially significant aluminium compounds, feature aluminium in the oxidation state 3+.
Is aluminum a mixture?
Aluminum, which is used in soda cans, is an element. A substance that can be broken down into chemically simpler components (because it has more than one element) is a compound. For example, water is a compound composed of the elements hydrogen and oxygen.
What is aluminum compound used for?
Aluminum compounds have many different uses, for example, as alums in water-treatment and alumina in abrasives and furnace linings. They are also found in consumer products such as antacids, astringents, buffered aspirin, food additives, and antiperspirants.
Is aluminum foil a compound?
The aluminum foil is simply a very large collection of ordered aluminum atoms that are connected through metallic bonding. Therefore, we classify aluminum foil as an element.
Where does aluminum come from?
Aluminum occurs in igneous rocks chiefly as aluminosilicates in feldspars, feldspathoids, and micas; in the soil derived from them as clay; and upon further weathering as bauxite and iron-rich laterite. Bauxite, a mixture of hydrated aluminum oxides, is the principal aluminum ore.
Why is aluminum added to metal?
Aluminum is added in small amounts to certain metals to improve their properties for specific uses, as in aluminum bronzes and most magnesium -base alloys; or, for aluminum-base alloys, moderate amounts of other metals and silicon are added to aluminum. The metal and its alloys are used extensively for aircraft construction, building materials, ...
What is the name of the element that is the most abundant in the Earth's crust?
Alternative Titles: Al, aluminium. Aluminum (Al), also spelled aluminium, chemical element, a lightweight silvery white metal of main Group 13 (IIIa, or boron group) of the periodic table. Aluminum is the most abundant metallic element in Earth ’s crust and the most widely used nonferrous metal. Because of its chemical activity, aluminum ...
What was the first compound used in pottery?
Before 5000 bce people in Mesopotamia were making fine pottery from a clay that consisted largely of an aluminum compound, and almost 4,000 years ago Egyptians and Babylonians used aluminum compounds in various chemicals and medicines. Pliny refers to alumen, now known as alum, a compound of aluminum widely employed in the ancient ...
How to detect aluminum?
Aluminum can be detected in concentrations as low as one part per million by means of emission spectroscopy. Aluminum can be quantitatively analyzed as the oxide (formula Al 2 O 3) or as a derivative of the organic nitrogen compound 8-hydroxyquinoline. The derivative has the molecular formula Al (C 9 H 6 ON) 3.
Which element has three electrons?
are boron (B), aluminum (Al), gallium (Ga), indium (In), thallium (Tl), and nihonium (Nh). They are characterized as a group by having three electrons in the outermost parts of their atomic structure. Boron, the lightest…. materials science: Aluminum.
How many elements are in the periodic table?
The periodic table is made up of 118 elements. How well do you know their symbols? In this quiz you’ll be shown all 118 chemical symbols, and you’ll need to choose the name of the chemical element that each one represents.
What is aluminum used for?
We use aluminum in a lot of our packaging, including as aluminum foil, in making mirrors, in pots and pans, and as a lightweight electrical wire. Aluminum is the most common element found in the earth's crust, at over 8%. It is typically found in the forms of potassium aluminum sulfate and aluminum oxide.
What is the charge of aluminum?
As mentioned, aluminum has a +3 charge , and sulfate has a -2 charge. In order to create of compound with no charge, we use 2 aluminum molecules and 3 sulfate molecules to form aluminum sulfate.
Is aluminum phosphate soluble in water?
Aluminum phosphate is also a white, crystalline powder, and is also soluble in water, although not nearly as soluble as aluminum sulfate. Once it is combined with other salts, such as potassium and nitrogen, it becomes highly insoluble.
What are binary compounds?
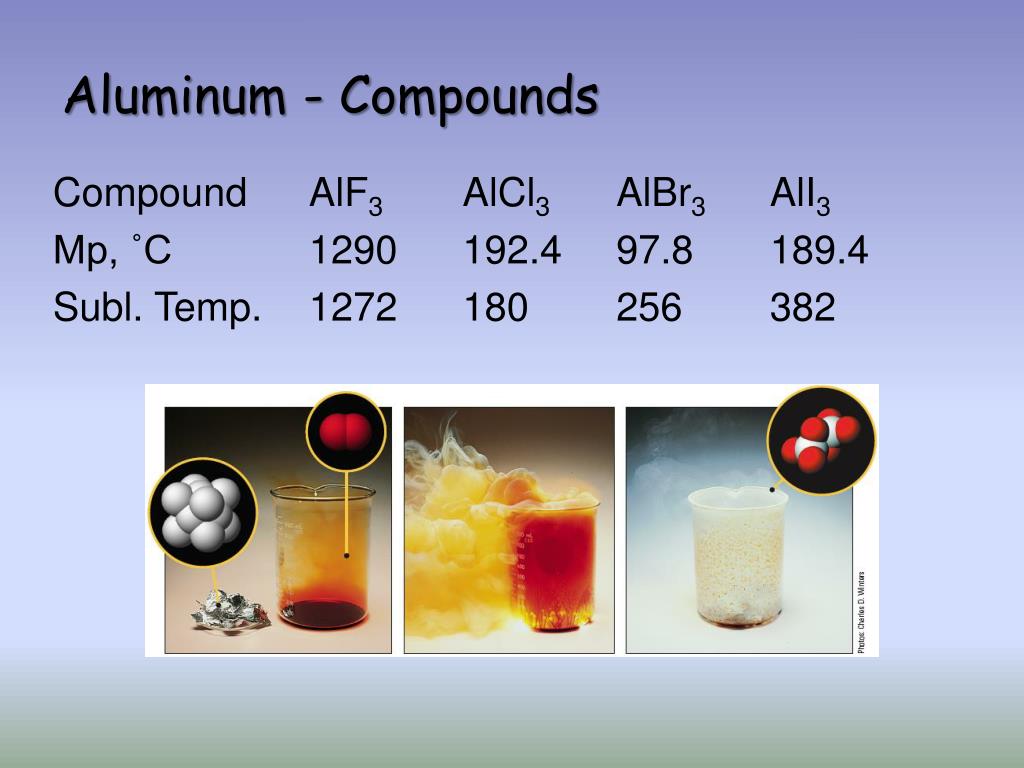
Binary compounds. Compound properties. Element reactions. This section lists some binary compounds with halogens (known as halides), oxygen (known as oxides), hydrogen (known as hydrides), and some other compounds of aluminium. For each compound, a formal oxidation number for aluminium is given, but the usefulness of this number is limited ...
What is the term for compounds of the type M x H y?
Hydrides. The term hydride is used to indicate compounds of the type M x H y and not necessarily to indicate that any compounds listed behave as hydrides chemically. Aluminium trihydride: AlH 3.

Overview
Monohalides
The aluminium(I) cation reacts with hydrogen halides to form the following aluminium monohalides:
• aluminium monofluoride (AlF)
• aluminium monochloride (AlCl)
• aluminium monobromide (AlBr)
Characteristics
Al(I) appears to be red, as solutions of AlBr and AlCl in organic solvents are both red. The presence of this color implies a relatively small HOMO/LUMO gap that is accessible by green light.
The geometry of compounds can be determined by analysis of the fine structure of the electronic spectra. Matrix isolation spectroscopy prevents disproportion…
Complex Chemistry
Aluminium is not only the most abundant metal in the earth's crust, but also an element of low toxicity. As such, aluminium (I) complexes attract considerable interest. These complexes can be supported by various ligands and used to activate small molecules.
In 2018, Liu et al. reviewed the chemistry of aluminium (I) with β-diketiminato li…
Metalloidal Clusters
Work in aluminium clusters has been done by Linti and Schnockel. These metalloidal clusters can be formed from Al(I) compounds, namely aluminium monohalides. These clusters are termed "metalloidal clusters" because the number of unbridged metal-metal bonds is greater than the number of localized metal-ligand bonds. On the way to metal formation, intermediates are trapped …
Natural Occurrence
Aluminium is rarely found in its +1 oxidation state in nature due to the immense stability of the +3 oxidation state.
Rotational transitions of AlF and AlCl have been detected in circumstellar shells near IRC +10216. The presence of AlF suggests that fluorine is produced in helium shell flashes instead of explosive nucleosynthesis.