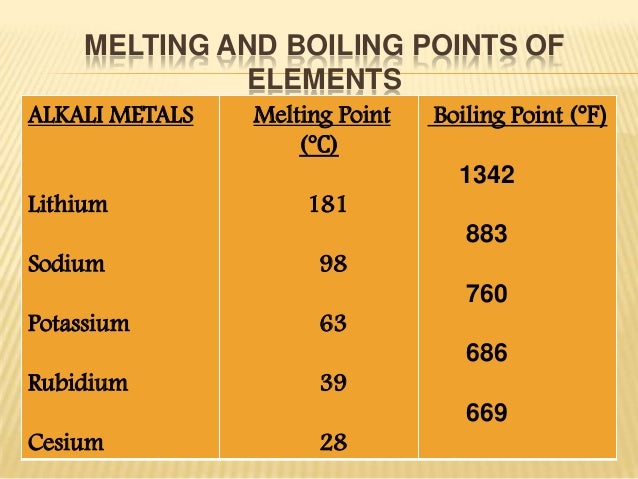
Alkaline earth metals facts
- They are shiny.
- They react with Halogens and form hydrated halides.
- All the alkaline earth metals react with water (except beryllium).
- They show a coloured flame when they are heated strongly.
- Magnesium is also found in our food.
- They do not exist as a pure form in nature.
- Calcium is the element which is found in building materials like cement, concrete, marbles, etc.
What are some interesting facts about alkaline earth metals?
What are 3 facts about alkali metals?
- Because they are so reactive with air and water, they are generally stored in oil.
- Cesium and rubidium are used to make atomic clocks.
- Sodium and potassium both play an important role in biological life on Earth.
- Sometimes cesium is also spelled “caesium.”
What is the most abundant elements of alkaline earth metals?
Visualizing the Abundance of Elements in the Earth’s Crust
- Oxygen. Oxygen is by far the most abundant element in the Earth’s crust, making up 46% of mass—coming up just short of half of the total.
- Silicon. More than 90% of the Earth’s crust is composed of silicate minerals, making silicon the second most abundant element in the Earth’s crust.
- Aluminum. ...
- Iron. ...
- Calcium. ...
What are the properties that make alkaline earth metals unique?
Alkaline earth metals have high electropositivity so their nature is electropositive. Among alkaline earth metals, magnesium does not give any colour on being exposed to flame. Alkaline earth metals are good conductors of heat. The density of alkaline earth metals is greater than those of alkali metals.
What group do the alkaline earth metals belong to?
The alkaline earth metals are the elements that correspond to group 2 of the modern periodic table. This group of elements includes beryllium, magnesium, calcium, strontium, barium, and radium. The elements of this group are quite similar in their physical and chemical properties. For example, all alkaline earth metals are silvery-white ...

What is common in alkaline earth metals?
Key PointsAlkaline earth metals have two valence electrons.They have low ionization energy, low electron affinity, and low electronegativity.They are highly reactive and often form divalent cations.They are good conductors of electricity.Alkaline earth metals have an oxidation state of +2.
What do the alkaline earth elements have in common?
The atoms of the alkaline-earth elements all have similar electronic structures, consisting of a pair of electrons (designated s electrons) in an outermost orbital, within which is a stable electronic configuration corresponding to that of a noble gas.
What properties do all alkaline earth metals have in common?
Summary of Common Alkaline Earth Properties Typically malleable and ductile. Relatively soft and strong. The elements readily form divalent cations (such as Mg2+and Ca2+). The alkaline earth metals are very reactive, although less so than the alkali metals.
What is special about alkaline earth metals?
The alkaline earth metals have the second-lowest first ionization energies in their respective periods of the periodic table because of their somewhat low effective nuclear charges and the ability to attain a full outer shell configuration by losing just two electrons.
What are three facts about alkaline earth metals?
Interesting Facts about Alkaline Earth Metals They are called alkaline because they form solutions with a pH greater than 7, making them bases or "alkaline." Radium is formed from the decay of uranium. It is very radioactive and is dangerous to handle. Calcium and magnesium are important for animal and plant life.
What do alkali metals and alkaline earth metals have in common?
Alkali metals and alkaline earth metals have the following properties: They are relatively soft metals. They are relatively low density metals. They have relatively low melting points compared to metals in general.
Are alkaline earth metals soft or hard?
Group 2A (or IIA) of the periodic table are the alkaline earth metals: beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). They are harder and less reactive than the alkali metals of Group 1A.
What is one characteristic of the alkali metals?
The alkali metals have the high thermal and electrical conductivity, lustre, ductility, and malleability that are characteristic of metals. Each alkali metal atom has a single electron in its outermost shell.
What do alkaline earth metals and transition metals have in common?
All alkaline earth elements have an oxidation number of +2, making them very reactive. The 38 elements in groups 3 through 12 of the periodic table are called "transition metals." As with all metals, the transition elements are both ductile and malleable, and conduct electricity and heat.
What are alkaline earth metals simple definition?
Definition of alkaline earth metal : any of the divalent strongly basic metals of group II of the periodic table comprising beryllium, magnesium, calcium, strontium, barium, and radium.
What is the difference between an alkali metal and an alkaline earth metal?
The main difference between alkali metals and alkaline earth metals is that alkali metals have one valence electron in the outermost orbit whereas alkaline earth metals have two valence electrons in the outermost orbit.
Are alkaline earth metals Basic?
Group 2 elements are referred to as “alkaline earth” metals (tan column below). The name “alkaline” comes from the fact that compounds of these elements form basic (pH greater than 7) or alkaline solutions when dissolved in water.
What do elements in the same group have in common?
The elements in each group have the same number of valence electrons. As a result, elements in the same group often display similar properties and reactivity.
What is the difference between alkali and alkaline earth metals?
5 days agoAlkali metals are the group 1 metals of the periodic table whereas alkaline earth metals are in the group 2. The main difference between alkali and alkaline is that alkali metals have one valence electron whereas alkaline earth metals have two valence electrons.
Do alkaline earth metals gain or lose electrons?
Answer and Explanation: Alkaline earth metals have two valence elctrons in their outermost shell. So, to complete their octet they need to lose two electrons. Therefore, 2 electrons are lost when alkaline earth metals form ions.
Which member of the alkaline earth metal family has?
Solution : The members of alkaline earth family (group 2) are : Be, Mg, Ca, Sr, Ba, Ra. The last element radium (Ra) is radioactive in nature. Step by step solution by experts to help you in doubt clearance & scoring excellent marks in exams.
The metals of which group are called alkaline earth metals?
Group 2 elements are called alkaline earth metals.
What is the most common oxidation state of alkaline earth metals?
+2 is the most common oxidation state of alkaline earth metals. They lose 2 electrons readily to achieve the nearest noble gas configuration.
Why are group 3 elements called alkaline earth metals?
The group 3 elements are called alkaline earth metals because the oxides of these metals produce a basic solution when they are dissolved in water.
What are the characteristics of alkaline earth metals?
Alkaline earth metals are reactive metals. They form divalent cations and are malleable and ductile.
Is copper an alkaline earth metal?
No, copper is not an alkaline earth metal. It is a transition element.
What are Alkaline Earth Metals?
Elements whose atoms have their s-subshell filled with their two valence electrons are called alkaline earth metals. Their general electronic configuration is [Noble gas] ns 2. They occupy the second column of the periodic table and so-called as group two metals also.
How are alkaline earth metals obtained?
Alkaline earth metals have low electrode potentials, and so are obtained by the electrolysis of the fused chlorides. Chlorides and fluorides of alkali and alkaline earth metals are added to reduce the melting point. Since magnesium burns readily in oxygen, the flow of a reducing gas like coal gas is maintained during electrolysis.
Why is the first ionization energy of alkaline earth metals larger than that of the alkali?
It is larger than that of the alkali metal atom for two reasons: Due to smaller radii and the electrons being held tightly by the higher nuclear charge, and.
What metals react with carbon to produce carbides?
Alkaline earth metals and their oxides, except beryllium, react with carbon to yield carbides. Carbides react with water to liberate acetylene gas and hence used as a source for the gas. M + 2C → MC 2 MC 2 + 2H 2 O → M (OH) 2 + C 2 H 2.
What is the most common oxidation state of alkaline earth metals?
Thus, the most common oxidation state exhibited by the alkaline earth metals is +2.
What are the elements in the alkaline earth?
This group of elements includes beryllium, magnesium, calcium, strontium, barium, and radium. The elements of this group are quite similar in their physical and chemical properties. For example, all alkaline earth metals are silvery-white coloured ...
Which has more density, alkaline or alkali?
In addition, due to the presence of two valence electrons, atoms have stronger metallic bonding. Hence, alkaline earth metals have more density and harder than alkali metals.
What is the color of alkaline earth metals?
All the alkaline earth metals are silvery-white, shiny and somewhat reactive.
What happens to the metallic character of alkaline earth metals as the atomic size increases?
Down the group, the atomic size increases. And as the atomic size increases, the electron will be lost easily. So the metallic character of alkaline earth metals increases from top to bottom in a group. Read more about: Metallic character trend in periodic table.
What metals react with water?
They also react with water and moisture of the air, if they are kept open. As alkaline earth metals are reactive, they always occur in the compound form in nature. For example; Beryllium is found in its mineral beryl, chrysoberyl and phenacite. Magnesium is found in its mineral magnesite.
What is the name of the metal that does not form an alkaline solution?
See the bold word “alkaline” and “earth” in the above 2 points, you will get the exact reason why they are called alkaline earth metals. ( Note: Beryllium (Be) is the only alkaline earth metal which does not form an alkaline solution on reacting with water.
How many valence electrons does an alkaline metal have?
Alkaline earth metals are similar in the following manner. All the alkaline earth metals have 2 valence electrons in their outermost orbit. They lose these two electrons and form cation with +2 charge. As you can see above that the alkaline earth metals have 2 electrons in the outermost shell.
Where is magnesium found?
Magnesium is found in its mineral magnesite.
Is magnesium a metal?
Also, magnesium is obtained from the mineral magnesite which is mostly found from the earth crust. So magnesium is alka line earth metal. Calcium reacts with water and forms calcium hydroxide which is alkaline in nature.