Why do particles move faster when heated?
With an increase in temperature, the particles move faster as they gain kinetic energy, resulting in increased collision rates and an increased rate of diffusion. … With an increase in temperature, the particles gain kinetic energy and vibrate faster and more strongly. Do hot or cold particles move faster?
How do particles move when they are heated?
Which particles move the fastest and have more thermal energy?
- That is one way that liquids and solids are alike. …
- Because the particles are freer to move around in gases than in solids or liquids, gas particles move the fastest. …
- Heating matter causes particles in that matter to speed up; cooling matter causes the particles to slow down.
What happens when PCl5 is heated?
What Happens When PCl 5 is Heated? When Phosphorus pentachloride (PCl 5 ) is heated, It will decompose into phosphorus trichloride(PCl 3 ) and chlorine gas (Cl 2 ). If placed in water, it will form phosphoric acid (H 3 PO 4 ) and hydrochloric acid (HCl) with the release of heat energy.
What happens to particles when they are dissolved?
There is a link between dissolving and diffusing the main difference is that dissolving involves the breaking of bonds like those that hold solid particles together. Once a solvent and a solute dissolve the particles mix together and then diffuse to form an even mixture of particles.

What happens to particles when they are heated or cooled?
When an object is heated the motion of the particles increases as the particles become more energetic. If it is cooled the motion of the particles decreases as they lose energy.
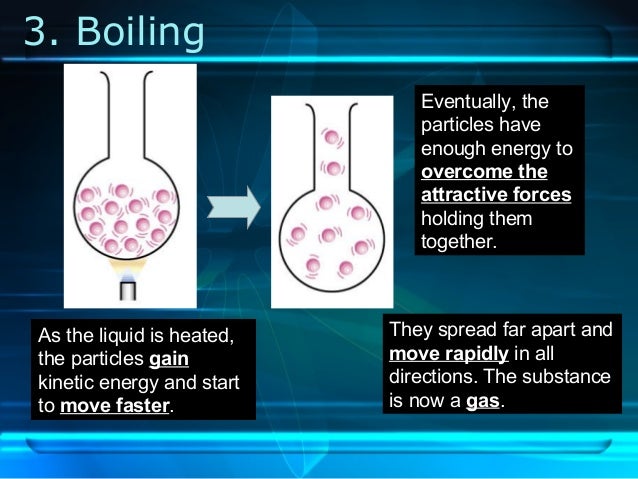
What happens to particles in a liquid when they are heated?
When a liquid is steadily heated, the particles move rapidly until many of them have sufficient energy to escape from the liquid and form a gas. This change of state is called evaporation. The liquid can also be changed to the solid state.
Do particles spread out when heated?
When a liquid or gas is heated, the molecules move faster, bump into each other, and spread apart. Because the molecules are spread apart, they take up more space. They are less dense.
What happens to particles when they are heated quizlet?
When the particles of a solid are heated, they move faster, move farther apart and take up more room. When the particles of a liquid, such as water, are heated, they move faster, move farther apart and take up more room.
What happens to atoms when they are heated quizlet?
When heat is added to a substance, the molecules and atoms vibrate faster. As atoms vibrate faster, the space between atoms increases. The motion and spacing of the particles determines the state of matter of the substance. The end result of increased molecular motion is that the object expands and takes up more space.
What happens to electrons when they are heated quizlet?
When you heat something, the heat gives the atoms in the substance energy. What happens when electrons are excited? The energy goes to the electron which give it the ability to jump to the higher energy level. The electron moves faster and further away from the nucleus.
What happens to the particles in a solid when they are heated to the point of melting?
Melting occurs when a solid is heated and turns to liquid. The particles in a solid gain enough energy to overcome the bonding forces holding them firmly in place. Typically, during melting, the particles start to move about, staying close to their neighbouring particles, then move more freely.
What happens to particles when heated in gas?
Molecules within gases are further apart and weakly attracted to each other. Heat causes the molecules to move faster, (heat energy is converted to kinetic energy ) which means that the volume of a gas increases more than the volume of a solid or liquid.