
What is the critical point or critical state?
The critical point or critical state is the point at which two phases of a substance initially become indistinguishable from one another. The critical point is the end point of a phase equilibrium curve, defined by a critical pressure Tp and critical temperature Pc. At this point, there is no phase boundary. Also Known As: critical state.
What is the critical point in a phase equation?
The critical point is the end point of a phase equilibrium curve, defined by a critical pressure T p and critical temperature P c. At this point, there is no phase boundary. Also Known As: critical state
What is the critical state of a pressure curve?
In thermodynamics, a critical point (or critical state) is the end point of a phase equilibrium curve. The most prominent example is the liquid–vapor critical point, the end point of the pressure–temperature curve that designates conditions under which a liquid and its vapor can coexist.
What is a critical point in thermodynamics?
In thermodynamics, a critical point (or critical state) is the end point of a phase equilibrium curve.
What does the critical point represent?
The critical point is the highest temperature and pressure at which a pure material can exist in vapor/liquid equilibrium. At temperatures higher than the critical temperature, the substance can not exist as a liquid, no matter what the pressure.
What is critical point in phase diagram of water?
The critical point is the point at which the critical temperature and pressure meet. Water particles in the gas phase are moving very quickly at 373.99°C. No matter how much pressure is applied to the gas, the gas phase cannot liquefy at any temperature higher than that.
What happens at the critical point?
The liquid expands and becomes less dense until, at the critical point, the densities of liquid and vapour become equal, eliminating the boundary between the two phases. If the average density at the start is too low, all the liquid will evaporate before the critical temperature is reached.
What does the critical point of water mean?
There's a special mix of temperature and pressure – we call it the critical point – where the difference between liquid and gas ceases to exist. For water, this happens at 374 °C (705 °F) and 218 atmospheres (normal air pressure is one atmosphere at sea level!).
How do you find critical points?
To find critical points of a function, first calculate the derivative. Remember that critical points must be in the domain of the function. So if x is undefined in f(x), it cannot be a critical point, but if x is defined in f(x) but undefined in f'(x), it is a critical point.
What is the difference between critical point and boiling point?
At the boiling point, molecular motion partially overcomes, and at the critical point, completely overcomes, liquefying forces.
What is the critical point called?
In thermodynamics, a critical point (or critical state) is the end point of a phase equilibrium curve. The most prominent example is the liquid–vapor critical point, the end point of the pressure–temperature curve that designates conditions under which a liquid and its vapor can coexist.
What is another word for critical point?
In this page you can discover 19 synonyms, antonyms, idiomatic expressions, and related words for critical-point, like: critical juncture, critical stage, pivotal point, turning point, climacteric, climax, crisis, critical mass, crucial moment, crucial point and crunch.
What is critical point and stationary point?
Critical point means where the derivative of the function is either zero or nonzero, while the stationary point means the derivative of the function is zero only.
What is critical point and stationary point?
Critical point means where the derivative of the function is either zero or nonzero, while the stationary point means the derivative of the function is zero only.
What will happen at the critical point of water?
Exactly at the critical point, these differences become zero, and at this point, the distinction between liquid and vapor disappears. The heat of vaporization also becomes zero at the critical point. Near the critical point, the physical properties of the liquid and the vapor change dramatically.
What is triple point and critical point?
The triple point represents a temperature and pressure combination where all three states of matter exist in equilibrium. Critical point is the temperature and pressure combination where the gas form of a substance can no longer be condensed back to a liquid, which becomes a supercritical fluid.
Where is the critical temperature in a phase diagram?
Point C is the critical point of the substance, which is the highest temperature and pressure at which a gas and a liquid can coexist at equilibrium. The figure below shows what happens when we draw a horizontal line across a phase diagram at a pressure of exactly 1 atm.
Can there be multiple critical points on a phase diagram?
There is only one critical point on a phase diagram. It can be found at the end of the equilibrium line between liquid and gas. This is the point t...
What is phase diagram and how can one interpret it?
A phase diagram is a representation of all the temperature and pressure combinations that create the different phases in a substance. Generally, th...
What is triple point and why is it important?
Triple point is found on a phase diagram where the three lines of equilibrium between states of matter converge. The triple point is a temperature...
What is the difference between triple point and critical point?
The triple point and critical point are both found on a phase diagram. The triple point represents a temperature and pressure combination where all...
What is the critical point in chemistry?
The critical point in chemistry is a temperature and pressure combination in which a given substance no longer can condense back into a gas. This p...
What is the critical point of a phase diagram?
The critical point on the phase diagram shows where the gas and liquid states of a liquid are identical and the substance is in one phase. Above the critical point, a substance is a supercritical fluid, where the gas and liquid phase of a substance are indistinguishable. Learning Outcomes.
What does the x axis of a phase diagram show?
The x -axis of the graph shows temperature. As temperature increases, most substances change from solid to liquid and then to gas. The y -axis shows pressure. Look at the three solid lines on the graph. These are the phase equilibrium lines. They are the lines on a phase diagram where two phases are in equilibrium.
What is phase equilibrium?
This is a state where the gas and liquid phase of a substance are indistinguishable. Lesson Summary. A phase diagram is a graph of the physical state of a substance (solid, liquid or gas) and the temperature and pressure of the substance. There are three solid lines on the phase diagram that are called the phase equilibrium lines.
What is the line on a phase diagram?
They are the lines on a phase diagram where two phases are in equilibrium. One line shows the equilibrium point between solid and liquid. One shows the equilibrium point between liquid and gas. The third shows the equilibrium point between solid and gas. Yes - solid and gas.
What is triple point?
This triple point is the point where the temperature and pressure conditions are right for all three states (solid, liquid and gas) to exist together at equilibrium. The triple point is like the peak of a pyramid.
Do you enter a different phase regardless of where you move from the triple point?
You enter a different phase regardless of where you move from the triple point.
What is the critical point of a pressure-temperature phase diagram?
The liquid–vapor critical point in a pressure–temperature phase diagram is at the high-temperature extreme of the liquid–gas phase boundary. The dotted green line shows the anomalous behavior of water.
What is the first critical point?
For simplicity and clarity, the generic notion of critical point is best introduced by discussing a specific example, the vapor-liquid critical point. This was the first critical point to be discovered, and it is still the best known and most studied one.
What does the principle of corresponding states mean?
The principle of corresponding states indicates that substances at equal reduced pressures and temperatures have equal reduced volumes. This relationship is approximately true for many substances, but becomes increasingly inaccurate for large values of pr .
What is the critical isotherm of matter?
Above the critical point there exists a state of matter that is continuously connected with (can be transformed without phase transition into) both the liquid and the gaseous state. It is called supercritical fluid.
What happens to liquid water at the critical point?
In the vicinity of the critical point, the physical properties of the liquid and the vapor change dramatically, with both phases becoming ever more similar. For instance, liquid water under normal conditions is nearly incompressible, has a low thermal expansion coefficient, has a high dielectric constant, and is an excellent solvent for electrolytes. Near the critical point, all these properties change into the exact opposite: water becomes compressible, expandable, a poor dielectric, a bad solvent for electrolytes, and prefers to mix with nonpolar gases and organic molecules.
What is a critical point in ethane?
Supercritical ethane, fluid. In thermodynamics, a critical point (or critical state) is the end point of a phase equilibrium curve. The most prominent example is the liquid–vapor critical point, the end point of the pressure–temperature curve that designates conditions under which a liquid and its vapor can coexist.
Who discovered the critical point?
The existence of a critical point was first discovered by Charles Cagniard de la Tour in 1822 and named by Dmitri Mendeleev in 1860 and Thomas Andrews in 1869. Cagniard showed that CO 2 could be liquefied at 31 °C at a pressure of 73 atm, but not at a slightly higher temperature, even under pressures as high as 3000 atm.
What is critical point?
Critical point, in physics, the set of conditions under which a liquid and its vapour become identical ( see phase diagram ). For each substance, the conditions defining the critical point are the critical temperature, the critical pressure, and the critical density. This is best understood by observing a simple experiment.
What happens at the critical point of a substance?
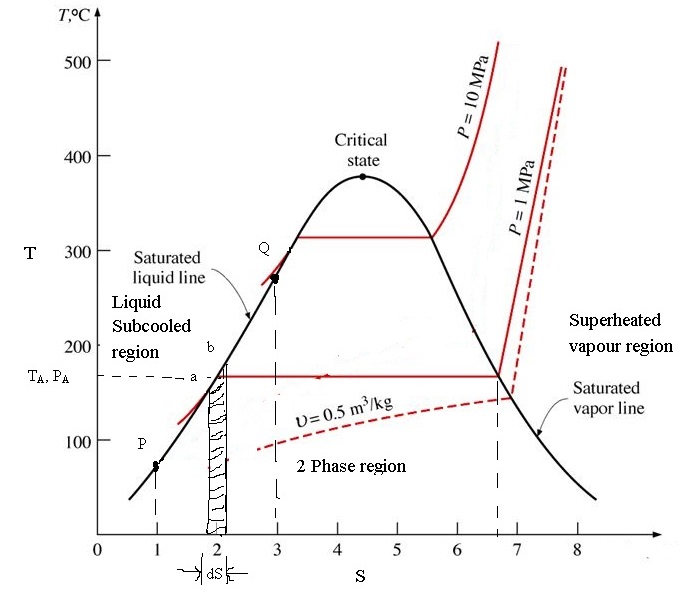
At the critical point, however, the vapour and liquid phases become identical, and above the critical point, the two phases are no longer distinct. Thus, if the substance moves from point P to S by the path PQRS so that no phase-change lines are crossed, the change…
What is phase diagram?
Phase diagram, graph showing the limiting conditions for solid, liquid, and gaseous phases of a single substance or of a mixture of substances while undergoing changes in pressure and temperature or in some other combination of variables, such as solubility and temperature. The Figure shows a typical phase diagram for…
What happens to the density of liquid at the critical point?
The liquid expands and becomes less dense until, at the critical point, the densities of liquid and vapour become equal, eliminating the boundary between the two phases. If the average density at the start is too low, all the liquid will evaporate before the critical temperature is reached. If the initial average density is too high, ...

Overview
Mixtures: liquid–liquid critical point
The liquid–liquid critical point of a solution, which occurs at the critical solution temperature, occurs at the limit of the two-phase region of the phase diagram. In other words, it is the point at which an infinitesimal change in some thermodynamic variable (such as temperature or pressure) leads to separation of the mixture into two distinct liquid phases, as shown in the polymer–solven…
Liquid–vapor critical point
For simplicity and clarity, the generic notion of critical point is best introduced by discussing a specific example, the vapor-liquid critical point. This was the first critical point to be discovered, and it is still the best known and most studied one.
The figure to the right shows the schematic PT diagram of a pure substance (a…
See also
• Conformal field theory
• Critical exponents
• Critical phenomena (more advanced article)
• Critical points of the elements (data page)
Footnotes
1. ^ Horstmann, Sven (2000). Theoretische und experimentelle Untersuchungen zum Hochdruckphasengleichgewichtsverhalten fluider Stoffgemische für die Erweiterung der PSRK-Gruppenbeitragszustandsgleichung [Theoretical and experimental investigations of the high-pressure phase equilibrium behavior of fluid mixtures for the expansion of the PSRK group contribution equation of state] (Ph.D.) (in German). Oldenburg, Germany: Carl-von-Ossietzky Univ…
External links
• "Critical points for some common solvents". ProSciTech. Archived from the original on 2008-01-31.
• "Critical Temperature and Pressure". Department of Chemistry. Purdue University. Retrieved 2006-12-03.