What can cause hyperchloremia?
Hyperchloremia can result from a variety of conditions including water depletion, excessive chloride exposure and metabolic acidosis. 3. The pathogenic cause of hyperchloremia will provide guidance on how the disturbance should be treated: water depletion is treated with judicious water repletion; excess chloride administration by withholding ...
What are some of the causes of metabolic alkalosis?
Some other Metabolic Alkalosis causes include:
- Use of steroids
- Excessive of antacids
- Overuse of alkaline drugs, like Bicarbonate
Why does metabolic acidosis cause hypochloremia?
There are several ways that hyperchloremia can occur. These include: intake of too much saline solution while in the hospital, such as during a surgery Hyperchloremic acidosis, or hyperchloremic metabolic acidosis, occurs when a loss of bicarbonate (alkali) tips the pH balance in your blood toward becoming too acidic (metabolic acidosis).
Why do loop diuretics cause metabolic alkalosis?
Loop diuretics act in the ascending limb of the loop of henle. They inhibit the Na-K-2Cl contransporter to inhibit sodium and chloride reabsorption. Loop and thiazide diuretics can cause metabolic alkalosis due to increased excretion of chloride in proportion to bicarbonate.

How does hypochloremia cause metabolic alkalosis?
Hypochloremia can contribute to the maintenance of metabolic alkalosis by increasing the reabsorption of and reducing the secretion of bicarbonate in the distal tubule.
What is alkalosis hypochloremic?
Hypochloremic alkalosis is caused by an extreme lack or loss of chloride, such as from prolonged vomiting. Hypokalemic alkalosis is caused by the kidneys' response to an extreme lack or loss of potassium. This can occur from taking certain water pills (diuretics).
How is hypochloremic metabolic alkalosis treated?
Metabolic alkalosis treatment uses an intravenous (IV) line to deliver fluid and other substances, such as:Saline infusion.Potassium replacement.Magnesium replacement.Chloride infusion.Hydrochloric acid infusion.Stopping the medications that caused the condition, for example high doses of diuretics.
What does metabolic alkalosis indicate?
Metabolic alkalosis is defined as a disease state where the body's pH is elevated to greater than 7.45 secondary to some metabolic process. Before going into details about pathology and this disease process, some background information about the physiological pH buffering process is important.
What is the most common cause of metabolic alkalosis?
The most common causes of metabolic alkalosis are the use of diuretics and the external loss of gastric secretions.
What are signs and symptoms of hypochloremia?
What are the symptoms of hypochloremia?fluid loss.dehydration.weakness or fatigue.difficulty breathing.diarrhea or vomiting, caused by fluid loss.
What are three causes of metabolic alkalosis?
Metabolic alkalosis, a disorder that elevates the serum bicarbonate, can result from several mechanisms: intracellular shift of hydrogen ions; gastrointestinal loss of hydrogen ions; excessive renal hydrogen ion loss; administration and retention of bicarbonate ions; or volume contraction around a constant amount of ...
What hypochloremia means?
So hypochloremia means that your concentration of blood chloride is below the normal range. If you have high levels of chloride in your blood, that's known as hyperchloremia. If you're healthy, your blood chloride levels don't change much during the day.
What medication is used for metabolic alkalosis?
Metabolic alkalosis is corrected with the aldosterone antagonist spironolactone or with other potassium-sparing diuretics (eg, amiloride, triamterene). If the cause of primary hyperaldosteronism is an adrenal adenoma or carcinoma, surgical removal of the tumor should correct the alkalosis.
What are three symptoms of metabolic alkalosis?
People may have irritability, muscle twitching, muscle cramps, or even muscle spasms. Blood is tested to diagnose alkalosis. Metabolic alkalosis is treated by replacing water and mineral salts such as sodium and potassium (electrolytes) and correcting the cause.
What disease processes can cause metabolic alkalosis?
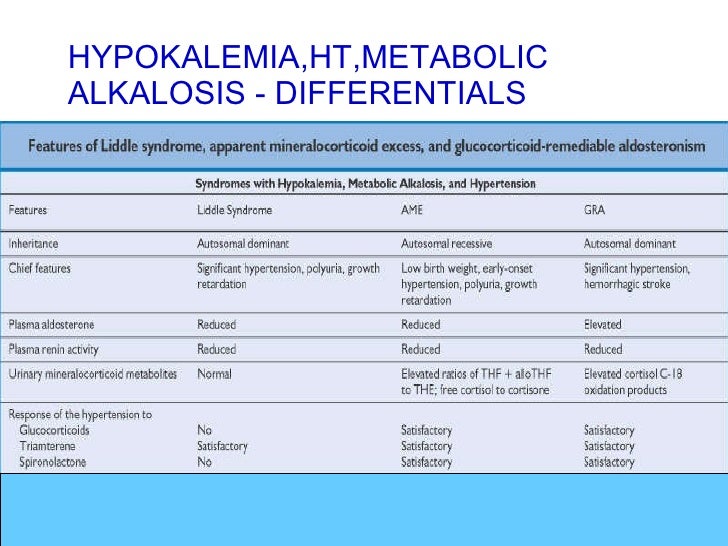
Five inherited diseases that can cause metabolic alkalosis are:Bartter syndrome.Gitelman syndrome.Liddle syndrome.glucocorticoid remediable aldosteronism.apparent mineralocorticoid excess.
Is metabolic alkalosis fatal?
The mortality associated with severe metabolic alkalosis is substantial; a mortality rate of 45% in patients with an arterial blood pH of 7.55 and 80% when the pH was greater than 7.65 has been reported (2).
What hypochloremia means?
So hypochloremia means that your concentration of blood chloride is below the normal range. If you have high levels of chloride in your blood, that's known as hyperchloremia. If you're healthy, your blood chloride levels don't change much during the day.
What causes hypochloremia?
Values below 95-105 mEq in adults and children are considered low (also known as hypochloremia). Potential causes include excessive chloride loss (such as from vomiting, watery diarrhea, laxative abuse), low-salt infusions, metabolic alkalosis, and many underlying electrolyte imbalances.
What is Hypochloremic acidosis?
Hyperchloremic acidosis is a disease state where acidosis (pH less than 7.35) develops with an increase in ionic chloride. Understanding the physiological pH buffering system is important. The major pH buffer system in the human body is the bicarbonate/carbon dioxide (HCO3/CO2) chemical equilibrium system.[1][2][3]
Who are risk of hypochloremia?
Chloride-deficientformulas, salt restricted diets, GI tube drainage, and severe vomiting and diarrheaare risk factors for hypochloremia. As chloride decreases(Usually because of volume depletion), sodium and bicarbonate ions are retained bythe kidney to balance the loss.
What is the difference between HCO3 and PCO2?
Where HCO3- represents in the plasma bicarbonate concentration and pCO2 is the plasma carbon dioxide tension in the blood. At normal conditions in the body, the CO2 production and excretion are equal and pCO2 is maintained at 40 mm Hg.
What causes bicarbonate levels to be elevated?
However, elevated urinary bicarbonate levels may also occur due to renal tubular acidosis which must be remembered in the differential diagnosis. Renal tubular acidosis (RTA) is characterized by the development of metabolic acidosis due to a defect in the ability of the renal tubules to perform normal functions [1, 6]. All forms of RTA are characterized by a normal anion gap (hyperchloremic) metabolic acidosis. This form of metabolic acidosis usually results from either the net retention of hydrogen chloride or its equivalent (such as ammonium chloride) or the net loss of sodium bicarbonate or its equivalent. The major cause of a normal anion gap acidosis in patients without renal failure is diarrhea.
How is bicarbonate reabsorption maintained?
At the cellular level, the balance between the excretion and retention of the bicarbonate in the plasma is maintained by the type A and B cells in the collecting tubules [4, 5]. Bicarbonate reabsorption in the medullary collecting tubule and in type A intercalated cell in the cortical collecting tubule is mediated by hydrogen secretion via H-ATPase pumps and passive cosecretion of chloride in the luminal membrane. Water within the cell dissociates into H+ and OH- ions. H+ ions are secreted into the lumen by H-ATPase pumps in the luminal membrane, where they primarily combine with NH3 to from NH4+. The OH- ions in the cell combine with CO2 to form HCO3- in a reaction catalyzed by carbonic anhydrase. Bicarbonate is then returned to the systemic circulation via Cl-HCO3 exchangers in the basolateral membrane. The favorable inward concentration gradient for Cl- provides the energy for HCO3- reabsorption. The intracellular bicarbonate is returned to the systemic circulation through the Cl/HCO3 exchangers in the basolateral membrane. A decline in the tubular fluid chloride concentration will promote both chloride and hydrogen secretion. H-K-ATPase pumps, which lead to both H+ secretion and K+ reabsorption, are also present in the luminal membrane. The number of these pumps increases with K+ depletion, suggesting that their main function may be to promote K+ conservation.
How long does it take for NaHCO3 to reach maximum?
The ability to enhance bicarbonate reabsorption takes 3 to 4 days to reach its maximum. Thus, there is increased NaHCO3 delivery to the collecting tubules.
Where is bicarbonate secreted in the cortical collecting tubule?
The type B intercalated cells in the cortical collecting tubule are able to directly secrete bicarbonate by reversing the location of the transporters as seen during the recovery phase of metabolic alkalosis [5]. The Cl/HCO3 exchangers are now located in the luminal membrane, leading to bicarbonate secretion into the tubular lumen. The activity of these cells is appropriately enhanced by alkalemia in an attempt to excrete the excess bicarbonate. These two mechanisms play a role in the conservation and excretion of the bicarbonate balance in the body depending on the body pH levels.
Why is bicarbonate in plasma higher?
Increased plasma bicarbonate concentration due to hydrogen loss in the urine or gastrointestinal tract, hydrogen movement into the cells, the administration of bicarbonate, or volume contraction.
What is the RTA?
All forms of RTA are characterized by a normal anion gap (hyperchloremic) metabolic acidosis. This form of metabolic acidosis usually results from either the net retention of hydrogen chloride or its equivalent (such as ammonium chloride) or the net loss of sodium bicarbonate or its equivalent.
What causes alkalosis in the body?
Other causes of metabolic alkalosis include medical conditions such as: Cystic fibrosis. Dehydration. Electrolyte imbalances, which affect levels of sodium, chloride, potassium and other electrolytes. High levels of the adrenal hormone aldosterone ( hyperaldosteronism ).
Why is metabolic alkalosis important?
Metabolic alkalosis is usually not life-threatening. It does not have lingering effects on your health once it is treated. But it’s important to seek medical care because it can lead to severe complications. Treatment with IV fluids helps many people make a full recovery. Addressing the cause can lower your risk of future episodes.
Why does alkalosis occur?
It can occur in a variety of conditions. It may be due to digestive issues, like repeated vomiting, that disrupt the blood ’s acid-base balance. It can also be due to complications of conditions affecting the heart, liver and kidneys.
What to do after alkalosis treatment?
You may wish to make small changes to lower your risk of future episodes. These changes may include: Decreasing the dose of or discontinuing steroids, laxatives, water pills or antacids.
What is the term for the body's acid-base balance?
What is alkalosis ? Alkalosis occurs when your blood and body fluids contain an excess of bases or alkali. Your blood’s acid-base (alkali) balance is critical to your well-being. When the balance is off, even by a small amount, it can make you sick.
What are the symptoms of metabolic alkalosis?
Many metabolic alkalosis symptoms are concerning and need prompt medical evaluation. If you are experiencing an arrhythmia, seizures or confusion, seek care right away.
What tests are used to check for metabolic alkalosis?
Blood tests to measure blood gases, acid-base balance and electrolyte levels. Electrocardiogram (EKG) to check for an arrhythmia. Urinalysis that may help find the cause of the metabolic alkalosis.
What happens if the expected pCO2 does not match the measured value?
If the expected pCO2 does not match the measured value, an underlying metabolic alkalosis is a likely present.
What causes bicarbonate to increase in blood?
Several etiologies lead to increases in bicarbonate within the blood. The simplest of which is an overdose of exogenous sodium bicarbonate in a medical setting. Milk-alkali syndrome is a pathology where the patient consumes excessive quantities of oral calcium antacids, which leads to hypercalcemia and varying degrees of renal failure. Additionally, since antacids are neutralizing agents, they add alkaline substances to the body while reducing acid levels thus increasing pH. A pathology that is in line with normal physiology is the body’s natural compensation mechanism for hypercarbia. When a patient hypoventilates, CO2 retention occurs in the lungs and subsequently reduces pH. Over time, the renal system compensates by retaining bicarbonate to balance pH. This is a slower process. Once the hypoventilation is corrected, such as with a ventilator-assisted respiratory failure patient CO2 levels will quickly decrease, but bicarbonate levels will lag in reducing. This causes post-hypercapnia metabolic alkalosis, which is self-correcting. It is possible to calculate the expected pCO2 in the setting of metabolic alkalosis to determine if it is a compensatory increase in bicarbonate, or if there is an underlying pathology driving alkalosis using the following equation:
How is CO2 regulated?
CO2 levels are physiologically regulated by the pulmonary system through respiration, whereas the HCO3 levels are regulated through the renal system with reabsorption rates. Therefore, metabolic alkalosis is an increase in serum HCO3. [1][2]
What is the term for a disease where the body's pH is elevated to greater than 7.45?
A decrease in pH below this range is acidosis, an increase over this range is alkalosis. Metabolic alkalosis is defined as a disease state where the body’s pH is elevated to greater than 7.45 secondary to some metabolic process.
What is chloride responsive metabolic alkalosis?
In chloride responsive metabolic alkalosis, this includes repletion of electrolytes, specifically chloride and potassium along with the replenishment of fluid. In scenarios, such as congestive heart failure (CHF) or edematous states, diuresis is essential using potassium-sparing diuretics.
What is metabolic alkalosis?
Metabolic alkalosis is defined as a disease state where the body’s pH is elevated to greater than 7.45 secondary to some metabolic process. Before going into details about pathology and this disease process, some background information about the physiological pH buffering process is important. The primary pH buffer system in ...
What is the normal pH of the human body?
Normal human physiological pH is 7.35 to 7.45. A decrease in pH below this range is acidosis, an increase over this range is alkalosis. Metabolic alkalosis is defined as a disease state where the body’s pH is elevated to greater than 7.45 secondary to some metabolic process.
How does hypochloremia affect metabolic alkalosis?
Hypochloremia can contribute to the maintenance of metabolic alkalosis by increasing the reabsorption of and reducing the secretion of bicarbonate in the distal tubule.
Why is urine chloride less than 10 mEq/L?
If urine chloride is less than 10 mEq/L, then hypochloremia is due to chloride responsive alkalosis. If greater than 40 mEq/L, hypochloremia is due to volume overload or dilution. These patients usually have a metabolic alkalosis due to excess mineralocorticoid or glucocorticoid.
Why is the colon not evident in the midgut volvulus?
With midgut volvulus, the colon is not evident as the abdomen is opened because it lies posteriorly. In addition, the small bowel may appear congested and blue, with dilated mesenteric veins ( Fig. 86-23, A ).
What are the side effects of thiazide diuretics?
Most serious adverse effects of thiazide diuretics are fluid and electrolyte imbalances and include ECV depletion, hypotension, hypokalemia, hyponatremia, hypochloremia, metabolic alkalosis, hypomagnesemia, hypercalcemia, and hyperuricemia. Electrolyte imbalances may lead to cardiac arrhythmias and orthostatic hypotension secondary to ECV depletion. Thiazide diuretics decrease glucose tolerance and increase the risk of precipitating new onset diabetes mellitus. Thiazide induced hyperglycemia is due to decreased insulin secretion and is secondary to extracellular potassium depletion, opening of ATP sensitive potassium channels and hyperpolarization of pancreatic islet cells. Thiazide diuretics also may increase plasma levels of low-density lipoprotein cholesterol, total cholesterol, and total triglycerides and have been associated with acute cholecystitis and rare cases of pancreatitis.
What is the cause of hypochloremia?
Hypochloremia is caused by prolonged vomiting involving a loss of chloride in excess of sodium, accompanied by a compensatory increase in plasma bicarbonate (hypochloremic alkalosis), as well as in diabetic ketoacidosis and in conditions associated with high serum bicarbonate concentrations, for example, in compensation of respiratory acidosis or metabolic alkalosis.
How to treat chloride resistant alkalosis?
Treatment is aimed at therapy for the underlying disorder. Chloride‐responsive alkalosis is treated with normal saline. Chloride‐resistant metabolic alkalosis requires IV normal saline plus K. Give one fourth as KCl and three fourths as NaCl.
What are electrolytes abnormalities?
Electrolyte abnormalities: Hypokalemia, hypochloremia, and metabolic alkalosis may occur. The hypokalemia can cause a prolonged QT interval and T-wave abnormalities. Esophageal: Acid reflux with esophagitis and (rarely) Mallory-Weiss tear may be found.
when to treat?
Patients with chronic hypercapneic respiratory failure will develop a chronic compensatory metabolic alkalosis.
What is the most commonly used diuretic for metabolic alkalosis?
Acetazolamide: ( more )#N#Acetazolamide is the most commonly used diuretic for metabolic alkalosis and perhaps the most effective.#N#Make sure to monitor potassium levels carefully (acetazolamide may induce hypokalemia, which will aggravate treatment of the metabolic alkalosis).
Is metabolic alkalosis a compensatory mechanism?
This is a compensatory mechanism which is generally beneficial. The metabolic alkalosis allows them to have a fairly normal pH, despite hypoventilation. Without metabolic compensation, these patients would be acidemic and have an increased respiratory drive.
What causes metabolic alkalosis?
Loss of acidic gastric contents will cause a metabolic alkalosis.
Is hypoventilation a sign of metabolic alkalosis?
Hypoventilation (due to respiratory compensation for the metabolic alkalosis). Generally not a significant issue. For patients with a weak respiratory drive (e.g., obesity hypoventilation syndrome or COPD), severe metabolic alkalosis may promote hypoventilation.
Can metabolic alkalosis be treated with volume resuscitation?
Instead, resolving the underlying cause is generally sufficient. For example, a patient with hypovolemia may be treated with volume resuscitation.
Is metabolic alkalosis a treatment for renal failure?
This is a potential treatment of metabolic alkalosis among patients with renal failure.
