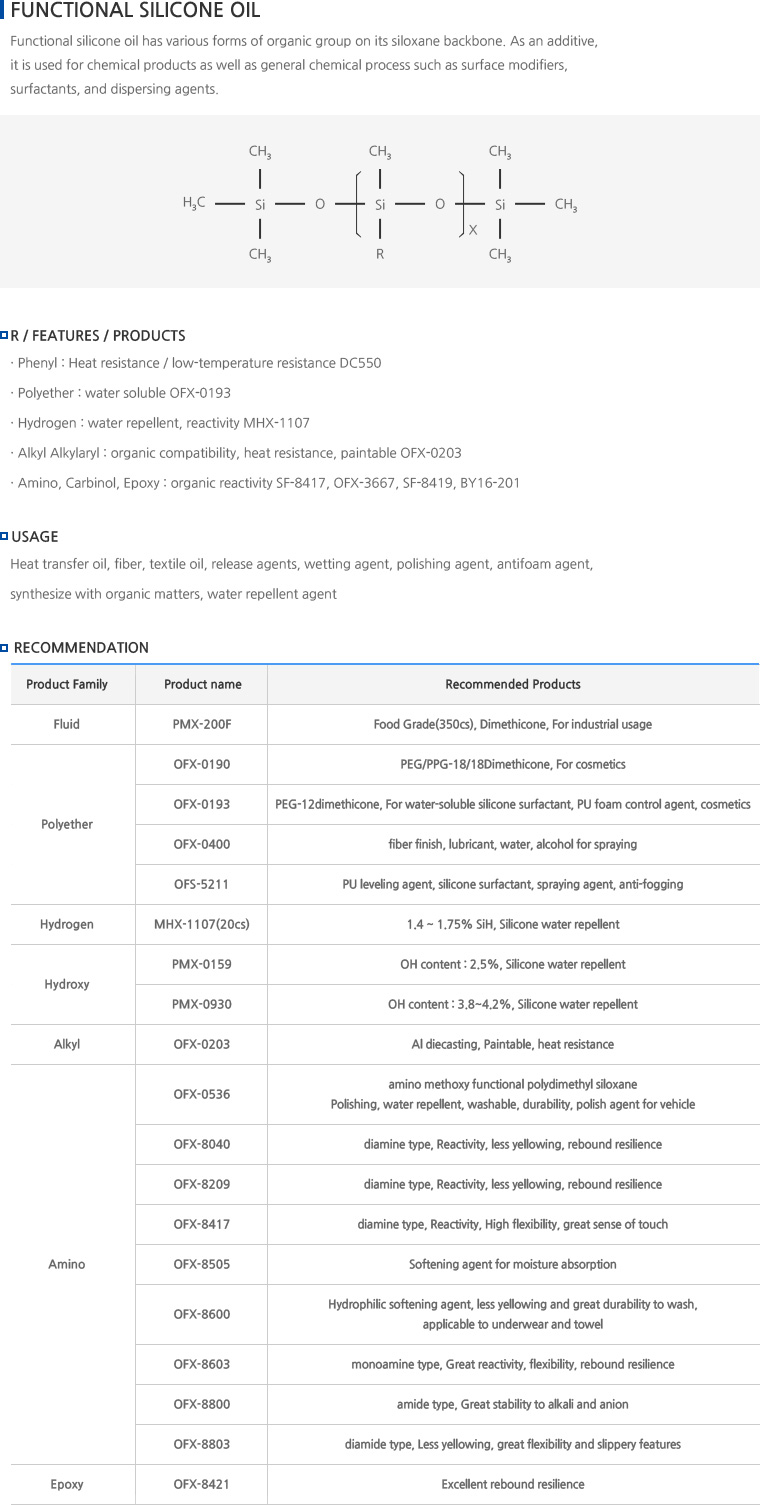
What are Silicone Oils?
- Silicone oil molecular structure. The repeating backbone of silicone oils and other siloxane polymers consists of alternating silicon and oxygen atoms.
- Discover our latest video "What are Silicone Oils": What are Silicone Oils?
- Silicone oil properties. ...
- Revision sheet. ...
What is silicone oil?
Jump to navigation Jump to search. A silicone oil is any liquid polymerized siloxane with organic side chains. The most important member is polydimethylsiloxane. These polymers are of commercial interest because of their relatively high thermal stability and their lubricating properties.
What is silicone made of?
In short, silicone is a synthetic substance made up of silicon, well, silicon is an element extracted from silica. 2. How is silicone made? Silica is put into a silicon furnace at a high temperature to drive the oxygen out of silica, the reaction between silica and carbon produces silicon.
Does silicone oil smell?
Silicone oil has no odor or taste, is fire resistant, and offers excellent electrical insulation. A liquid polymerized siloxane, with organic side chains, can be termed as silicone oil. Silicone oil consists of alternating atoms of silicone and oxygen (..Si-O-Si-O-Si..). This odorless oil is nonflammable and nontoxic.
What happens when silicone oil is mixed with water?
When the mixing energy of silicone oil is sufficient to disrupt the hydrogen bonding between water molecules, it can be introduced into water. However, on stoppage of mixing, the oil is forced out of the water by the reformation of the hydrogen bonds between water molecules, and rises to the surface of the substrate.

What are the ingredients in silicone oil?
A silicone oil is any liquid polymerized siloxane with organic side chains. The most important member is polydimethylsiloxane. These polymers are of commercial interest because of their relatively high thermal stability, lubricating, and dielectric properties.
How do you manufacture silicone oil?
This is done by heating a large volume of quartz sand to temperatures as high as 1800˚C. The result is pure, isolated silicon, which is allowed to cool and then ground into a fine powder. To make silicone, this fine silicon powder is combined with methyl chloride and heated once again.
What is silicon oil used for?
The silicone–silica greases are used primarily as electrical greases for such applications as aircraft and car ignition systems. The fluids are also used in shock absorbers, hydraulic fluids, dashpots and other damping systems designed for high-temperature operation.
What is the main ingredient in silicone?
While the main chain of common organic synthetic polymers consists of repeating carbon (C) atoms, silicone is an "inorganic synthetic polymer" whose main chain is made of polysiloxane, which is the repetition of silicon(Si) and oxygen(O) atoms(1,2).
Is silicone oil petroleum based?
Non-petroleum oils include synthetic oils, such as silicone fluids, tung oils, and wood-derivative oils such as resin/rosin oils.
What is similar to silicone oil?
Dimethicone. This is a skin-safe alternative to silicone which is used in many beauty products. Another common usage is personal lubricants.
Is wd40 silicone oil?
WD-40 Specialist® Silicone Lubricant safely lubricates, waterproofs and protects metal and non-metal surfaces such as rubber, plastic and vinyl.
Is silicone oil harmful to humans?
The bottom line. When used in household products such as cooking utensils, silicone is largely a safe material. However, research suggests that liquid silicone can be dangerous if it gets inside your body through ingestion, injection, absorption, or leakage from an implant.
Is silicone oil safe on skin?
The Cosmetic Ingredient Review Expert Panel concluded that silicone in skin care products, such as moisturizers and patches, is safe. Forms of silicone, such as dimethicone, do not interact with the epidermis, the upper layer of the skin, and are therefore unlikely to penetrate the skin barrier.
Where is silicon naturally found?
Natural abundance Silicon makes up 27.7% of the Earth's crust by mass and is the second most abundant element (oxygen is the first). It does not occur uncombined in nature but occurs chiefly as the oxide (silica) and as silicates. The oxide includes sand, quartz, rock crystal, amethyst, agate, flint and opal.
What does silicone do to skin?
Silicone clogs your pores It traps everything like bacteria, impurities, dirt, and sebum in your skin by forming a barrier on the skin. Silicone also causes pores to enlarge, and can also encourage blackheads and acne to form. It can affect the natural process of sweating which can lead to blemishes forming.
Is silicone natural or manmade?
Many people seem to think they are a natural material derived directly from sand. Not so. Like any plastic polymer, silicones are synthetic and include a mix of chemical additives derived from fossil fuels.
How do you make silicone oil emulsion?
In a process for the production of a silicone in water emulsion in which a polysiloxane fluid, at least one surfactant and water are continuously fed to a high shear mixer in such proportions as to form a viscous oil in water emulsion which is continuously withdrawn from the mixer.
Is silicone A oil?
Silicone oil is similar to a traditional hydrocarbon oil except that its molecular chain replaces carbon units with siloxane units. This oil is a nonflammable, nontoxic, tasteless, and odorless material. It is widely used by several industries including laboratories and medical facilities.
How do you make silicone softener?
The preparation method of silicone softener is to take metallic zinc particles and selenium powder, stir and mix them and place them in a ball mill tank, ball mill, sieving, weigh methanol, deionized water, ethylenediamine, ball mill powder and hydrazine hydrate in a beaker.
What is silicone emulsion?
Silicone emulsions are insoluble silicones evenly dispersed in water with the aid of an emulsifying surfactant. They can be divided into three groups, depending on the type of emulsifier system used, i.e. they can be anionic, cationic and non-ionic.
What is the difference between silicon and silicon?
1. What is silicone? 1 Silicon#N#Silicone vs silicon, many take the two as one thing, because they are almost spelled the same and sound similar. But, in fact, they are two very different things, silicone should not be confused with silicon. Silicon (Si) is the fourteenth element on the periodic table, which is a metalloid, meaning it has the properties of both metal and nonmetal, and it is the second most abundant element in the earth’s crust, follow oxygen. Silicon is most commonly combined with oxygen in the form of silicon dioxide or silica, it doesn’t occur in it’s pure form in nature. 2 Silica#N#Silica, also known as silicon dioxide, it is reduced to produce silicon metal, which is reacted with other compounds to produce silicones. Found in nature as quartz, silica or silicon dioxide is the most common component of beach sand, that’s why we say silicones are made of sands. 3 Silicone#N#Silicone also known as siloxanes, is a man-made polymer made up of silicon, oxygen, carbon and hydrogen. Silicones are produced in forms including silicone fluids, resins, liquid or flexible rubber like solid silicone.
What is silicone used for?
It is consistently marketed as inert and versatile, thus silicones have many uses in a variety of industries and consumer products, silicone cookware is one of the most popular application, silicone bake mold, silicone ice tray, silicone fermentation lids, etc. 1.
What is the most common component of beach sand?
Found in nature as quartz, silica or silicon dioxide is the most common component of beach sand, that’s why we say silicones are made of sands. Silicone. Silicone also known as siloxanes, is a man-made polymer made up of silicon, oxygen, carbon and hydrogen.
What is the reaction between silica and carbon?
Silica is put into a silicon furnace at high temperature to drive the oxygen out of silica, the reaction between silica and carbon produce silicon. The powdered silicon then combined with methyl chloride and heated once again, forming methysilane, phenylsiane, vinylsilane and fluoroalkylsilane, which after further reaction and refinement, made the silicones including fluids, rubbers, liquid rubbers, resins and silanes.
When did silicone start environmental monitoring?
But the silicone industry, independent scientific panels and government authorities in several world regions initiated environmental monitoring program in 2016, had produced a robust data demonstrates that:
Can silicone be recycled?
Wasted silicone products can be recycled and process ed back into silicone oil used as lubricants, thermic fluid oil or hydraulic fluids.
Is silicon a metalloid?
Silicon (Si) is the fourteenth element on the periodic table, which is a metalloid, meaning it has the properties of both metal and nonmetal, and it is the second most abundant element in the earth’s crust, follow oxygen.
What is silicone oil?
A liquid polymerized siloxane, with organic side chains, can be termed as silicone oil. Silicone oil consists of alternating atoms of silicone and oxygen (..Si-O-Si-O-Si..). This odorless oil is nonflammable and nontoxic. Due to its lubricating properties and magnificent thermal stability, this oil is used in several industries, ...
Does silicone oil cause irritation?
But it can cause mild irritation, temporarily, if it comes in contact with the eyes. ✦ Silicone oil has a low vapor pressure, i.e., 5 mm Hg at 20°C. ✦ It has anti-foaming properties and is widely used in the food service industry. ✦ Silicone oil has a boiling point that is greater than 140°C/0.002 mmHg (lit.).
Does silicone oil help with skin?
✦ It is used in ointments and skin creams. As silicone oil is a water repellent, it prevents the skin from drying by forming a protective layer across the skin, but it does allows the skin to breathe.
What is silicone made of?
Silicones are a family of polymers containing silicon, hydrogen, and oxygen ( Figure 9.10 ). Unlike other polymers, this product family has silicon and not carbon along the main chain. The pendant side groups can be aliphatic, aromatic, or fluorinated. Most commercially available silicones contain methyl groups and are called polydimethylsiloxanes. Silicones are also known as siloxanes, polyorganosiloxanes, or polysiloxanes.
What is the role of silicone oil in vitreous fluid?
Silicone oils act as a good tamponade against the effusion of subretinal fluid in to the vitreous cavity and support the detached retina onto the choroid.
What is the name of the liquid that is used to treat gas?
Dimeticone (polydimethyl siloxane) is a fluid silicone which, when mixed with silicon dioxide, is known as activated dimeticone (dimethicone) or simeticone (simethicone) and is used therapeutically as an antifoaming or antiflatulent agent. Administered orally as an emulsion or liquid, activated dimeticone relieves excess gastrointestinal gas in conditions such as infantile colic and wind pains Berstein and Kasich (1974). It is also used as an antifoaming agent in radiography, gastroscopy Bertoni et al (1992) and anorectal endoluminal ultrasonography de la Portilla et al (2003) to enhance visualization. Chemically inert, dimeticones have no known systemic action.
What is the name of the compound that is used to make antifoam?
Dimeticone (polydimethyl siloxane) is a fluid silicone which, when mixed with silicon dioxide, is known as activated dimeticone (dimethicone) or simeticone (simethicone) and is used therapeutically as an antifoaming or antiflatulent agent.
How to sterilize silicone?
Silicones can be sterilized by steam, autoclave, EtO and gamma and e-beam radiation ( Table 9.13, Section 9.4.4 ). Figure 9.13 shows the effect of autoclave, EtO, and gamma radiation sterilization on a silicone rubber. Over 90% of the properties are retained with all forms of sterilization [8]. When sterilized with EtO, sufficient time (∼24 h) must be given to aerate the material or device to remove any residual EtO. High doses of gamma radiation (10–100 kGy) will cross-link polydimethylsiloxanes via radical formation at the methyl groups. This may result in a decrease in flexibility and an increase in stiffness and hardness.
How to make crosslinked silicone?
Cross-linked silicone elastomers or resins can be produced by two methods ( Figure 9.12 ). Adding a trichlorosilane (or triacetoxy silane) will produce a cross-linked material during hydrolysis ( Figure 9.12 a). Alternatively, the vinyl-containing fluid can be cross-linked via the mechanism shown in Figure 9.12b.
How many monomer units are in a silicon fluid?
Silicone fluids—have a repeat unit of less than 3000 monomer units.
What is silicone fluid?
The silicone fluids form a range of colourless liquids with viscosities from 1 to 1 000 000 centistokes. High molecular weight materials also exist but these may be more conveniently considered as gums and rubbers (see Section 29.6). It is conveinient to consider the fluids in two classes:
Why are silicones used as lubricants?
Silicone fluids, or oils as they are sometimes known, have been used for many decades as excellent lubricants for polymers and rubbers because, by nature, they have very low coefficients of friction. The viscosity of silicones is much less dependent on temperature compared to petroleum oils. In addition, silicones are more stable to heat oxidation and chemical attack, and are capable of providing anti-corrosion protection for metal surfaces. They can be used in their pure form or alternatively they can be processed to make grease, or combined with water to make oil in water silicone emulsions. These lubricants can be used at the interface between most rubbers and polymers, and when metals are in contact with rubber or plastic. Furthermore, silicones were successfully introduced as an internal lubricant in polymeric materials.
Why is silicone fluid used in a variety of applications?
Silicone fluids find a very wide variety of applications mainly because of their water-repellency, anti-stick properties, low surface tension and thermal properties.
What oil is used to lubricate lycra?
Silicone Oil. Silicone oil used to lubricate lycra during knitting has a tendency to redeposit onto the fabric during rinsing, producing random dye resist marks. From: Fundamentals and Practices in Colouration of Textiles, 2010. Download as PDF.
How are greases made?
Greases may be made by blending the polymer with an inert filler such as a fine silica, carbon black or metallic soap. The silicone–silica greases are used primarily as electrical greases for such applications as aircraft and car ignition systems.
Is silicone a water repellent?
The silicones have established their value as water-repellent finishes for a range of natural and synthetic textiles. A number of techniques have been devised which result in the pick-up of 1–3% of silicone resin on the cloth. The polymer may be added as a solution, an emulsion or by spraying a fine mist; alternatively, intermediates may be added which either polymerise in situor attach themselves to the fibre molecules.
Is silicone good for metal?
In addition, silicones are more stable to heat oxidation and chemical attack, and are capable of providing anti-corrosion protection for metal surfaces. They can be used in their pure form or alternatively they can be processed to make grease, or combined with water to make oil in water silicone emulsions.
What are silicones made of?
More precisely called polymerized siloxanes or polysiloxanes, silicones consist of an inorganic silicon–oxygen backbone chain (⋯−Si−O−Si−O−Si−O−⋯) with two organic groups attached to each silicon center. Commonly, the organic groups are methyl. The materials can be cyclic or polymeric. By varying the −Si−O− chain lengths, side groups, and crosslinking, silicones can be synthesized with a wide variety of properties and compositions. They can vary in consistency from liquid to gel to rubber to hard plastic. The most common siloxane is linear polydimethylsiloxane (PDMS), a silicone oil. The second-largest group of silicone materials is based on silicone resins, which are formed by branched and cage-like oligosiloxanes.
What is the chemical structure of silicone?
Chemical structure of the silicone polydimethylsiloxane (PDMS) More precisely called polymerized siloxanes or polysiloxanes, silicones consist of an inorganic silicon–oxygen backbone chain (⋯−Si−O−Si−O−Si−O−⋯) with two organic groups attached to each silicon center. Commonly, the organic groups are methyl. The materials can be cyclic or polymeric.
What are the properties of silicon?
Silicones exhibit many useful characteristics, including: 1 Low thermal conductivity 2 Low chemical reactivity 3 Low toxicity 4 Thermal stability (constancy of properties over a wide temperature range of −100 to 250 °C) 5 The ability to repel water and form watertight seals. 6 Does not stick to many substrates, but adheres very well to others, e.g. glass 7 Does not support microbiological growth 8 Resistance to oxygen, ozone, and ultraviolet (UV) light. This property has led to the widespread use of silicones in the construction industry (e.g. coatings, fire protection, glazing seals) and the automotive industry (external gaskets, external trim). 9 Electrical insulation properties. Because silicone can be formulated to be electrically insulative or conductive, it is suitable for a wide range of electrical applications. 10 High gas permeability: at room temperature (25 °C), the permeability of silicone rubber for such gases as oxygen is approximately 400 times that of butyl rubber, making silicone useful for medical applications in which increased aeration is desired. Conversely, silicone rubbers cannot be used where gas-tight seals are necessary such as seals for high-pressure gasses or high vacuum.
What is the difference between silicone and benzophenone?
Kipping coined the word silicone in 1901 to describe the formula of polydiphenylsiloxane, Ph 2 SiO (Ph denoting phenyl, C 6 H 5 ), by analogy with the formula of the ketone benzophenone, Ph 2 CO (his term was originally silicoketone ). Kipping was well aware that polydiphenylsiloxane is polymeric whereas benzophenone is monomeric and noted the contrasting properties of Ph 2 SiO and Ph 2 CO. The discovery of the structural differences between Kipping's molecules and the ketones means that silicone is no longer the correct term (though it remains in common usage) and that the term siloxane is preferred according to the nomenclature of modern chemistry.
Why are electronics encased in silicone?
Electronic components are sometimes encased in silicone to increase stability against mechanical and electrical shock, radiation and vibration, a process called "potting". Silicones are used where durability and high performance are demanded of components under hard conditions, such as in space (satellite technology).
What is silicone caulk used for?
Silicone caulk can be used as a basic sealant against water and air penetration.
Why is silicon used in aerospace?
Silicone is a widely used material in the aerospace industry due to its sealing properties, stability across an extreme temperature range, durability, sound dampening and anti-vibration qualities, and naturally flame retardant properties. Maintaining extreme functionality is paramount for passenger safety in the aerospace industry, so each component on an aircraft requires high-performance materials.
What is Silicone?
The plastics industry considers silicone a plastic, and so do we, regardless of much of the green marketing claiming it is not a plastic.
What is the backbone of silicone?
Thus, while most plastics have a polymer backbone of hydrogen and carbon, silicones have a backbone made of silicon and oxygen, and hydrocarbon side groups - all of which gives them plastic-like characteristics.
How long does it take for siloxanes to release into milk?
One study tested the release of siloxanes from silicone nipples and bakeware into milk, baby formula and a simulant solution of alcohol and water. Nothing was released into the milk or formula after six hours, but after 72 hours in the alcohol solution several siloxanes were detected.
Is silicone made of sand?
Silica: When people say silicones are made of sand, they are not incorrect, though that’s too simplistic a description. Silica—or silicon dioxide—is what they are referring to. Silica is the raw material used to make silicone resins. Beach sand is practically pure silica, as is quartz.
Is silicone a good material for heating food?
While silicone is durable and has a high temperature resistance, it makes us queasy to be heating food to very high temperatures in a material like silicone which has been shown to leach chemicals and is not completely inert and stable.
Is silicone a good seal?
Silicone has become a standard high quality seal for products requiring an airtight watertight seal, and a suitable alternative has not yet become available. Natural rubber can be a good alternative for things like soothers and bottle nipples, as long as there is no risk of rubber allergy.
Is silicone cookware safe?
Scientific American reports that in 1979 the US Food and Drug Administration determined silicon dioxides—the raw material for silicone products— were safe for food-grade applications. However, the first silicone cookware only appeared a decade later (e.g., spatulas) and no follow-up studies were done to assess whether silicone cookware leaches anything potentially harmful.
How does silicone oil work?
The silicone oil rises to the surface, bringing colors with it and creating movement in the paint, which creates cells. Typically torching leads to lots of small cells, rather than fewer, larger cells.
What is the first ingredient in silicone lubricant?
Look for silicone-based lube, or products where dimethicone is used instead. Just make sure that silicone or dimethicone is the first or only ingredient.
What is the best lubricant for treadmill belts?
Liquid silicone oils are what the vast majority of us use to create stunning cells. We recommend treadmill belt lubricant because it’s 100 percent silicone oil with nothing else added. It’s completely clear, doesn’t smell bad like the sprays, and usually comes in a convenient dropper bottle, making it easy to dispense the right amount into your paints.
What oil is used to make cells for paint?
Silicone oil: Works well to make cells for most paints and applications. B’laster :Alternative; to be used in a pinch. Liquid Wrench: Spray lubricant; to be used in a pinch. WD40 : Spray lubricant; to be used in a pinch. CRC Heavy Duty : Spray lubricant; to be used in a pinch.
What is the best material to use for acrylic pouring?
While there are lots of different factors that make up a great piece of art (color, texture, finish, etc.), for acrylic pouring, no single aesthetic feature is as sought-after as cells. Silicone, as most experienced pour artists will tell you, is the key to creating dynamic cells in your acrylic pour paintings. While technique can certainly make a difference, it mostly comes down to using the right materials. You need to use the right mix of paints, mediums, and additives—specifically silicone —to create a space for cells to form.
What is silicone oil used for?
You can certainly find other products in this category, since silicone oil is used to maintain everything from locks to sewing machines. Just be sure to choose 100 percent silicone.
What is the purpose of a torch for silicone?
Most folks who use silicone also make use of a culinary torch, to help finish up the cell-making process. When you run the torch lightly over the surface of your painting it serves two functions:
