How to find the freezing point on a phase diagram?
By the end of this section, you will be able to:
- Explain the construction and use of a typical phase diagram
- Use phase diagrams to identify stable phases at given temperatures and pressures, and to describe phase transitions resulting from changes in these properties
- Describe the supercritical fluid phase of matter
What is the importance of phase diagram?
Phase diagrams can be used to predict the phase changes that have occurred in an alloy that has been exposed to a particular heat treatment process. This is important because the properties of a metal component depend on the phases present in the metal.
Which point on the phase diagram represents a boiling point?
The line from A to B is the vapor-pressure curve of the liquid. It represents the equilibrium between the liquid and gas phases. The point on this curve where the vapor pressure is 1 atm is the normal boiling point of the substance. Click to explore further. Simply so, what is normal melting point on a phase diagram?
How can I determine the type of the critical point?
Types of Critical Points
- A critical point is a local maximum if the function changes from increasing to decreasing at that point and is a local minimum if the function changes from decreasing to ...
- A critical point is an inflection point if the function changes concavity at that point.
- A critical point may be neither. ...
.PNG)
What is critical point in phase rule?
The critical point (on a phase diagram) can only exist at one temperature and pressure for a substance or system and thus the degrees of freedom at any critical point is zero.
How do you find the critical point on a phase diagram?
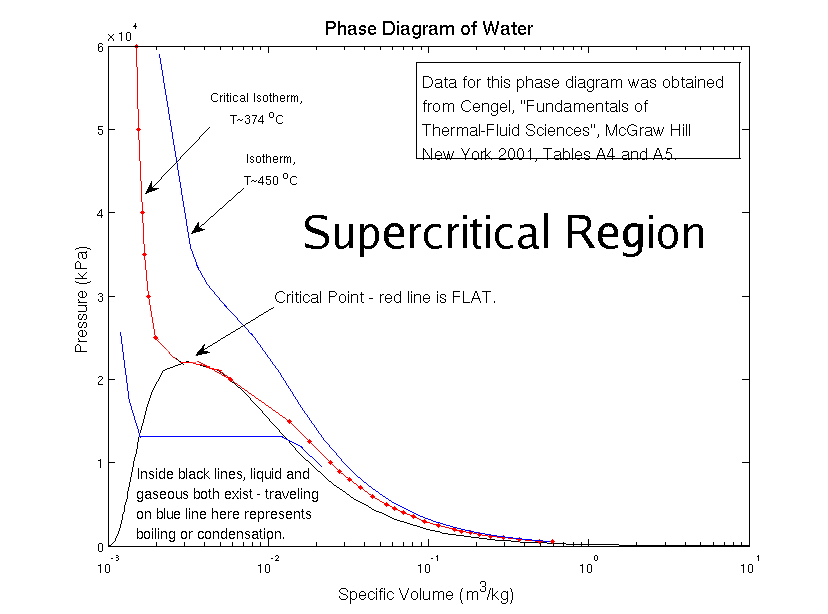
There is only one critical point on a phase diagram. It can be found at the end of the equilibrium line between liquid and gas. This is the point that once passed, the substance becomes a supercritical fluid.
What does the critical point represent?
The critical point is the highest temperature and pressure at which a pure material can exist in vapor/liquid equilibrium. At temperatures higher than the critical temperature, the substance can not exist as a liquid, no matter what the pressure.
How do you find critical points?
To find the critical points of a function y = f(x), just find x-values where the derivative f'(x) = 0 and also the x-values where f'(x) is not defined. These would give the x-values of the critical points and by substituting each of them in y = f(x) will give the y-values of the critical points.
What is critical point and stationary point?
Critical point means where the derivative of the function is either zero or nonzero, while the stationary point means the derivative of the function is zero only.
Is critical point same as triple point?
0:512:15What is the Difference Between Critical Point and Triple Point | HeatYouTubeStart of suggested clipEnd of suggested clipMeet is called digital Point. An example is the liquid vapor critical point the endpoint of theMoreMeet is called digital Point. An example is the liquid vapor critical point the endpoint of the pressure temperature curve that designates conditions under which a liquid and its vapor can coexist.
What is critical point in phase diagram?
What is a critical point in a phase diagram? In thermodynamics, a critical point (or critical state) is the end point of a phase equilibrium curve. The most prominent example is the liquid-vapor critical point, the end point of the pressure-temperature curve that designates conditions under which a liquid and its vapor can coexist.
What is critical point?
A critical point (or critical state) is the end point of a phase equilibrium . Triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium . Correspondingly, what happens at critical point?
What is a critical point in math?
A critical point of a continuous function f is a point at which the derivative is zero or undefined. Critical points are the points on the graph where the function's rate of change is altered—either a change from increasing to decreasing, in concavity, or in some unpredictable fashion.
Where is the critical point?
When dealing with functions of a real variable, a critical point is a point in the domain of the function where the function is either not differentiable or the derivative is equal to zero.
What occurs at the triple point?
In thermodynamics, the triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium. For example, the triple point of mercury occurs at a temperature of −38.83440 °C and a pressure of 0.2 mPa.
What makes something a critical point?
Points on the graph of a function where the derivative is zero or the derivative does not exist are important to consider in many application problems of the derivative. The point ( x, f (x)) is called a critical point of f (x) if x is in the domain of the function and either f′ (x) = 0 or f′ (x) does not exist.
How is boiling point affected by pressure?
The boiling point is reached when the vapor pressure of a liquid matches the atmospheric pressure. Raising the atmospheric pressure will raise the boiling point. Conversely, lowering the atmospheric pressure will lower the boiling point of the liquid.
What is critical volume?
Critical volume is the volume of a fixed mass of a fluid at critical temperature and pressure.
How do you find the triple point?
Sometimes only the term triple point is used, which refers to both the temperature and the pressure at which the three phases co-exist in equilibrium. The triple point of water is 273.16 K at 611.73 Pa. This is the basis of the Kelvin scale and it is equal to 0.01 оC, which is the freezing point of water.
What is a critical point in ethane?
Supercritical ethane, fluid. In thermodynamics, a critical point (or critical state) is the end point of a phase equilibrium curve. The most prominent example is the liquid–vapor critical point, the end point of the pressure–temperature curve that designates conditions under which a liquid and its vapor can coexist.
Who discovered the critical point?
The existence of a critical point was first discovered by Charles Cagniard de la Tour in 1822 and named by Dmitri Mendeleev in 1860 and Thomas Andrews in 1869. Cagniard showed that CO 2 could be liquefied at 31 °C at a pressure of 73 atm, but not at a slightly higher temperature, even under pressures as high as 3000 atm.
What is the critical isotherm of matter?
Above the critical point there exists a state of matter that is continuously connected with (can be transformed without phase transition into) both the liquid and the gaseous state. It is called supercritical fluid.
Does van der Waals hold near critical point?
However, the van der Waals equation, based on a mean-field theory, does not hold near the critical point. In particular, it predicts wrong scaling laws . To analyse properties of fluids near the critical point, reduced state variables are sometimes defined relative to the critical properties.
What is critical point?
Critical point, in physics, the set of conditions under which a liquid and its vapour become identical ( see phase diagram ). For each substance, the conditions defining the critical point are the critical temperature, the critical pressure, and the critical density. This is best understood by observing a simple experiment.
What is phase diagram?
Phase diagram, graph showing the limiting conditions for solid, liquid, and gaseous phases of a single substance or of a mixture of substances while undergoing changes in pressure and temperature or in some other combination of variables, such as solubility and temperature. The Figure shows a typical phase diagram for…
What happens to the density of liquid at the critical point?
The liquid expands and becomes less dense until, at the critical point, the densities of liquid and vapour become equal, eliminating the boundary between the two phases. If the average density at the start is too low, all the liquid will evaporate before the critical temperature is reached. If the initial average density is too high, ...
What is the critical point of a phase diagram?
The critical point on the phase diagram shows where the gas and liquid states of a liquid are identical and the substance is in one phase. Above the critical point, a substance is a supercritical fluid, where the gas and liquid phase of a substance are indistinguishable. Learning Outcomes.
What does the x axis of a phase diagram show?
The x -axis of the graph shows temperature. As temperature increases, most substances change from solid to liquid and then to gas. The y -axis shows pressure. Look at the three solid lines on the graph. These are the phase equilibrium lines. They are the lines on a phase diagram where two phases are in equilibrium.
What is phase equilibrium?
This is a state where the gas and liquid phase of a substance are indistinguishable. Lesson Summary. A phase diagram is a graph of the physical state of a substance (solid, liquid or gas) and the temperature and pressure of the substance. There are three solid lines on the phase diagram that are called the phase equilibrium lines.
What is the line on a phase diagram?
They are the lines on a phase diagram where two phases are in equilibrium. One line shows the equilibrium point between solid and liquid. One shows the equilibrium point between liquid and gas. The third shows the equilibrium point between solid and gas. Yes - solid and gas.
What is triple point?
This triple point is the point where the temperature and pressure conditions are right for all three states (solid, liquid and gas) to exist together at equilibrium. The triple point is like the peak of a pyramid.
How many phases of a substance can be indistinguishable from each other?
The three phases of a substance can sometimes be indistinguishable from each other. Look at the line on this graph separating liquid from gas: At the critical point, the gas and liquid states are identical. As the temperature increases, the substance reaches a critical point.
How to describe a phase diagram?
By the end of this section, you will be able to: 1 Explain the construction and use of a typical phase diagram 2 Use phase diagrams to identify stable phases at given temperatures and pressures, and to describe phase transitions resulting from changes in these properties 3 Describe the supercritical fluid phase of matter
What is phase diagram?
The temperature and pressure conditions at which a substance exists in solid, liquid, and gaseous states are summarized in a phase diagram for that substance . Phase diagrams are combined plots of three pressure-temperature equilibrium curves: solid-liquid, liquid-gas, and solid-gas.
What temperature does the liquid vapor curve end?
Notice that the liquid-vapor curve terminates at a temperature of 374 °C and a pressure of 218 atm, indicating that water cannot exist as a liquid above this temperature, regardless of the pressure. The physical properties of water under these conditions are intermediate between those of its liquid and gaseous phases.
What is triple point?
triple point. temperature and pressure at which the vapor, liquid, and solid phases of a substance are in equilibrium. Previous: Phase Transitions.
How many phases does carbon have?
Elemental carbon has one gas phase, one liquid phase, and two different solid phases, as shown in the phase diagram: (a) On the phase diagram, label the gas and liquid regions. (b) Graphite is the most stable phase of carbon at normal conditions. On the phase diagram, label the graphite phase.
What does the point of intersection of all three curves represent?
The point of intersection of all three curves represents the substance’s triple point —the temperature and pressure at which all three phases are in equilibrium. At pressures below the triple point, a substance cannot exist in the liquid state, regardless of its temperature.
Overview
Mixtures: liquid–liquid critical point
The liquid–liquid critical point of a solution, which occurs at the critical solution temperature, occurs at the limit of the two-phase region of the phase diagram. In other words, it is the point at which an infinitesimal change in some thermodynamic variable (such as temperature or pressure) leads to separation of the mixture into two distinct liquid phases, as shown in the polymer–solven…
Liquid–vapor critical point
For simplicity and clarity, the generic notion of critical point is best introduced by discussing a specific example, the vapor-liquid critical point. This was the first critical point to be discovered, and it is still the best known and most studied one.
The figure to the right shows the schematic PT diagram of a pure substance (a…
See also
• Conformal field theory
• Critical exponents
• Critical phenomena (more advanced article)
• Critical points of the elements (data page)
Footnotes
1. ^ Horstmann, Sven (2000). Theoretische und experimentelle Untersuchungen zum Hochdruckphasengleichgewichtsverhalten fluider Stoffgemische für die Erweiterung der PSRK-Gruppenbeitragszustandsgleichung [Theoretical and experimental investigations of the high-pressure phase equilibrium behavior of fluid mixtures for the expansion of the PSRK group contribution equation of state] (Ph.D.) (in German). Oldenburg, Germany: Carl-von-Ossietzky Univ…
External links
• "Critical points for some common solvents". ProSciTech. Archived from the original on 2008-01-31.
• "Critical Temperature and Pressure". Department of Chemistry. Purdue University. Retrieved 2006-12-03.