What is the half life of iridium 192?
Iridium-192 (symbol 192 Ir) is a radioactive isotope of iridium, with a half-life of 73.83 days. It decays by emitting beta (β) particles and gamma (γ) radiation.
What is Iridium 192 used for in medicine?
It is also used in radiotherapy as a radiation source, in particular in brachytherapy . Iridium-192 has accounted for the majority of cases tracked by the U.S Nuclear Regulatory Commission in which radioactive materials have gone missing in quantities large enough to make a dirty bomb.
What are the isotope properties of iridium?
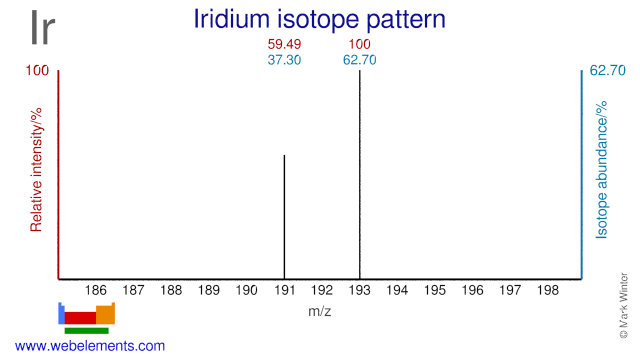
Iridium: isotope data. Available iridium properties... Iridium has two isotopes, Ir-191 and Ir-193 and both are used in the production of radioactive material. Ir-191 is used for the production of radioactive Ir-192. This Ir-192 is used as a radiation source in gamma cameras that are used for non-destructive testing.
How do you make iridium 192?
Iridium-192 is normally produced by neutron activation of natural-abundance iridium metal. Iridium-192 is a very strong gamma ray emitter, with a gamma dose-constant of approximately 1.54 μSv ·h −1 · MBq −1 at 30 cm, and a specific activity of 341 TBq ·g −1 (9.22 kCi ·g −1 ).

What is the isotope iridium-192 used for?
Ir-192 is used in industrial gauges that inspect welding seams and in medicine to treat certain cancers. Where does it come from? Ir-192 is a manmade radioactive element that is formed from nonradioactive iridium metal in a nuclear reactor.
What is the T 1/2 of iridium-192?
Isotopes of iridiumIsotopeabundancehalf-life (t1/2)191Ir37.3%stable192Irsyn73.827 d192m2Irsyn241 y8 more rows
What is the atomic number of iridium-192?
Iridium is a chemical element with the symbol Ir and atomic number 77.
Does iridium have any isotopes?
Naturally occurring iridium has two stable isotopes.
What does Ir-192 decay into?
platinum 192Ir decays into platinum 192 (192Pt) via β− decay 95.1% of the time and the remaining 4.9% in osmium 192 (192Os) by EC. This leads to a complex decay pattern resulting in 29 gamma emission peaks from 0.110 to 1.378 MeV, various characteristic x-rays, and numerous electrons up to 1.377 MeV.
How do you make iridium-192?
Iridium-192 does not typically occur naturally. Instead, scientists must put iridium-191 in a nuclear reactor and bombard it with neutrons. The iridium-191 then takes up an extra neutron to become iridium-192.
What is the chemical symbol of iridium?
IrIridium / Symboliridium (Ir), chemical element, one of the platinum metals of Groups 8–10 (VIIIb), Periods 5 and 6, of the periodic table. It is very dense and rare and is used in platinum alloys.
Who discovered Ir-192?
The physicist Enrico Fermi was born in Rome, Italy. In 1926 Fermi discovered the statistical laws governing the behaviour of particles of quantum spin one half, which are now known as fermions.
What are 3 interesting facts about iridium?
Just the factsAtomic number (number of protons in its nucleus): 77.Atomic symbol (on the Periodic Table of Elements): Ir.Atomic weight (average mass of the atom): 192.217.Melting point: 4,435 F (2,446 C)Boiling point: 8,002.4 F (4,428 C)More items...•
Is iridium a rare Earth metal?
Iridium is one of the rarest elements in the earth's crust.
Is iridium harmful to humans?
Health effects of iridium Highly flammable. Potential health effects: Eye: may cause eye irritation. Skin: low hazard for usual industrial handling. Ingestion: may cause irritation of the digestive tract.
What is iridium worth?
The average annual price for iridium in 2020 was 2,550 U.S. dollars per troy ounce. The price of iridium dramatically increased in March 2021, to 6,000 U.S. dollars per troy ounce as a result of supply shortages combined with its prospective use to produce green hydrogen.
What is IR 192?
Ir-192 is a manmade radioactive element that is formed from nonradioactive iridium metal in a nuclear reactor.
What is the reaction of ir-192?
Metallic Ir-192 will react with fluorine gas to form iridium fluoride (IV), IrF 6.
Can IR 192 cause cancer?
Exposure to Ir -192 can increase the risk for cancer because of its high-energy gamma radiation. External exposure to Ir-192 can cause burns, acute radiation sickness, and even death. Internal exposure could occur only if a person were to swallow one of the Ir-192 seeds or pellets. Internal exposure from Ir-192 could cause burns in ...
How strong is iridium 192?
Iridium-192 is a very strong gamma ray emitter, with a gamma dose-constant of approximately 1.54 μSv ·h −1 · MBq −1 at 30 cm, and a specific activity of 341 TBq ·g −1 (9.22 kCi ·g −1 ). There are seven principal energy packets produced during its disintegration process ranging from just over 0.2 to about 0.6 MeV .
What is iridium 192 used for?
Iridium-192 is commonly used as a gamma ray source in industrial radiography to locate flaws in metal components. It is also used in radiotherapy as a radiation source, in particular in brachytherapy .
How does iridium 192 decay?
Iridium-192 (symbol 192 Ir) is a radioactive isotope of iridium, with a half-life of 73.83 days. It decays by emitting beta (β) particles and gamma (γ) radiation. About 96% of 192 Ir decays occur via emission of β and γ radiation, leading to 192 Pt. Some of the β particles are captured by other 192 Ir nuclei, which are then converted to 192 Os. Electron capture is responsible for the remaining 4% of 192 Ir decays. Iridium-192 is normally produced by neutron activation of natural-abundance iridium metal.
How many isotopes of iridium are there?
There are two natural isotopes of iridium ( 77 Ir), and 34 radioisotopes, the most stable radioisotope being 192 Ir with a half-life of 73.83 days, and many nuclear isomers, the most stable of which is 192m2 Ir with a half-life of 241 years. All other isomers have half-lives under a year, most under a day.
What is iridium 192?
Overview. Iridium-192 () is the most common isotope used in high dose rate (HDR) brachytherapy. It is a man-made isotope produced by neutron bombardment of Iridium-191. decays primarily through decay to an excited state of Platinum-192 which is stable. Approximately 5% of decays occur via electron capture yielding an excited state ...
Which is better, Osmium 192 or Cesium 137?
It is regarded as superior to cobalt-60 and cesium-137 due to it’s higher specific activity which allows for smaller source sizes.
What is the iridium isotope used for?
Iridium has two isotopes, Ir-191 and Ir-193 and both are used in the production of radioactive material. Ir-191 is used for the production of radioactive Ir-192. This Ir-192 is used as a radiation source in gamma cameras that are used for non-destructive testing.
When was Mason in multinuclear NMR?
J. Mason in Multinuclear NMR, Plenum Press, New York, USA, 1987. Where given, data for certain radioactive nuclei are from this reference.
Can Ir-191 be used as an enriched radioisotope?
Although Ir-192 can be produced from natural Ir, the use of enriched Ir-191 gives a much higher specific activity and allows the use of smaller sources. The use of Ir-193 has been suggested for the production of the therapeutic radioisotope Pt-195m.