Table of Contents
| Types of Chemical Reactions | Explanation | General Reaction |
| Combination reaction | Two or more compounds combine to form on ... | A + B → AB |
| Decomposition reaction | The opposite of a combination reaction – ... | AB → A + B |
| Precipitation reaction | Two solutions of soluble salts are mixed ... | A + Soluble salt B → Precipitate + solub ... |
| Neutralization reaction | An acid and a base react with each other ... | Acid + Base → Salt + Water |
Full Answer
What are the 5 different chemical reactions?
What are the 6 types of chemical reactions?
- synthesis reaction.
- decomposition reaction.
- single displacement reaction.
- double displacement reaction.
- combustion reaction.
- acid-base reaction.
How to identify chemical reactions?
Method 6 Method 6 of 6: Recognizing a Reaction through Observation Download Article
- Feel for heat in exothermic reactions. Many synthesis and replacement (single and double) reactions are exothermic, meaning they release heat.
- Look for the formation of precipitate. Again, in many synthesis and replacement (single and double) reactions, a precipitate will form at the bottom of the tube.
- Add heat for endothermic reactions. ...
What are the different reactions in chemistry?
Types of Reaction Mechanisms in Organic Chemistry
- Sn1 Reactions. Sn1 reactions are substitution reactions that use a weak nucleophile or base. ...
- Sn2 Reactions. Sn2 reactions are also substitution reactions, but they require a strong nucleophile. ...
- E1 Reactions. E1 reactions are elimination reactions that use a weak nucleophile or base. ...
What are the examples of chemical reaction?
Types of Chemical Reactions
- Decomposition reaction
- Combination reaction
- Combustion reaction
- Neutralization reaction
- Single displacement reaction
- Double displacement reaction
- Precipitation reaction
- Redox reaction
- Decomposition Reaction – In a decomposition reaction, molecules or compounds break down into two or more than two simpler chemically new substances. ...

How do you identify a chemical reaction?
A chemical reaction is typically followed by physical signs that are readily detected, such as heat and light emission, precipitate formation, gas...
How do you identify physical and chemical changes?
The shape or form of the matter varies through a physical transition, but the sort of matter in the material does not. In a chemical shift, however...
Why do we write a chemical equation?
The purpose of writing a balanced chemical equation is to explain the occurring reactants (starting material) and products (end results). The ratio...
What is chemical reaction and equation?
A chemical equation is the symbolic representation in the form of symbols and formulas of a chemical reaction in which the reactant entities on the...
What is the skeleton equation?
A skeleton equation is when each product that takes part in the reaction is written with the chemical formulas describing the chemical reaction. Ex...
How many types of chemical reactions are there?
There are hundreds or even thousands of types of chemical reactions! If you are asked to name the main 4, 5 or 6 types of chemical reactions, here is how they are categorized. The main four types of reactions are direct combination, analysis reaction, single displacement, and double displacement. If you're asked the five main types of reactions, it is these four and then either acid-base or redox (depending who you ask). Keep in mind, a specific chemical reaction may fall into more than one category.
What is chemical reaction?
A chemical reaction is a process generally characterized by a chemical change in which the starting materials (reactants) are different from the products. Chemical reactions tend to involve the motion of electrons, leading to the formation and breaking of chemical bonds.
What is the reaction between an acid and a base?
An acid-base reaction is a type of double displacement reaction that occurs between an acid and a base. The H + ion in the acid reacts with the OH - ion in the base to form water and an ionic salt: HA + BOH → H 2 O + BA. The reaction between hydrobromic acid (HBr) and sodium hydroxide is an example of an acid-base reaction: ...
What is the reaction of a combustible material?
Combustion. A combustion reaction is a type of redox reaction in which a combustible material combines with an oxidizer to form oxidized products and generate heat ( exothermic reaction ). Usually, in a combustion reaction oxygen combines with another compound to form carbon dioxide and water.
What is an example of a decomposition reaction?
In a decomposition reaction, a compound is broken into smaller chemical species.#N#AB → A + B#N#The electrolysis of water into oxygen and hydrogen gas is an example of a decomposition reaction:#N#2 H 2 O → 2 H 2 + O 2
What is an example of a synthesis reaction?
In a synthesis reaction, two or more chemical species combine to form a more complex product.#N#A + B → AB#N#The combination of iron and sulfur to form iron (II) sulfide is an example of a synthesis reaction:#N#8 Fe + S 8 → 8 FeS
What is a double displacement reaction?
Metathesis or Double Displacement Reaction. In a double displacement or metathesis reaction two compounds exchange bonds or ions in order to form different compounds. An example of a double displacement reaction occurs between sodium chloride and silver nitrate to form sodium nitrate and silver chloride.
What are the different types of chemical reactions?
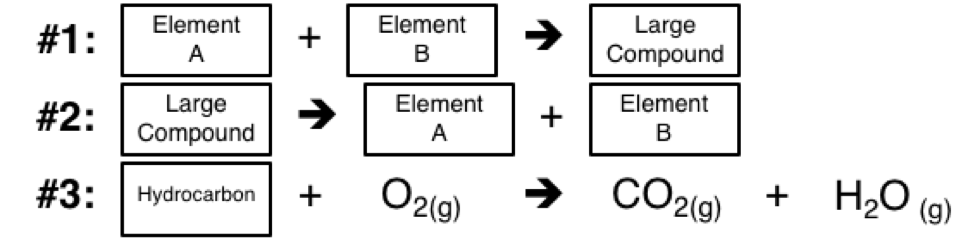
4 Main Types of Chemical Reactions 1 Synthesis reaction (also known as a direct combination reaction)#N#In this reaction, reactants combine to form a more complex product. Often there are two or more reactants with only a single product. The general reaction takes the form:#N#A + B → AB 2 Decomposition reaction (sometimes called an analysis reaction)#N#In this kind of reaction, a molecule breaks into two or more smaller pieces. It's common to have one reactant and multiple products. The general chemical reaction is:#N#AB → A + B 3 Single displacement reaction (also called a single replacement reaction or substitution reaction)#N#In this type of chemical reaction, one reactant ion changes place with another. The general form of the reaction is:#N#A + BC → B + AC 4 Double displacement reaction (also called a double replacement reaction or metathesis reaction)#N#In this type of reaction, both cations and anions exchange places, according to the general reaction:#N#AB + CD → AD + CB
What is a reaction called when reactants combine to form a more complex product?
It's a good idea to become familiar with the various names so that you can identify a reaction and communicate with people who may have learned it under a different name. Synthesis reaction (also known as a direct combination reaction) In this reaction, reactants combine to form a more complex product.
What is the reaction of a molecule that breaks into two or more smaller pieces called?
Decomposition reaction (sometimes called an analysis reaction) In this kind of reaction, a molecule breaks into two or more smaller pieces. It's common to have one reactant and multiple products. The general chemical reaction is: AB → A + B.
Is it a good idea to memorize the main types of chemical reactions?
It's a good idea to memorize the main types of chemical reactions. Trish Gant / Getty Images. Dr. Helmenstine holds a Ph.D. in biomedical sciences and is a science writer, educator, and consultant. She has taught science courses at the high school, college, and graduate levels.
Can a Reaction Be More Than One Type?
As you start adding more and more types of chemical reactions, you'll notice a reaction may fit into multiple categories. For example, a reaction may be both an acid-base reaction and a double displacement reaction.
How many types of chemical reactions are there?
Chemical reactions are of many types. Here, we are covering most common eight types of chemical reactions which are part of class X chemistry syllabus as well.
What are the characteristics of chemical reactions?
Characteristics of a Chemical Reaction 1 Evolution of gas 2 Change in color 3 Change in temperature 4 Change in energy 5 Formation of precipitate 6 Change in state
What is Chemical Reaction?
A chemical reaction is the process in which reactants react chemically and convert into products by chemical transformation. For example – Respiration – we inhale oxygen which reacts with glucose and produces carbon dioxide, water and energy. Reaction is given below –
What chemical reaction is accompanied by the formation of a precipitate?
Formation of Precipitate – Some chemical reactions are accompanied by formation of precipitate. For example, barium chloride reacts with sodium sulphate and forms sodium chloride and precipitate of barium sulfate.
What are some examples of neutralization reactions?
For example, when we burn methane it gives carbon dioxide and water. Neutralization Reaction – In these reactions acid and base reacts with each other and form salt and water. For example, hydrochloric acid reacts with sodium hydroxide (base) and forms sodium chloride (salt) and water.
What is a reactivity series?
Reactivity series is a series in which elements are arranged in decreasing order of their reactivity. It means the elements present at the top of this reactivity series are more reactive than the elements present at the bottom. Reaction of potassium with magnesium chloride is an example of single displacement reaction.
What is the reaction between hydrochloric acid and sodium hydroxide?
For example, hydrochloric acid reacts with sodium hydroxide (base) and forms sodium chloride (salt) and water. Single Displacement Reaction – In these reactions more reactive metal displaces less reactive metal from its salt. In these reactions, products can be determined through reactivity series.
What are the different types of chemical reactions?
This article will cover the main classifications of chemical reactions: synthesis reaction, decomposition reaction, single replacement reaction, and double replacement reaction. We also discuss what is a combustion reaction, precipitation reaction, and acid base reaction.
What is synthesis reaction?
A synthesis reaction occurs when two reactants interact to form one product. The product created is different from both of the reactants. The general equation represents this type of reaction:
What is acid base reaction?
Acid base reactions, or neutralization reactions are double replacement reactions that occur between acids and bases. Typically, acid-base neutralizations produce water and a salt. A common example of neutralization is between hydrochloric acid, a strong acid, sodium hydroxide, and a strong acid.
When does a decomposition reaction occur?
A decomposition reaction occurs when the reactant breaks down into simpler products. Here is the genera l equation that represents this type of reaction:
What is a single replacement reaction?
Single replacement reactions occur when a single element replaces an element in another compound. This produces a new compound and a new element. This type of reaction is represented by the general equation
What is combustion reaction?
Combustion reactions are those that involve the burning of compounds. A reactant, usually a hydrocarbon, reacts with oxygen gas (O 2 ), to produce carbon dioxide gas (CO 2) and water vapor (H 2 O). Combustion reactions also produce energy in the form of heat and/or light.
What is the reaction that separates from the solution called?
These reactions both result in two completely new compounds through double replacement. A precipitation reaction occurs when two soluble compounds mix to form an insoluble solid. The solid that separates from the solution is called the precipitant. A classic example of a precipitation reaction is Silver nitrate’s reaction with Potassium chloride, which forms Silver chloride, a white solid.
How many types of chemical reactions are there?
Based on this rule, chemical reactions are often divided into five common types.
What are some examples of chemical reactions?
Smelting iron, burning fuels, pottery and glass manufacturing, production of cheese and wine, and brewing processes are typical chemical reaction examples. We balance complex chemical equations for getting desired results.
What is a chemical reaction?
A chemical reaction can be defined as a process in which one or more substances undergo a chemical change, such as a change in the structure or positioning of atoms.
What is a combination reaction?
So, whenever you notice a chemical reaction where two elements interact to form a single compound, know that it is a combination reaction. This can be processed by using chemical equation calculator online.
How long have chemical reactions been used?
These processes have been used for thousands of years. There are many conversion or chemical factors are involved in this. These processes force us to think what is a chemical reaction and the types of chemical reactions and how it works? But, to understand the process thoroughly, you first have to learn the ropes.
How to predict the type of reaction?
Answer: Analyze the nature of reactants and products taking part in the reaction. You will be able to predict the types of reaction occurring among them. In some cases, there are even such reactions that fall into more than one category.
What is a reaction called when a new substance is synthesized?
Since a new substance is being synthesized, such reactions are also being referred to as synthesis reactions. To illustrate it further, let's have a look at the general equation of combination reaction.

4 Main Types of Chemical Reactions
5 Main Types of Chemical Reactions
- You simply add one more category: the combustion reaction. The alternate names listed above still apply. 1. synthesis reaction 2. decomposition reaction 3. single displacement reaction 4. double displacement reaction 5. combustion reaction A general form of a combustion reaction is: hydrocarbon + oxygen → carbon dioxide + water
6 Main Types of Chemical Reactions
- The sixth type of chemical reaction is an acid-base reaction. 1. synthesis reaction 2. decomposition reaction 3. single displacement reaction 4. double displacement reaction 5. combustion reaction 6. acid-base reaction
Other Major Categories
- Other main categories of chemical reactions include oxidation-reduction (redox) reactions, isomerization reactions, and hydrolysis reactions.
Can A Reaction Be More Than One Type?
- As you start adding more and more types of chemical reactions, you'll notice a reaction may fit into multiple categories. For example, a reaction may be both an acid-base reaction and a double displacement reaction.
Characteristics of A Chemical Reaction
- Chemical reactions show one or more than one of the following characteristics. 1. Evolution of gas 2. Change in colour 3. Change in temperature 4. Change in energy 5. Formation of precipitate 6. Change in state Now understand these characteristics in detail. 1. Evolution of Gas– Some chemical reactions take place with the evolution of gas. For example, when zinc reacts with hydr…
Types of Chemical Reactions
- Chemical reactions are of many types. Here, we are covering the most common eight types of chemical reactions, which are part of the class X chemistry syllabus as well. 1. Decomposition reaction 2. Combination reaction 3. Combustion reaction 4. Neutralization reaction 5. Single displacement reaction 6. Double displacement reaction 7. Precipitation ...
List of Common Chemical Reactions
- We are giving here a general list of common chemical reactions. This ends our coverage on a list of Various Types of Chemical Reactions. We hope you enjoyed learning and were able to grasp the concepts. We hope after reading this article you will be able to differentiate various reactions easily and it will help you in experiments as well because we have covered almost all-important …