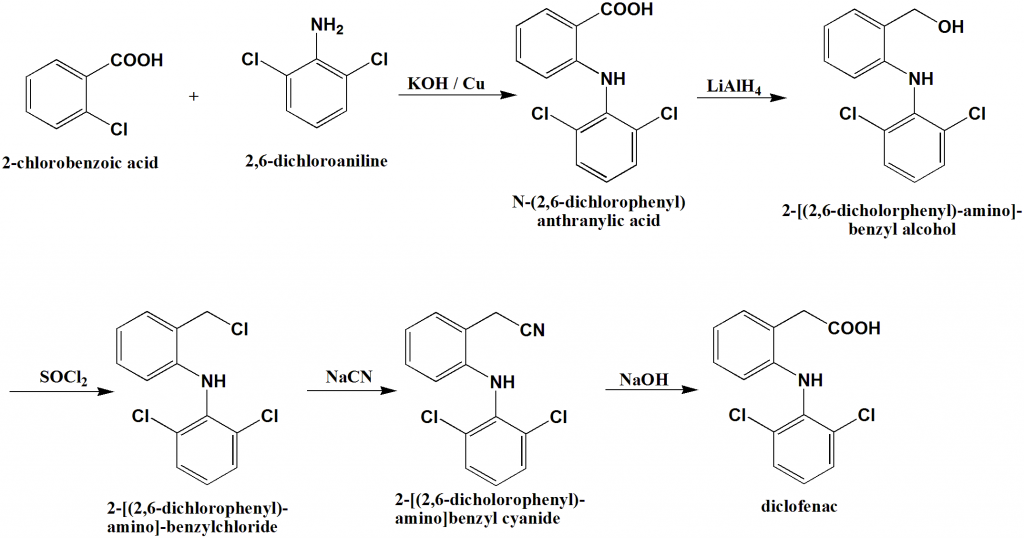
A synthesis reaction or direct combination reaction is one of the most common types of chemical reaction. In a synthesis reaction, two or more chemical species combine to form a more complex product: A + B → AB. In this form, a synthesis reaction is easy to recognize because you have more reactants than products.
Full Answer
What are the reactants and products of synthesis?
Reactants and products are the two major components of a chemical reaction. Reactants are the starting material of a chemical reaction. Products are the chemical species that can be found after the completion of the reaction. There are various types of chemical reactions such as acid-base reactions, redox reactions, and combustion reactions.
Does a synthesis reaction always involve bond formation?
A chemical synthesis usually involves the breaking of existing bonds and the formation of new ones. Synthesis of a complex molecule may involve a considerable number of individual reactions leading in sequence from available starting materials to the desired end product. Each step usually involves reaction at only one chemical bond in the molecule.
What are the common chemical reactions?
Types of Chemical Reactions
- Oxidation-Reduction or Redox Reaction. ...
- Direct Combination or Synthesis Reaction. ...
- Chemical Decomposition or Analysis Reaction. ...
- Single Displacement or Substitution Reaction. ...
- Metathesis or Double Displacement Reaction. ...
- Acid-Base Reaction. ...
- Combustion. ...
- Isomerization. ...
- Hydrolysis Reaction. ...
- The Main Reaction Types. ...
What is a Decomp reaction?
A decomposition reaction is a type of chemical reaction in which one reactant yields two or more products . The general form for a decomposition reaction is: AB → A + B Decomposition reactions are also known as analysis reactions or chemical breakdowns.
What is a synthesis reaction simple definition?
Synthesis reactions are reactions that occur when two different atoms or molecules interact to form a different molecule or compound.
What are some synthesis reactions?
Here are some examples of synthesis reactions:Water: 2 H2(g) + O2(g) → 2 H2O(g)Carbon dioxide: 2 CO(g) + O2(g) → 2CO2(g)Ammonia: 3 H2(g) + N2(g) → 2 NH3(g)Aluminum oxide: 4 Al(s) + 3 O2(g) → 2 Al2O3(s)Iron sulfide: 8 Fe + S8 → 8 FeS.Potassium chloride: 2 K(s) + Cl2(g) → 2 KCl(s)
Why is it called a synthesis reaction?
Answer: Solution: When two or more substances (element or compounds) react together to combine and form a new product, which is a compound with new properties. They synthesize to form a new substance, hence it is called a synthesis reaction.
What is synthesis reaction give two examples?
The formation of water from hydrogen and oxygen.. [ 2 H2(g) + O2(g) → 2 H2O(g) ] Synthesis of Ammonia.. [3 H2(g) + N2(g) → 2 NH3(g) ]
How do you identify a synthesis reaction?
A synthesis reaction occurs when two or more reactants combine to form a single product. This type of reaction is represented by the general equation: A + B → AB. An example of a synthesis reaction is the combination of sodium (Na) and chlorine (Cl) to produce sodium chloride (NaCl).
What is an example of synthesize?
It's simply a matter of making connections or putting things together. We synthesize information naturally to help others see the connections between things. For example, when you report to a friend the things that several other friends have said about a song or movie, you are engaging in synthesis.
What's another name for a synthesis reaction?
Synthesis reactions are also called combination reactions because multiple reactants form a single product.
What are some examples of synthesis reactions in everyday life?
Examples of Synthesis Chemical Reactionshydrogen + oxygen ---> water.iron + oxygen ---> rust.potassium and chlorine gas ---> chloride.lime + carbon dioxide ---> calcium carbonate (used to strengthen masonry)water + carbon dioxide + light ---> glucose and oxygen (photosynthesis)More items...
Is synthesis reaction the same as combination?
→→ Combination reaction is also known as synthesis reaction because in these reactions, two or more substances chemically combine to form a new compound.
How many products are in a synthesis reaction?
Answer and Explanation: There is one product in a synthesis reaction. A synthesis reaction occurs when two or more reactants combine to form a single, more complex molecule (A + B ---> AB).
What types of products does a synthesis reaction always have?
Synthesis reactions always yield one product. Reversing a synthesis reaction will give you a decomposition reaction. This is the most well-known example of a synthesis reaction—the formation of water via the combustion of hydrogen gas and oxygen gas.
What Happens in a Synthesis Reaction?
In a synthesis reaction, two or more chemical species combine, forming a more complex product in the reaction. It is also called a direct reaction and is one of the most common chemical reactions. When the two or more reactants combine they make a larger compound. A synthesis reaction is the opposite of a decomposition reaction, which is when the bonds are broken in a complex product, and it splits the product into its respective components or elements.
What Is a Synthesis Reaction in Organic Chemistry?
Organic synthesis reactions involve organic compounds. Organic molecules are more complex than their inorganic counterparts are. In many cases, because of the complexity, synthesis reactions of organic compounds require several steps one after the other to create a single product. This makes intermediate compounds for each step before the final single product.
What is the reaction of taurine?
It's highly likely if you consumed taurine, which is the result of an organic synthesis reaction and commonly found in milk and eggs. In chemistry, a synthesis reaction is when two or more chemicals combine and form a more complex product. You will also have more reactants than products since two or more chemical species combine to form one new ...
What is the reaction that occurs when combining elements and producing a new compound?
A synthesis reaction can occur when combining elements and producing a new compound, combining compounds to produce a new compound, or combining both elements and compounds to result in a new compound.
What is the opposite of a decomposition reaction?
A synthesis reaction is the opposite of a decomposition reaction, which is when the bonds are broken in a complex product, and it splits the product into its respective components or elements.
What does synthesis mean?
The word synthesis means to put together. When two or more products are put together it produces a new single product. The basic form of the chemical equation is written as: 00:00. 00:00 12:50. GO LIVE. Facebook.
What happens when two non-metals combine?
When two non-metals combine, they produce a covalent compound. When combining metal oxide and water (both compounds), it produces a new compound of a metal hydroxide. Non-metal and water combinations result in an oxy acid compound. Metal oxides and carbon dioxide combined produce metal carbonates.
What is a Synthesis Reaction?
Synthesis reactions are reactions that occur when two different atoms or molecules interact to form a different molecule or compound. Most of the time, when a synthesis reaction occurs, energy is released and the reaction is exothermic. However, an endothermic outcome is also possible. Synthesis reactions are one of the major classes of chemical reactions, which include single displacement, double displacement, and combustion reactions.
What is a successful synthesis reaction?
A successful synthesis reaction maximizes the creation of desired molecules and minimizes byproduct molecules. A thorough understanding of reaction kinetics, mechanism, and effect of reaction variables are keys to successful synthesis reactions.
What are the key stages of a reaction?
Investigate key stages of a reaction or process, such as reaction start, induction period, accumulation, conversion, and endpoint. Detect reaction stalling or upsets
What is a smart chemical synthesis reactor?
Smart chemical synthesis reactors, combined with unattended dosing and automated sampling, provide a simple and safe way to precisely control reaction parameters and obtain reaction information unattended and around the clock.
What is reaction kinetics?
Reaction Kinetics - A thorough understanding of reaction rates are crucial to ensure optimized product yield and minimum byproducts. Through data-rich experiments, ReactIR simplifies and speeds the measurement of kinetic factors in synthesis reactions.
What are intermediate molecules?
Intermediate molecules may form that can lead to the formation of byproducts. In addition, depending on how the two colliding reactant molecules orient, both the desired product and byproducts may form - which may effect product purity. There are various types of synthesis reactions. For example, nucleophilic and electrophilic addition ...
What are the major classes of chemical reactions?
Synthesis reactions are one of the major classes of chemical reactions, which include single displacement, double displacement, and combustion reactions.
What is the hallmark of a synthesis reaction?
The hallmark of a synthesis reaction is that a more complex product is formed from the reactants. One easy-to-recognize type of synthesis reaction occurs when two or more elements combine to form a compound. The other type of synthesis reaction happens when an element and a compound combine to form a new compound.
What are the different types of chemical reactions?
While there are many types of chemical reactions, they all fall into at least one of four broad categories: synthesis reactions, decomposition reactions, single displacement reactions, and double displacement reactions. A synthesis reaction or direct combination reaction is a type of chemical reaction in which two or more simple substances combine ...
How to identify a reaction?
Basically, to identify this reaction, look for a product that contains all the reactant atoms. Be sure to count the number of atoms in both the reactants and the products. Sometimes when a chemical equation is written, "extra" information is given that might make it hard to recognize what is going on in a reaction. Counting numbers and types of atoms makes it easier to identify reaction types.
What is a direct combination reaction?
A synthesis reaction or direct combination reaction is a type of chemical reaction in which two or more simple substances combine to form a more complex product. The reactants may be elements or compounds, while the product is always a compound.
What is a Synthesis Reaction?
Did you know those are great examples of products made from a synthesis reaction? It may seem odd to mix a food term with a science term but it is definitely true. Before we discuss how this is so, let's go over what a synthesis reaction is.
What is the reaction that produces a compound called?
A synthesis reaction is the joining together of two reactants, or compounds, to produce a complex product, also called a compound. Sometimes synthesis reactions can result in the formation of more than one product, as we'll see shortly with the process of photosynthesis.
Why is chlorine a balanced reactant?
Because two molecules of chlorine (Cl 2) are present as a reactant, this needs to be balanced for the product LiCl (where only one chlorine atom is present). The addition of a coefficient of 2 (in front of LiCl) will ensure all chlorine atoms, on both the reactant and product side, are balanced.
Why is rust a combination reaction?
Synthesis reactions are also called combination reactions because multiple reactants form a single product. The carbon in steel is added to toughen the steel and won't prevent the reaction of the iron in the steel with oxygen in the atmosphere. Rust is iron oxide, which is the chemical combination of iron and oxygen.
What is the function of coefficients in synthesis reactions?
As you will see shortly, coefficients, for example numbers, are sometimes required to ensure what is present on the left side of a reaction is equal in amount to what is present on the right side of a reaction.
What is binary compound?
First, binary compounds can be produced from a synthesis reaction. A binary compound is a compound that contains only two different elements. For example, the combination of two reactants, iron and oxygen, will produce the binary compound iron oxide, commonly known as rust.
Is a synthesis reaction a multi step process?
As you can gather from the equation shown in Diagram 1, a synthesis reaction is not a multi-step process. That is, there is no need to be concerned with learning multiple steps for this reaction.
Synthesis Reaction
When performing an experiment, it is common to look and see what type of chemical reaction has occurred. If the two reactants, which can be compounds or elements, react and rearrange to create a new product, then a synthesis reaction has taken place. A synthesis reaction is when the compounds or elements combine and turn into a new compound.
Mechanism of Synthesis Reaction
One of the most basic of reactions is the creating of water. To create water, 2 molecules of hydrogen gas (2H2) combine with 1 molecule of oxygen gas (O2). During the reaction, the molecules break apart and recombine to create 2 molecules of water (2H2O). This process is an example of a synthesis reaction because a new substance has formed.
Types of Synthesis Reactions
There are multiple ways of creating a synthesis reaction. A synthesis reaction can be created by combining two different elements to create a compound. Two different compounds can be combined to create a new compound, or an element and a compound can combine to create a new compound.
Synthesis Reactions
Observing a chemical reaction can be a good way to remember how to categorize a reaction based on the various reaction types. In this activity, we're going to conduct an experiment with a single replacement reaction. We're going to react steel, which is an alloy of iron, with oxygen.
What is a synthesizing reaction?
Synthesis reactions are one of those chemical reactions which are greatly used in the labs for the production of highly essential products. These reactions are very useful for equation balancing process. The balanced chemical equation calculator can help you a lot in learning and calculating equations.
What is synthesis in chemistry?
The synthesis in chemistry refers to how complex chemical entities or compounds are formed from smaller and simpler units. Generally, synthesis reactions can be demonstrated, such as
Why is synthesis important for chemists?
Similarly, synthesis allows chemists to form chemical compounds that don't exist in nature to elevate the living standards and for research purposes.
What is the chemical reaction in which two or more products are formed?
On the other hand, the chemical reactions in which two or more products are formed are called complex synthesis reactions.
What happens when sodium and chlorine react?
Related: Sodium react with chlorine to form sodium chloride NaCl. Learn more about the reaction of sodium and chlorine and what happens when they react with each other.
Why are organic synthesis reactions used?
The organic synthesis reactions are used greatly for the manufacturing of substances required for daily life.
How are chemical compounds formed?
As we know, chemical compounds are formed by the combination of multiple atoms of different elements.
What is Synthesis Reaction?
Synthesis reaction is one type of chemical reaction in which two different atoms involve in the reaction, react with each other to form a totally different molecular compound. In most of the synthesis reaction, energy is released from the reaction medium and known as exothermic reaction.
Synthesis of ethyl methyl ether
Williamson synthesis process is the best method to synthesis ethyl methyl ether (CH 3 -O- CH 2 CH 3 ). This reaction proceeds through SN 2 pathway. To obtain ethyl methyl ether as the synthesized product, sodium methoxide (CH 3 ONa) and ethyl chloride (C 2 H 5 Cl) reacts with each other.
Synthesis of anisole
This ether can also be synthesized by Williamson ether synthesis. To obtain anisole, sodium phenoxide (C 5 H 5 ONa) will react with methyl iodide (CH 3 I) and sodium phenoxide (nucleophile) attacks the electrophilic centre of methyl iodide. Iodide (I –) will be eliminated as it is a good leaving group and anisole is formed.
Synthesis of 2-Ethoxynaphthalene
To proceed this reaction, hydroxyl group should be inserted at the 2 position of naphthalene group and reacts with bromoethane. The reaction medium should be basis. Thus, sodium hydride (NaH) is used. Nucleophilic oxygen atom of OH group in naphthalene attacks the CH 2 centre of CH 3 CH 2 Br and Br – is eliminated as the leaving group.
Synthesis of Phenyl Propyl Ether
To synthesis phenyl propyl ether the reactants that are chosen are phenol, sodium metal and n-propyl bromide. Solvent that is used in this synthesis reaction is a polar aprotic solvent. The first step is to react phenol with sodium to form sodium phenoxide (active nucleophile).
Synthesis of Benzyl-tertbutyl ether
William synthesis pathway is followed for the formation of benzyl-tertbutyl ether. Sodium tert-butoxide and benzyl bromide is taken as the reactants. O – ion from sodium tert-butoxide attacks the electron deficient centre of benzyl bromide Br – is eliminated as the leaving group to form the desired product.
Synthesis of tert-butyl methyl ether
This synthesis process almost similar to the synthesis of benzyl tert-butyl ether. One of the reactants is also same, sodium tert-butoxide and the another reactant is methyl bromide (CH 3 Br). Tertiary sodium tert butoxide reacts as nucleophile and attacks the methyl carbon center to eliminate bromide ion.